“Can I take melatonin with Ozempic” is a common supplement-interaction question for GLP-1 drugs. Drugs.com answers with a one-line “no known interaction.” That is a reasonable narrow interaction answer, but it does not cover the metabolic context: melatonin’s effects on beta-cell insulin secretion and the possible sleep-disruption pathways that can show up during GLP-1 treatment.
The question of drug interactions is simpler than the question of whether a supplement addresses the problem. GLP-1 drugs may affect sleep through appetite suppression, glucose variability, GI effects, and arousal pathways; each points to a different approach. The supplement that helps may depend on which contributor is dominant in your case.
This article evaluates melatonin, magnesium (multiple forms), L-theanine, tart cherry, and prescription orexin antagonists through the lens of possible GLP-1-related sleep-disruption pathways. It does not cover general insomnia supplements. For the full overview of GLP-1 sleep effects, see the parent article.
Can You Take Melatonin While on Ozempic or Wegovy
The primary metabolic pathway for exogenous melatonin is CYP1A2-mediated 6-hydroxylation in the liver. Papagiannidou et al. (2014) tested multiple commonly co-administered drugs for their capacity to inhibit melatonin metabolism using human hepatic preparations. Of all drugs tested, only 5-methoxypsoralen — a potent CYP1A2 inhibitor — impaired melatonin metabolism at relevant concentrations. Diazepam, tamoxifen, and acetaminophen did not meaningfully inhibit melatonin’s metabolic conversion. Semaglutide, as a peptide degraded by proteolytic cleavage and beta-oxidation rather than CYP enzymes, does not enter this enzyme pathway as its primary clearance route.
The actual drug interactions to monitor are CYP1A2 inhibitors: fluvoxamine (an SSRI) and ciprofloxacin (a fluoroquinolone antibiotic) both potently inhibit CYP1A2 and can raise melatonin exposure. Semaglutide is not among these.
Ha et al. (2024) analyzed 35,479 adverse event reports associated with exogenous melatonin from 1996 through September 2022 in the WHO VigiBase global safety database. When compared against all other drugs in the database, melatonin showed elevated reporting odds ratios for nightmares (ROR 4.90), abnormal dreams (ROR 3.68), and falls (ROR 2.24). However, when compared specifically to other sleep medications rather than all drugs, these safety findings largely disappeared — indicating that melatonin’s safety profile is broadly comparable to other pharmacological sleep aids. The fall-risk finding is worth noting for anyone combining melatonin with other sedating agents such as orexin antagonists.
Where melatonin falls short for GLP-1 users is mechanism: melatonin addresses circadian timing — the internal clock cue that tells the brain when to sleep. It does not block orexin receptors. If arousal is being sustained through orexin/hypocretin signaling, melatonin may be insufficient on its own.
What Sleep Supplements Are Safe to Take With Glucagon-Like Peptide-1 Drugs
Magnesium glycinate/bisglycinate operates through a dual mechanism. The magnesium component can potentiate GABA-A receptor activity and has been associated with higher serum melatonin in one magnesium RCT. Abbasi et al. (2012) conducted an 8-week RCT of 500 mg magnesium versus placebo in 46 elderly participants. The magnesium group showed improvements in total sleep time (P = 0.002) and sleep efficiency (P = 0.03). Biochemical analysis revealed that magnesium supplementation increased serum melatonin concentrations and decreased serum cortisol (P = 0.008), providing a direct mechanistic link between magnesium and the hormonal regulators of sleep in elderly adults with primary insomnia.
Schuster et al. (2025) extended this evidence to a younger population. In a randomized, placebo-controlled trial of 155 adults aged 18-65 years, magnesium bisglycinate reduced Insomnia Severity Index scores versus placebo (Cohen’s d = 0.2), with greater benefit in those with low dietary magnesium intake. The bisglycinate chelate form was selected for its superior gastrointestinal tolerability and higher bioavailability compared to inorganic magnesium salts such as oxide.
