Nighttime anxiety on semaglutide is reported in pharmacovigilance datasets and social-media analyses, but the evidence does not yet isolate nighttime anxiety as its own studied outcome.
This article explains why GLP-1 drugs may contribute to nighttime anxiety or panic-like arousal through several plausible pathways: orexin-related wakefulness, HPA-axis signaling, lower overnight fuel availability, nausea-related visceral input, and limbic stress circuits. It also addresses the gap between pharmacovigilance data, where anxiety is a recurring report, and controlled trial data, where broad psychiatric harm has not been shown in selected trial populations. For the full overview of GLP-1 sleep effects, see the parent article on Ozempic and insomnia.
Nighttime anxiety is one possible sleep-disrupting pattern reported by some people taking GLP-1 drugs. Other articles in this series cover orexin-driven wakefulness, blood sugar instability, and vivid dreams.
Why Does Ozempic Cause Anxiety or Restlessness at Night
Three plausible pathways can produce anxiety-like states at night in people taking semaglutide, although each pathway has different levels of human evidence.
Orexin activation. GLP-1 depolarizes orexin neurons in the lateral hypothalamus in mouse brain-slice work (Acuna-Goycolea & van den Pol, 2004). Orexin is a wakefulness-promoting neuropeptide that helps sustain arousal and sleep-wake stability. If GLP-1-related orexin signaling increases arousal near bedtime, the experience could feel like restlessness, racing thoughts, or difficulty winding down.
HPA axis engagement. GLP-1 receptors are expressed in the paraventricular nucleus (PVN), a region involved in corticotropin-releasing hormone (CRH), ACTH, and cortisol signaling. A crossover trial in 20 healthy volunteers found that dulaglutide at approved doses did not increase 24-hour urinary cortisol or alter circadian cortisol rhythm (Winzeler et al., 2019). This null cortisol finding argues against sustained cortisol elevation as the main explanation for every GLP-1-related arousal complaint, at least at the dulaglutide dose and timeframe studied.
Blood sugar instability. Appetite suppression from semaglutide can lead to lower food intake, and lower evening intake can make some people more vulnerable to overnight glucose dips. When blood glucose falls during sleep, the counter-regulatory response can include epinephrine and other stress hormones; in controlled hypoglycemia studies, epinephrine can rise before awakening. That pattern can feel like rapid-onset, panic-like arousal from a metabolic trigger rather than a primary anxiety disorder.
During waking hours, external activity competes with internal sensations. At bedtime, the same internal arousal or visceral discomfort may be more noticeable.
A social media analysis of 43,710 posts across Reddit, YouTube, and TikTok found insomnia was the top-reported mental health concern with GLP-1 drugs (620 mentions), followed by anxiety (353 mentions). Users described bidirectional mood effects — some reported improvement, others reported worsening — suggesting individual variation in how people experience GLP-1 receptor agonists (Arillotta et al., 2023).
