Exercise is one of the few approaches that builds new mitochondria rather than optimizing existing ones. For people whose sleep has degraded with age or chronic stress, patching a depleted mitochondrial network is fundamentally different from expanding it. The research connecting exercise type, mitochondrial biogenesis, and measurable sleep architecture changes has matured in 2024 and 2025.
This article covers which exercise types produce the best sleep outcomes, what the molecular biogenesis pathway looks like, how long results take to appear, and when exercise volume crosses from helpful to counterproductive. For supplement-based mitochondrial support, see CoQ10, PQQ: Can Mitochondrial Supplements Fix Your Sleep?. Mitochondrial reduction is one of several metabolic causes of sleep disruption — for the full overview, see Metabolic Sleep Disruption.
Which Type of Exercise Produces the Best Sleep — High-Intensity Intervals, Cardio, or Weights?
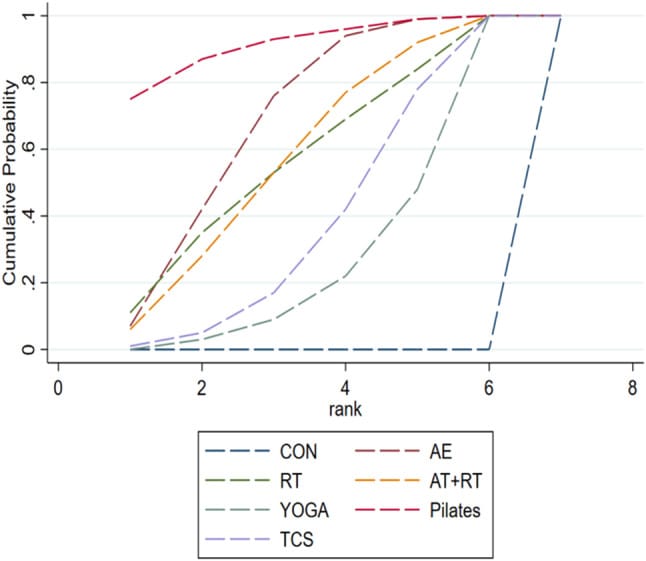
The 2024 network meta-analysis by Gao et al. synthesized 28 randomized controlled trials (3,460 middle-aged and older adults) comparing aerobic exercise, resistance training, combined training, and yoga. Aerobic exercise ranked first for overall sleep quality (Surface Under the Cumulative Ranking Curve 93.2%) and sleep latency reduction (96.8%). Yoga ranked highest for sleep efficiency (95.9%), sleep duration (93.8%), and daytime impairment (98.3%) — suggesting complementary mechanisms between aerobic and mind-body modalities.
The 2025 meta-analysis by Wang et al. expanded the evidence to 86 randomized controlled trials and 7,276 participants. All six modalities produced significant sleep improvements: Pilates (standardized mean difference -1.65), aerobic exercise (-1.21), resistance training (-1.12), combined training (-1.11), traditional Chinese sports (-0.94), and yoga (-0.82). Pilates ranked first but based on fewer trials; aerobic exercise has the largest evidence base.
For mitochondrial biogenesis, the 2025 meta-analysis by Abrego-Guandique et al. found interval training (Hedges’ g = 1.29) and continuous training (Hedges’ g = 1.01) produce equivalent large PGC-1alpha responses with no statistically significant difference. The 2025 biopsy study by Li et al. (20 young men, 6 weeks) added nuance: both modalities increase citrate synthase and Complex I activity, but high-intensity interval training produces more pronounced fusion protein upregulation and longitudinal mitochondrial network architecture, while moderate-intensity continuous training produces grid-like cross-sarcomere patterns.
For deep sleep specifically, Park et al. (2021) demonstrated in a small crossover study (9 participants) that 60 minutes of aerobic exercise at 60% of maximal oxygen uptake increased delta power during N3 slow-wave sleep (p < 0.05) and enhanced slow-wave stability. Subjective sleep quality was judged lower — indicating aerobic exercise reshapes sleep architecture at the electroencephalogram level even when subjective perception does not reflect the objective improvement. Larger trials are needed to show these findings across broader populations.
How Much Exercise Improves Sleep — and When Does It Disrupt Sleep?
The 2025 dose-response analysis by Wang et al. (86 randomized controlled trials) identified a U-shaped relationship across modalities: both under-dosing and over-dosing exercise reduced sleep benefits. The overall optimal dose was approximately 920 MET-minutes per week, equivalent to roughly 200 minutes of moderate-intensity aerobic activity per week. Optimal dose varied by modality — Pilates peaked at 390 MET-minutes per week, while aerobic exercise peaked at 1,100 MET-minutes per week. The therapeutic window concept applies: more is not always better.