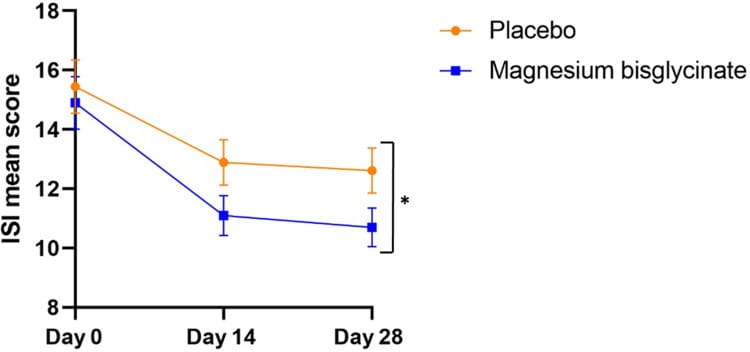
Glycine — the amino acid component of glycinate chelate — has been reported to lower core body temperature, one physiological change associated with sleep onset. This is why the glycinate form is relevant for sleep, not just the magnesium.
Magnesium L-threonate crosses the blood-brain barrier more efficiently than other magnesium forms due to the threonate carrier molecule. Hausenblas et al. (2024) conducted a 21-day RCT of 80 adults using Oura ring tracking alongside subjective instruments. The magnesium L-threonate group demonstrated improvements in Oura-estimated deeper sleep stages, increased REM sleep duration, and enhanced readiness scores compared to placebo. This is one of the few magnesium sleep trials with wearable-tracked outcomes rather than self-report alone.
L-theanine has been studied for stress physiology and alpha oscillatory activity, including one L-theanine-based drink trial showing increased posterior alpha oscillations in higher-anxiety participants. For GLP-1 users experiencing subjective restlessness, L-theanine may reduce mental agitation, although this has not been tested in GLP-1-treated populations.
Tart cherry extract contains small amounts of melatonin and procyanidins/phenolic compounds that may inhibit indoleamine 2,3-dioxygenase — an enzyme that degrades tryptophan before it converts to serotonin and melatonin. For GLP-1 users who are undereating, tryptophan availability may already be reduced from lower protein intake, making this tryptophan-preservation mechanism plausible but untested in GLP-1 users.
Rondanelli et al. (2011) tested a nightly combination of 5 mg melatonin, 225 mg magnesium, and 11.25 mg zinc in a double-blind, placebo-controlled RCT of 43 long-term care residents. The supplement group showed a between-group difference of 6.8 points on the Pittsburgh Sleep Quality Index (P < .001) -- providing RCT support for this specific combination compared with placebo.
Could Prescription Orexin-Blocking Sleep Medications Counteract Semaglutide Insomnia
The mechanistic match is plausible but not clinically proven for GLP-1 users. GLP-1 receptor signaling has been shown in preclinical work to depolarize orexin/hypocretin neurons in the lateral hypothalamus, increasing their firing rate and arousal output. Dual orexin receptor antagonists (DORAs) block OX1R and OX2R — the downstream receptors through which orexin neurons promote arousal. This is the sleep-medication class that most directly targets orexin signaling, if orexin-mediated arousal is part of the sleep pattern.
Khazaie et al. (2022) conducted a meta-analysis of eight randomized, double-blind, placebo-controlled trials — five evaluating suvorexant and three evaluating lemborexant. Both drugs demonstrated statistically significant superiority over placebo on subjective and objective sleep onset latency, wake after sleep onset, and total sleep time. The commonly reported adverse effects were somnolence, fatigue, and abnormal dreams. Their safety profile differs from GABAergic hypnotics, but individual risks, next-day effects, and respiratory comorbidities still belong in a prescribing conversation.
Owen (2016) reviewed suvorexant’s long-term safety profile and found no strong signals for rebound insomnia or physical withdrawal after 1-12 months of use — a distinction from benzodiazepines. Somnolence occurred in 7% or fewer of those taking approved doses.
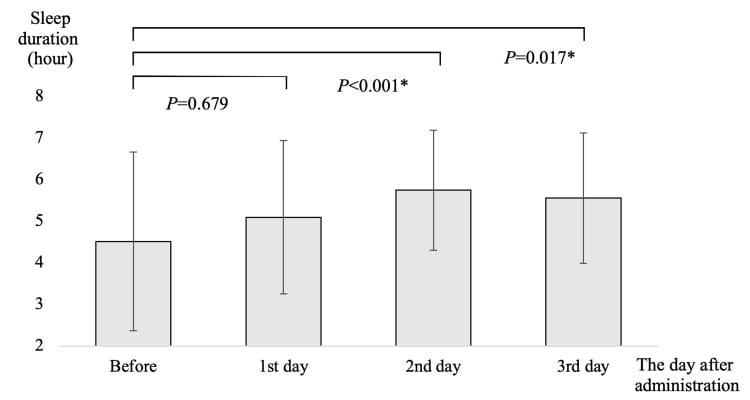
Mori et al. (2024) compared suvorexant and lemborexant head-to-head in a retrospective study of 105 hospitalized people. Lemborexant produced longer sleep on the first night of treatment (5.93 hours vs. 5.10 hours for suvorexant; P = 0.017), which the authors discussed in relation to lemborexant’s greater potency at OX2R — the orexin receptor subtype that governs sleep maintenance. Suvorexant reached significance only on days 2-3. On fall safety, no falls were recorded in the suvorexant group compared to three falls (5.3%) in the lemborexant group, though this difference was not statistically significant (P = 0.248).