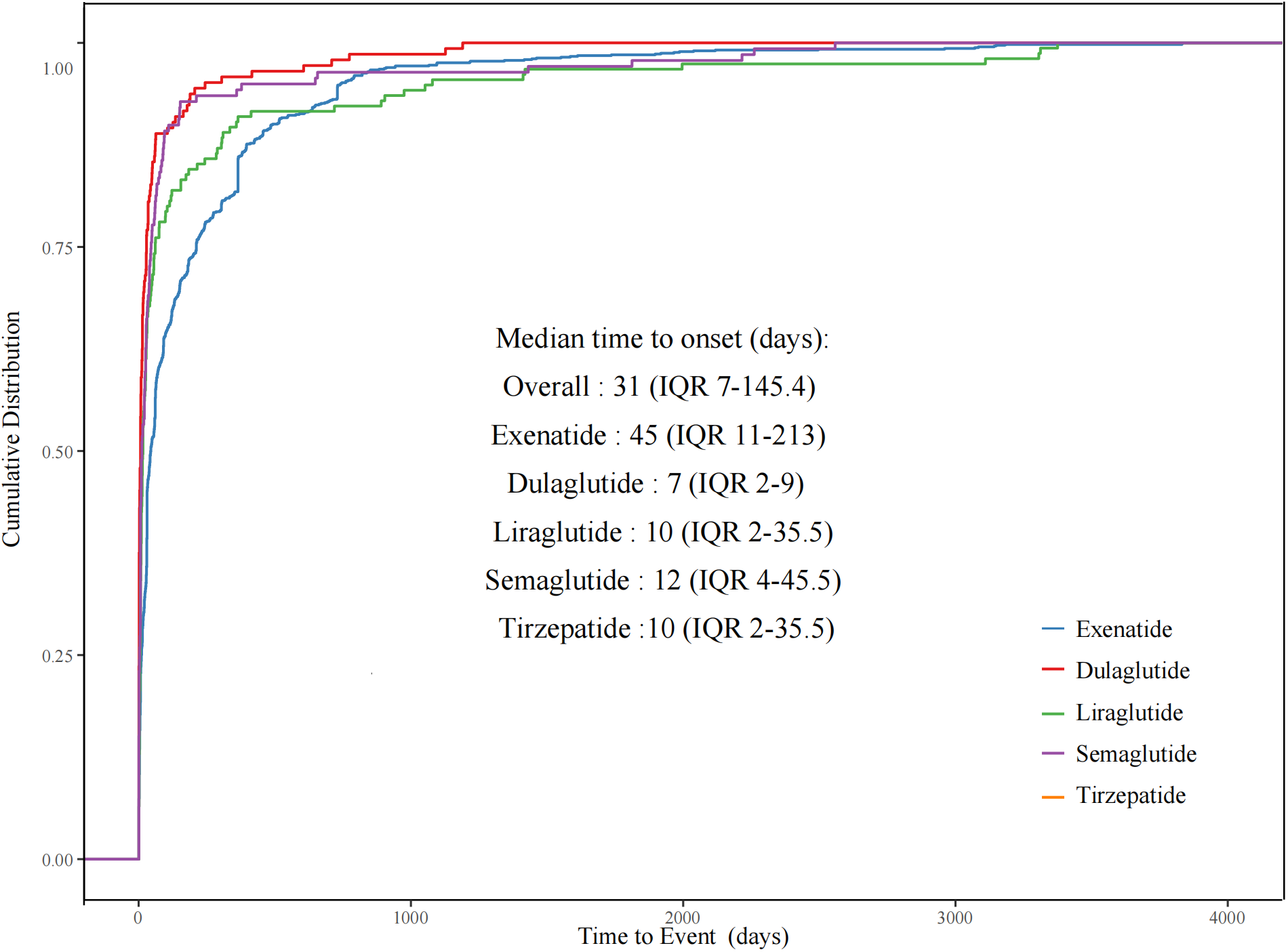
What Do Pharmacovigilance Databases Show About Semaglutide and Anxiety
FAERS database. An analysis of 181,238 GLP-1 receptor agonist adverse event reports in the FDA’s FAERS database identified 8,240 psychiatric adverse events (4.5% of reports). Eight distinct psychiatric categories showed disproportionality, including nervousness (reporting odds ratio [ROR] 1.97) and fear of eating (ROR 3.35). The population was 65.89% female with a median age of 56, and the median time from drug initiation to psychiatric adverse event was 31 days (Chen et al., 2024).
EudraVigilance. An analysis of 31,444 European adverse event reports identified 372 psychiatric cases. Anxiety accounted for 38.7% (n=144), depression for 50.3% (n=187), and suicidal ideation for 19.6% (n=73). Nine deaths were reported (8 liraglutide, 1 semaglutide), with fatal cases occurring predominantly among men (8 of 9) (Tobaiqy & Elkout, 2024).