The mechanistic explanation comes from Powers et al. (2024): exercise-generated ROS activate PGC-1alpha, Nrf2, NF-kappaB, and HSF1 as adaptive pathway activity molecules. At moderate intensity, ROS drive mitochondrial biogenesis and upregulate endogenous antioxidant defenses (superoxide dismutase, catalase, glutathione peroxidase). At excessive intensity or with inadequate recovery, ROS overwhelm antioxidant capacity and produce oxidative stress instead of adaptation.
Saner et al. (2018) proposed, based on mechanistic reasoning rather than direct experimental evidence, that exercise and sleep reduction act on the same AMPK/PGC-1alpha/SIRT1 molecular targets in opposing directions. This means exercising hard on insufficient sleep may not produce the expected biogenesis response because the pathway activity machinery is already compromised. For people caught in the cycle described in Wired But Tired: When Your Mitochondria Can’t Power Down, the optimization window is narrow — enough exercise to activate biogenesis, not so much that it compounds existing stress.
One practical implication from Powers et al. (2024): taking antioxidant supplements around exercise can blunt the adaptive ROS cues needed for PGC-1alpha activation. The common pattern of exercising and then supplementing antioxidants may undermine the mitochondrial benefits exercise produces.
How Long Does It Take for an Exercise Program to Measurably Improve Sleep?
Little et al. (2011) tracked 8 young men through a single high-intensity interval training session (four 30-second maximal-effort cycling sprints). post-exercise, p38 MAPK and AMPK activated. At 3 hours, nuclear PGC-1alpha was elevated. By 24 hours, mitochondrial protein content and enzyme activity were measurably increased. These results come from a small mechanistic study, but the pathway activity timeline has been replicated in subsequent research: a single high-intensity session triggers the full PGC-1alpha-mediated biogenesis pathway activity cascade.
Little et al. (2010) established the two-week adaptation window in another small study: 7 young men completed 6 high-intensity interval training sessions over 2 weeks. Nuclear PGC-1alpha protein increased approximately 24%. SIRT1 protein increased approximately 56%. Citrate synthase, cytochrome c oxidase, and mitochondrial transcription factor A all increased, showing activation of the complete PGC-1alpha-to-mitochondrial-DNA-transcription axis.
The gap between molecular changes (hours), structural adaptation (2 weeks), and practical sleep outcomes (8 to 12 weeks) reflects the time needed for mitochondrial network expansion to translate into consistent sleep architecture changes. Park et al. (2021) showed delta power increases after a single aerobic session, but the meta-analyses by Gao et al. (2024) and Wang et al. (2025) pooled 8- to 12-week approaches for sustained sleep quality improvement.
Whether exercise can rescue mitochondria already stressd by sleep deprivation remains an open question. Saner et al. (2018) proposed that it can, since exercise and sleep reduction act on the shared AMPK/PGC-1alpha/SIRT1 pathways in opposing directions — but no direct interventional study has tested this in sleep-deprived populations. Maintaining exercise during periods of poor sleep is reasonable based on this mechanistic logic, even if the biogenesis response is blunted. For more on what happens when mitochondria not work to produce sufficient energy despite exhaustion, see Why You’re Exhausted But Can’t Sleep.
What Is Mitochondrial Biogenesis and How Does Exercise Trigger It?
The convergence model, detailed by Hood et al. (2011), explains why exercise is such a potent biogenesis stimulus. A single bout of exercise activates parallel pathway activity cascades simultaneously: AMPK senses the falling ATP-to-ADP ratio as muscles consume energy; p38 MAPK responds to calcium flux and ROS; CaMKII responds to calcium release from muscle contraction. All three pathways converge independently on PGC-1alpha, which then co-activates transcription factors for both nuclear-encoded mitochondrial genes and mitochondrial-DNA-encoded genes. Because multiple exercise-derived cues independently drive the same biogenesis outcome, no single pathway is a bottleneck — the process has built-in redundancy.