DORAs are schedule IV controlled substances. The rationale for their use in GLP-1-related insomnia is mechanistic, not established by trial — no published trial has tested DORAs specifically in GLP-1-treated populations. This is a prescriber conversation, not a self-treatment decision.
Does Melatonin Interact With Semaglutide’s Effect on Blood Sugar
Melatonin acts on MT1 and MT2 receptors expressed on pancreatic beta cells — the same cells that semaglutide stimulates through GLP-1 receptors. MT1 activation generally suppresses insulin secretion through Gi-coupled pathways, which could theoretically oppose the Gs-coupled GLP-1 receptor pathway on the same cells. This is the mechanism Drugs.com does not address when it reports “no known interaction.”
MT2 activation can potentiate insulin release in some contexts. Exogenous melatonin activates both receptor subtypes simultaneously, meaning the net effect on insulin secretion depends on relative receptor expression, timing of administration, and the individual’s metabolic state.
The distinction matters for different populations. People with type 2 diabetes depend on semaglutide’s insulinotropic effect for glycemic control. Melatonin-mediated MT1 suppression could theoretically blunt this insulin-stimulating action, reducing the drug’s glucose-lowering benefit. Weight-management users without diabetes have intact endogenous insulin regulation and are less likely to notice a clinically important change, but direct co-administration data are lacking.
Abbasi et al. (2012) demonstrated that 500 mg magnesium supplementation increased serum melatonin concentrations — meaning magnesium users are also raising their endogenous melatonin exposure at beta cells. This is relevant for people stacking magnesium with exogenous melatonin: both supplements increase melatonin activity at the beta-cell level.
No evidence demonstrates meaningful glucose disruption from melatonin and semaglutide co-administration. But people with type 2 diabetes adding melatonin should consider monitoring fasting glucose with guidance from the clinician managing their diabetes medication, especially when adding or changing melatonin dose or timing.
GLP-1 insomnia is rarely caused by a single mechanism. Orexin activation, blood sugar instability, caloric deficit, and neurotransmitter changes may each contribute in different people — and the best-fitting supplement may depend on which pathways are disrupted. Sleep disruption from metabolic causes often overlaps with hormonal, circadian, or autonomic factors that no single supplement can address.
Find out which causes might be driving your sleep disruption
Can Magnesium Glycinate Help With Ozempic Insomnia Specifically
The glycinate/bisglycinate form is a plausible candidate for GLP-1 users because it delivers magnesium in a chelated form with high bioavailability and minimal gastrointestinal side effects. Magnesium oxide — the form sold in the highest volume — has poor absorption and frequently causes diarrhea, which is counterproductive for GLP-1 users already dealing with gastrointestinal side effects from semaglutide.
Magnesium L-threonate is another option when the goal is brain magnesium delivery. Because L-threonate crosses the blood-brain barrier more efficiently, it may produce stronger effects on Oura-estimated sleep-stage metrics — deeper sleep stages and increased REM — as demonstrated by Hausenblas et al. (2024) using objective Oura ring tracking.
The Abbasi et al. (2012) cortisol-reduction finding is relevant to GLP-1 users as an indirect sleep-regulation clue, not GLP-1-specific proof. GLP-1-related insomnia may overlap with stress-axis arousal when appetite suppression, under-eating, or glucose variability is present. Magnesium’s documented ability to reduce serum cortisol addresses this secondary arousal pathway independently from its effects on melatonin and GABA.