WHO VigiBase. The largest pharmacovigilance psychiatric analysis to date examined 2,061,901 total reports across dulaglutide, semaglutide, and liraglutide, of which 21,414 involved psychiatric adverse reactions. Semaglutide showed an adjusted ROR of 1.26 (95% CI 1.18–1.35) for anxiety and 1.70 (95% CI 1.57–1.84) for depressed mood. A sensitivity analysis restricted to reports filed before the weight-management approval date found no psychiatric disproportionality, which suggests reporting context and indication may matter (Nishida et al., 2025).
FAERS depression and suicide analysis. A separate FAERS analysis comparing semaglutide, liraglutide, and tirzepatide found semaglutide was the only agent with disproportionality for both depression (ROR 1.87) and suicide/self-injury (ROR 1.73). Neither liraglutide nor tirzepatide reached disproportionality for either outcome (Wang et al., 2025).
These databases point in the same general direction: psychiatric and arousal-related reports exist in post-marketing data, especially for semaglutide. They cannot prove causality, and they do not establish that nighttime anxiety occurs at a specific rate.
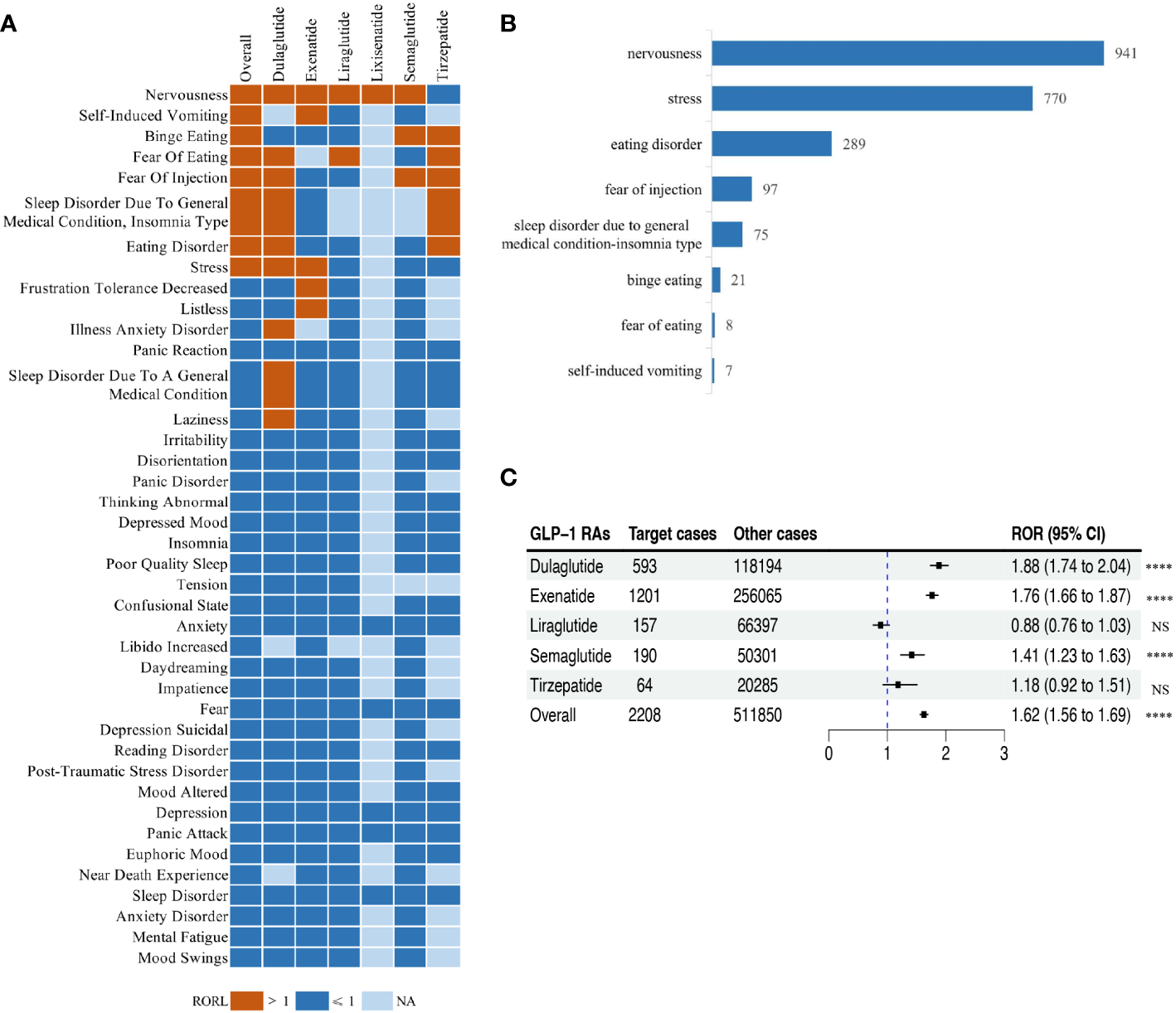
Why Do Controlled Trials Show No Anxiety When People Report It Consistently
The STEP 1–5 post-hoc analysis. Pooled data from four randomized controlled trials (STEP 1, 2, 3, and 5) included 3,681 participants without known major psychopathology at baseline. At week 68, semaglutide recipients had mean PHQ-9 scores of 2.0 ± 2.9 versus placebo at 2.4 ± 3.3 — statistically detectable but not considered clinically meaningful. Semaglutide recipients were less likely to move to a more severe PHQ-9 depression category (OR 0.63; 95% CI 0.50–0.79). Suicidal ideation was reported in 1% or fewer of both arms (Wadden et al., 2024).
Why the trials may miss nighttime anxiety. Several design features limit what these trials can say about nighttime arousal:
1. Excluding individuals with known major psychopathology limits generalizability to people with active psychiatric conditions.
2. The PHQ-9 measures depression severity; it does not isolate nighttime arousal, restlessness, or racing thoughts at bedtime.
3. No trial endpoint measured restlessness at bedtime or racing thoughts at night as a distinct outcome.