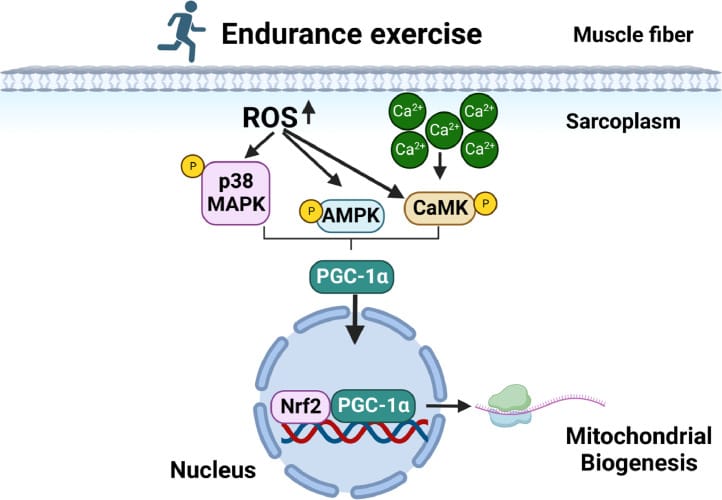
ROS play a required pathway activity role. Powers et al. (2024) established that exercise-generated ROS from NADPH oxidase, mitochondria, and xanthine oxidase are pathway activity molecules that drive adaptation. ROS activate p38 MAPK pathway activity necessary for PGC-1alpha activation. ROS also activate NF-kappaB, which upregulates endogenous antioxidant defenses. Without the ROS cue, biogenesis does not fully proceed.
The 2025 meta-analysis by Abrego-Guandique et al. quantified biogenesis magnitude: pooled effect on PGC-1alpha expression was large (Hedges’ g = 1.17, 95% confidence interval: 0.14 to 2.19). Both interval training (g = 1.29) and continuous training (g = 1.01) drove large responses with no significant difference between modalities.
Li et al. (2025) revealed that exercise intensity shapes mitochondrial network architecture, not just quantity. High-intensity interval training produced longitudinal networks with greater fusion protein expression; moderate-intensity continuous training produced grid-like cross-sarcomere networks. Both are functional but structurally distinct — among the first human evidence that training intensity determines mitochondrial network geometry.
The connection to sleep is direct: the mitochondrial network that exercise builds generates the ROS sleep-pressure cue discussed in How Your Mitochondria Decide When You Sleep, produces ATP for neuronal housekeeping during sleep, and drives the circadian clock through NAD+-dependent SIRT1 activity. A depleted network does not work at all three functions.
Exercise rebuilds mitochondrial capacity, but in adults over 40, it is one of several metabolic processes affecting sleep. Blood sugar instability, hormonal changes, inflammatory pathway activity, and cortisol rhythm disruption can compound alongside depleted mitochondrial networks — and exercise addresses only the mitochondrial piece of that process.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Does Exercise Increase Deep Sleep?
Park et al. (2021) compared a 60-minute aerobic session against a resting control in 9 participants with full-night polysomnography. Delta power during N3 slow-wave sleep increased (p < 0.05), and slow-wave stability was enhanced — captured by envelope analysis of the electroencephalogram but not by standard sleep staging. The dissociation between objective improvement and subjective perception explains why some people conclude exercise "does not help" their sleep when polysomnography shows otherwise. The paper found that exercise improved the efficiency of early sleep processes rather than increasing overall homeostatic sleep pressure. This was a small crossover study, and showation in larger samples would strengthen the finding.
Can Exercise Counteract the Effects of Poor Sleep on Mitochondria?
Saner et al. (2018) proposed exercise as a "viable, suitable, and potent alternative approach" for sleep-reduction-induced mitochondrial impairment. Sleep reduction impairs mitochondrial respiratory function through disrupted hormonal secretion, dysregulated circadian clock genes, and impaired AMPK/PGC-1alpha/SIRT1 pathway activity — the same pathways exercise activates. This proposal is based on shared molecular targets, not on a randomized controlled trial directly testing exercise in sleep-deprived populations. Maintaining exercise during periods of poor sleep is reasonable based on this logic, even if the biogenesis response is reduced. For more on the metabolic cascade during 3am waking, see The 3am Wakeup Is a Metabolic Emergency.
Can Overexercising Make Sleep Lower?
Wang et al. (2025) provide the strongest evidence for an exercise therapeutic ceiling: beyond approximately 920 MET-minutes per week, sleep benefits reduced across modalities. Modality-specific ceilings differed — Pilates peaked at 390 MET-minutes per week, aerobic exercise at 1,100. For people combining high-intensity interval training and resistance training, total weekly volume matters more than any single session.
The hormetic framework from Powers et al. (2024) explains the mechanism: moderate ROS production drives PGC-1alpha activation and mitochondrial biogenesis, while excessive production overwhelms antioxidant defenses and produces oxidative stress — elevating sympathetic tone, increasing cortisol output, and fragmenting sleep architecture.