Is It Safe to Take Prescription Sleep Aids Like Ambien While on Ozempic
Z-drugs suppress brain activity broadly by enhancing GABA-mediated inhibition across the cortex. This induces sleep but does not selectively address the wakefulness drive from orexin neurons that GLP-1 signaling may activate in preclinical physiology. DORAs (suvorexant, lemborexant) target orexin receptors involved in arousal.
Owen (2016) documented that suvorexant showed no strong evidence of rebound insomnia or physical withdrawal after 1-12 months, while Z-drugs carry established risks for both. If the sleep pattern appears arousal-driven, the mechanistic logic may favor orexin antagonists for GLP-1 insomnia specifically. This is a discussion to have with a prescribing physician who understands both drug classes.
Does Cannabidiol or Cannabinol Interact With Semaglutide for Sleep
The evidence base does not yet support cannabinoid use for the specific mechanisms that may drive GLP-1-related sleep disruption. CB1 and CB2 receptors are part of the endocannabinoid pathway, which can influence sleep through routes that have not been tested against the orexin or metabolic circuits potentially relevant to semaglutide. No controlled trial has evaluated cannabinoids in GLP-1-treated populations. The mechanistic mismatch — cannabinoid receptors versus orexin and metabolic pathways — suggests limited targeted benefit for this specific type of insomnia.
Can Tart Cherry Extract Help With Ozempic-Related Sleep Problems
The connection to GLP-1 insomnia is through the undereating pathway. Semaglutide suppresses appetite, and many users consume less protein as a result. Lower protein intake means less dietary tryptophan — the amino acid precursor to serotonin and then melatonin. Tart cherry’s phenolic compounds may reduce tryptophan degradation, but no study has shown that tart cherry compensates for reduced protein intake in GLP-1 users.
Tart cherry is not a standalone solution for GLP-1 insomnia but may complement magnesium in a multi-pathway approach. Rondanelli et al. (2011) demonstrated that combination supplementation (melatonin, magnesium, and zinc together) produced a 6.8-point improvement on the Pittsburgh Sleep Quality Index (P < .001) -- supporting that this specific combination improved sleep quality compared with placebo in older long-term care residents.
Should You Take Sleep Supplements at a Different Time Than Your Ozempic Injection
The injection-day sleep pattern is commonly reported among GLP-1 users: insomnia may feel worse on the night of injection and the following 1-2 nights, then improves toward the end of the dosing interval. This corresponds to semaglutide’s pharmacokinetic profile — peak plasma concentration occurs 1-3 days after dosing, but human brain exposure and orexin activation timing have not been established.
Supplements may be most useful on the nights when symptoms cluster, not necessarily every night. For users who prefer not to take supplements daily, concentrating melatonin and magnesium on the 2-3 nights following injection may provide targeted support during the reported higher-arousal window.
Related Reading
- Metabolic Sleep Disruption — The full metabolic sleep framework covering glucose regulation, insulin resistance, mitochondrial decline, NAD+ loss, and fat metabolism
- Does Ozempic Cause Insomnia What GLP-1 Drugs Do to Your Sleep — The broad overview of GLP-1 sleep side effects and mechanisms
- Why Does Ozempic Make You Tired But Keep You Awake — How GLP-1 drugs activate orexin/wakefulness circuitry while appetite suppression can create fatigue
- Why Does Ozempic Wake You Up at 3am — How lower overnight fuel availability and counter-regulatory hormones can contribute to 3am waking
- Does Ozempic Give You Vivid Dreams — How GLP-1 drugs may affect dream intensity through arousal and neuropsychiatric pathways
- Does Eating Too Little on Ozempic Cause Insomnia — How appetite suppression can affect tryptophan, serotonin, melatonin, and overnight glucose stability