4. Psychiatric adverse-event categories are broader than the specific nighttime pattern described by some GLP-1 users.
Swedish national cohort. A Lancet Psychiatry study of 95,490 individuals with existing depression or anxiety found semaglutide was associated with lower risk of worsening mental illness (aHR 0.58; 95% CI 0.51–0.65). The population differs from pharmacovigilance reports: this study measured worsening of existing conditions, not onset of new nighttime anxiety in previously unaffected individuals (Taipale et al., 2026).
100-million-patient electronic health record study. Semaglutide showed no increased insomnia, anxiety, or depression risk versus three active comparators over 12 months. However, this was a type 2 diabetes population, and the results do not necessarily extend to higher-dose obesity use or to newer dual-agonist drugs (De Giorgi et al., 2024).
The synthesis. Large controlled and observational studies do not show population-level psychiatric harm in the groups studied. Pharmacovigilance can still capture uncommon or subgroup-specific reports that broad trials are less suited to characterize. Both findings can be true at the same time, and a 2026 review calls for dedicated prospective trials with prespecified psychiatric endpoints to clarify the psychiatric effects of GLP-1 receptor agonists (Sa et al., 2026).
How Does Semaglutide Affect the Brain’s Stress and Anxiety Circuits
Paraventricular nucleus (PVN). The PVN helps coordinate the HPA axis: CRH signaling can trigger ACTH and cortisol release. GLP-1 receptors are expressed in stress-related brain regions, and animal work links central GLP-1 signaling to endocrine and anxiety responses. Winzeler et al. (2019) showed no 24-hour cortisol increase from approved-dose dulaglutide, so persistent cortisol elevation is unlikely to explain every GLP-1-related arousal report.
Locus coeruleus (LC). The LC is a major noradrenaline source involved in arousal, autonomic responses, and vigilance. In rat work, LC GLP-1 receptors contributed to food-intake suppression, nausea-like behavior, and autonomic effects. That makes LC signaling a plausible contributor to restlessness or panic-like arousal, but direct evidence in humans taking semaglutide is still limited.