Related Reading
- Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep — the pillar article for metabolic sleep disruption, covering glucose regulation, cortisol rhythm, mitochondrial stress, NAD+ reduction, and fat metabolism
- Why Are You Exhausted But Can’t Sleep? Is It a Mitochondrial Energy Paradox? — mitochondrial fatigue, unrefreshing sleep, ATP production, and why rest may not restore cellular energy
- Do Your Mitochondria Control When You Sleep? and Why Does It Break Down After 40? — ROS sleep pressure, deep sleep reduction after 40, mitochondrial DNA aging, and sleep timing
- Can CoQ10 and PQQ Supplements Improve Your Sleep? — CoQ10, PQQ, magnesium, supplement timing, and the evidence for mitochondrial sleep support
- Is Your 3am Wakeup a Metabolic Emergency? Blood Sugar, Cortisol, and Mitochondrial reduction? — the 2-4am vulnerability window, glucose drops, cortisol rescue, and mitochondrial energy buffering
- Why Are You Wired but Tired, and What Role Do Mitochondria Play? — hyperarousal, cortisol rhythm changes, stress physiology, and mitochondrial power-down reduction
- What Is the Measured Cellular Impact of Sleep Fragmentation? — fragmented sleep, oxidative stress, vascular injury, inflammation, and cellular aging impact
References
Abrego-Guandique, D. M., Aguilera Rojas, N. M., Chiari, A., Luciani, F., Cione, E., & Cannataro, R. (2025). The impact of exercise on mitochondrial biogenesis in skeletal muscle: A review and meta-analysis of randomized trials. Biomolecular Concepts, 16(1). https://pubmed.ncbi.nlm.nih.gov/40459444/
Gao, X., Qiao, Y., Chen, Q., Wang, C., & Zhang, P. (2024). Effects of different types of exercise on sleep quality based on Pittsburgh Sleep Quality Index in middle-aged and older adults: A network meta-analysis. Journal of Clinical Sleep Medicine, 20(7), 1193-1204. https://pubmed.ncbi.nlm.nih.gov/38450497/
Hood, D. A., Uguccioni, G., Vainshtein, A., & D’souza, D. (2011). Mechanisms of exercise-induced mitochondrial biogenesis in skeletal muscle: Implications for health and disease. Comprehensive Physiology, 1(3), 1119-1134. https://pubmed.ncbi.nlm.nih.gov/23733637/
Li, Y., Zhao, W., & Yang, Q. (2025). Effects of high-intensity interval training and moderate-intensity continuous training on mitochondrial dynamics in human skeletal muscle. Frontiers in Physiology, 16, 1554222. https://pubmed.ncbi.nlm.nih.gov/40313872/
Little, J. P., Safdar, A., Bishop, D., Tarnopolsky, M. A., & Gibala, M. J. (2011). An acute bout of high-intensity interval training increases the nuclear abundance of PGC-1alpha and activates mitochondrial biogenesis in human skeletal muscle. American Journal of Physiology: Regulatory, Integrative and Comparative Physiology, 300(6), R1303-R1310. https://pubmed.ncbi.nlm.nih.gov/21451146/
Little, J. P., Safdar, A., Wilkin, G. P., Tarnopolsky, M. A., & Gibala, M. J. (2010). A clinical model of low-volume high-intensity interval training induces mitochondrial biogenesis in human skeletal muscle: Potential mechanisms. The Journal of Physiology, 588(Pt 6), 1011-1022. https://pubmed.ncbi.nlm.nih.gov/20100740/
Park, I., Diaz, J., Matsumoto, S., Iwayama, K., Nabekura, Y., Ogata, H., Kayaba, M., Aoyagi, A., Yajima, K., Satoh, M., Tokuyama, K., & Vogt, K. E. (2021). Exercise improves the quality of slow-wave sleep by increasing slow-wave stability. Scientific Reports, 11(1), 4410. https://pubmed.ncbi.nlm.nih.gov/33627708/
Powers, S. K., Radak, Z., Ji, L. L., & Jackson, M. (2024). ROS promote endurance exercise-induced adaptations in skeletal muscles. Journal of Sport and Health Science, 13(6), 780-792. https://pubmed.ncbi.nlm.nih.gov/38719184/
Saner, N. J., Bishop, D. J., & Bartlett, J. D. (2018). Is exercise a viable therapeutic approach to mitigate mitochondrial impairment and insulin resistance induced by sleep reduction? Sleep Medicine Reviews, 37, 60-68. https://pubmed.ncbi.nlm.nih.gov/29056415/
Wang, H., Xin, X., & Pan, Y. (2025). The best approaches and doses of exercise for improving sleep quality: A network meta-analysis and dose-response relationship study. BMC Public Health, 25(1), 1371. https://pubmed.ncbi.nlm.nih.gov/40217183/
Written by Kat Fu, M.S., M.S.? Last reviewed: May 2026? 10 references cited