- Does Your Ozempic Injection Day Disrupt Your Sleep The 24-48 Hour Pattern — Why sleep disruption can follow a weekly pattern tied to injection timing and peak drug exposure
- Does Your Semaglutide Dose Increase Restart Your Insomnia Why Each Titration Can Disrupt Sleep Again — Why each titration step can restart sleep disruption before adaptation catches up
- Does Ozempic Give You Anxiety at Night Why GLP-1 Drugs May Contribute to Nighttime Panic — How orexin activation, HPA-axis signaling, and glucose instability may contribute to nighttime anxiety
- Do GLP-1 Drugs Disrupt Your Body Clock Ozempic, Circadian Rhythms, and Melatonin — How GLP-1 secretion rhythms, food timing, peripheral clocks, and melatonin intersect
- Ozempic Night Sweats: How GLP-1 Drugs May Affect Temperature at Night — How GLP-1 drugs may affect nighttime sweating through autonomic tone and thermoregulation
- Ozempic Nausea and Sleep: How Stomach Side Effects Keep You Awake at Night — How delayed gastric emptying, reflux, and nausea can fragment sleep
- Will Your Sleep Go Back to Normal When You Stop Ozempic — What is known and not known about sleep after semaglutide discontinuation
- Does Ozempic Muscle Loss Affect Your Sleep Sarcopenia, Temperature, and Deep Sleep — How lean-mass loss, thermoregulation, and deep sleep can interact during GLP-1 weight loss
- Could Ozempic Contribute to Restless Legs at Night Iron, B12, and the Dopamine Connection — How iron, B12, dopamine, and rapid weight loss may intersect with restless legs at night
- Does Compounded Semaglutide Affect Sleep Differently Than Brand-Name Ozempic — How dose variability, formulation differences, and adverse-event tracking can affect sleep side effects
- Ozempic and Sleep After 40: Why GLP-1 Side Effects Can Feel Different as You Age — Why GLP-1 sleep effects can land differently after 40 because of muscle, hormones, and glucose regulation
References
- Abbasi, B., Kimiagar, M., Sadeghniiat, K., Shirazi, M. M., Hedayati, M., & Rashidkhani, B. (2012). The effect of magnesium supplementation on primary insomnia in elderly: A double-blind placebo-controlled clinical trial. Journal of Research in Medical Sciences, 17(12), 1161-1169. https://pubmed.ncbi.nlm.nih.gov/23853635/
- Acuna-Goycolea, C., & van den Pol, A. (2004). Glucagon-like peptide 1 excites hypocretin/orexin neurons by direct and indirect mechanisms: Implications for viscera-mediated arousal. The Journal of Neuroscience, 24(37), 8141-8152. https://pubmed.ncbi.nlm.nih.gov/15371515/
- Bannai, M., & Kawai, N. (2012). New therapeutic strategy for amino acid medicine: Glycine improves the quality of sleep. Journal of Pharmacological Sciences, 118(2), 145-148. https://pubmed.ncbi.nlm.nih.gov/22293292/
- Garaulet, M., Gomez-Abellan, P., Rubio-Sastre, P., Madrid, J. A., Saxena, R., & Scheer, F. A. J. L. (2015). Common type 2 diabetes risk variant in MTNR1B worsens the deleterious effect of melatonin on glucose tolerance in humans. Metabolism: Clinical and Experimental, 64(12), 1650-1657. https://pubmed.ncbi.nlm.nih.gov/26440713/
- Ha, M., Yoon, D., Lee, C. Y., Lee, M., Kim, Y. W., Lee, J. M., & Shin, J. Y. (2024). Investigating the safety profiles of exogenous melatonin and associated adverse events: A pharmacovigilance study using WHO-VigiBase. Journal of Pineal Research, 76(2), e12949. https://pubmed.ncbi.nlm.nih.gov/38528668/
- Hausenblas, H. A., Lynch, T., Hooper, S., Shrestha, A., Rosendale, D., & Gu, J. (2024). Magnesium-L-threonate improves sleep quality and daytime functioning in adults with self-reported sleep problems: A randomized controlled trial. Sleep Medicine: X, 8, 100121. https://pubmed.ncbi.nlm.nih.gov/39252819/
- Hausenblas, H., Hooper, S., & Lynch, T. (2025). Effectiveness of a cannabinoids supplement on sleep and mood in adults with subthreshold insomnia: A randomized double-blind placebo-controlled crossover pilot trial. Health Science Reports, 8(2), e70481. https://pubmed.ncbi.nlm.nih.gov/39980821/
- Khazaie, H., Sadeghi, M., Khazaie, S., Hirshkowitz, M., & Sharafkhaneh, A. (2022). Dual orexin receptor antagonists for treatment of insomnia: A systematic review and meta-analysis on randomized, double-blind, placebo-controlled trials of suvorexant and lemborexant. Frontiers in Psychiatry, 13, 1070522. https://pubmed.ncbi.nlm.nih.gov/36578296/
- Lavender, I. G., Marshall, N. S., McCartney, D., Cho, G., Irwin, C., Suraev, A., Gordon, R., Arnold, J. C., D’Rozario, A. L., Gordon, C. J., Saini, B., Sivam, S., Zheng, Y., Grunstein, R. R., Yee, B. J., McGregor, I. S., & Hoyos, C. M. (2026). Cannabinol for acute treatment of insomnia disorder in a randomized placebo-controlled crossover trial. Journal of Sleep Research, e70284. https://pubmed.ncbi.nlm.nih.gov/41698831/
- Losso, J. N., Finley, J. W., Karki, N., Liu, A. G., Prudente, A., Tipton, R., Yu, Y., & Greenway, F. L. (2018). Pilot study of the tart cherry juice for the treatment of insomnia and investigation of mechanisms. American Journal of Therapeutics, 25(2), e194-e201. https://pubmed.ncbi.nlm.nih.gov/28901958/
- Mori, K., Kimura, M., & Usami, E. (2024). Short-term efficacy and safety of suvorexant and lemborexant: A retrospective study. Cureus, 16(10), e71049. https://pubmed.ncbi.nlm.nih.gov/39512953/
- Muhlbauer, E., Albrecht, E., Bazwinsky-Wutschke, I., & Peschke, E. (2012). Melatonin influences insulin secretion primarily via MT1 receptors in rat insulinoma cells (INS-1) and mouse pancreatic islets. Journal of Pineal Research, 52(4), 446-459. https://pubmed.ncbi.nlm.nih.gov/22288848/
- Owen, R. T. (2016). Suvorexant: Efficacy and safety profile of a dual orexin receptor antagonist in treating insomnia. Drugs of Today, 52(1), 29-40. https://pubmed.ncbi.nlm.nih.gov/26937493/
- Papagiannidou, E., Skene, D. J., & Ioannides, C. (2014). Potential drug interactions with melatonin. Physiology & Behavior, 131, 17-24. https://pubmed.ncbi.nlm.nih.gov/24732412/
- Ranum, R. M., Whipple, M. O., Croghan, I., Bauer, B., Toussaint, L. L., & Vincent, A. (2023). Use of cannabidiol in the management of insomnia: A systematic review. Cannabis and Cannabinoid Research, 8(2), 213-229. https://pubmed.ncbi.nlm.nih.gov/36149724/
- Rondanelli, M., Opizzi, A., Monteferrario, F., Antoniello, N., Manni, R., & Klersy, C. (2011). The effect of melatonin, magnesium, and zinc on primary insomnia in long-term care facility residents in Italy: A double-blind, placebo-controlled clinical trial. Journal of the American Geriatrics Society, 59(1), 82-90. https://pubmed.ncbi.nlm.nih.gov/21226679/
- Schuster, J., Cycelskij, I., Lopresti, A., & Hahn, A. (2025). Magnesium bisglycinate supplementation in healthy adults reporting poor sleep: A randomized, placebo-controlled trial. Nature and Science of Sleep, 17, 2027-2040. https://pubmed.ncbi.nlm.nih.gov/40918053/
- Suraev, A. S., Marshall, N. S., Vandrey, R., McCartney, D., Benson, M. J., McGregor, I. S., Grunstein, R. R., & Hoyos, C. M. (2020). Cannabinoid therapies in the management of sleep disorders: A systematic review of preclinical and clinical studies. Sleep Medicine Reviews, 53, 101339. https://pubmed.ncbi.nlm.nih.gov/32603954/
- White, D. J., de Klerk, S., Woods, W., Gondalia, S., Noonan, C., & Scholey, A. B. (2016). Anti-stress, behavioural and magnetoencephalography effects of an L-theanine-based nutrient drink: A randomised, double-blind, placebo-controlled, crossover trial. Nutrients, 8(1), 53. https://pubmed.ncbi.nlm.nih.gov/26797633/
- Yang, X. D., & Yang, Y. Y. (2024). Clinical pharmacokinetics of semaglutide: A systematic review. Drug Design, Development and Therapy, 18, 2555-2570. https://pubmed.ncbi.nlm.nih.gov/38952487/
- Zibolka, J., Bazwinsky-Wutschke, I., Muhlbauer, E., & Peschke, E. (2018). Distribution and density of melatonin receptors in human main pancreatic islet cell types. Journal of Pineal Research, 65(1), e12480. https://pubmed.ncbi.nlm.nih.gov/29464840/
Written by Kat Fu, M.S., M.S. – Last reviewed: May 2026 – 21 references cited