Nucleus of the solitary tract (NTS) to amygdala. The NTS receives visceral input from the gut and is part of the central GLP-1 network. Animal work shows central GLP-1 signaling can affect anxiety-related responses through the amygdala. If nausea or gastrointestinal discomfort is present at bedtime, visceral input may contribute to anxiety-like arousal.
Dopamine and serotonin modulation. GLP-1 receptors are expressed in brain regions involved in reward and mood. A 2026 review found preliminary evidence for modest antidepressant effects and potential psychiatric benefits in some contexts, while also noting inconsistent evidence and underrepresentation of people with psychiatric comorbidities (Sa et al., 2026).
Why nighttime is different. During waking hours, external activity competes with internal sensations. At bedtime, reduced external input can make arousal, nausea, palpitations, or glucose-related sensations more noticeable.
Nighttime anxiety on GLP-1 drugs can stem from orexin-related arousal, lower overnight fuel availability, autonomic activation, nausea-related visceral input, or stress-circuit signaling. These pathways can also overlap with hormonal, circadian, and metabolic causes of nighttime waking that exist independently of the drug. Naming the likely contributors is the first step toward a more specific response.
Find out which causes might be driving your nighttime anxiety →
Does Semaglutide Cause Panic Attacks or Just General Anxiety
The distinction between pharmacological arousal and panic disorder matters. If the timing tracks closely with dose escalation, appetite suppression, nausea, or overnight glucose instability, a drug-related arousal pattern becomes more plausible. Panic disorder, by contrast, is recurrent and often accompanied by avoidance behavior.
The FAERS analysis identified several arousal-adjacent categories — nervousness, fear of eating, and fear of injection — consistent with GLP-1 receptor agonists activating arousal-related pathways in some reports (Chen et al., 2024).
Is Nighttime Anxiety on Ozempic a Psychiatric Condition or a Drug Side Effect
This distinction has practical implications. A drug-related arousal pattern may track with dose changes, injection timing, reduced food intake, nausea, or sleep disruption. An anxiety disorder has its own pattern and may persist outside the medication timeline. If anxiety persists or intensifies over weeks, discussion with a qualified medical professional is reasonable (Taipale et al., 2026).
Does Ozempic Anxiety Improve When You Stop Taking the Drug
The washout timeline matters for interpretation. If anxiety improves as semaglutide exposure falls, that would be consistent with a drug-related contributor. If anxiety persists after drug levels have fallen, another contributor may be involved. The current research does not provide a measured anxiety-resolution timeline after stopping semaglutide.
Can Anti-Anxiety Medication Help With GLP-1-Related Nighttime Restlessness
GLP-1 signaling can excite orexin neurons in preclinical models, and DORAs block orexin receptors. That makes orexin a rational discussion point for medication review, not a proven treatment path for semaglutide-related nighttime anxiety. Any medication decision depends on individual circumstances, other medications, and whether the anxiety is dose-related or persistent.
Does Mounjaro Cause the Same Nighttime Anxiety as Ozempic
The Wang et al. (2025) FAERS analysis found tirzepatide showed no psychiatric disproportionality versus semaglutide’s ROR of 1.87 for depression and 1.73 for suicide/self-injury. The authors suggested tirzepatide may be a more appropriate choice for some people with psychiatric comorbidities, pending dedicated head-to-head comparisons (Wang et al., 2025).
Whether tirzepatide’s dual GIP/GLP-1 agonism produces different arousal-circuit effects is not yet established. The GIP receptor component may affect the overall GLP-1 response differently, but this remains an open question.
Related Reading
- Metabolic Sleep Disruption — The full metabolic sleep framework covering glucose regulation, insulin resistance, mitochondrial decline, NAD+ loss, and fat metabolism
- Does Ozempic Cause Insomnia What GLP-1 Drugs Do to Your Sleep — The broad overview of GLP-1 sleep side effects and mechanisms
- Why Does Ozempic Make You Tired But Keep You Awake — How GLP-1 drugs activate orexin/wakefulness circuitry while appetite suppression can create fatigue
- Why Does Ozempic Wake You Up at 3am — How lower overnight fuel availability and counter-regulatory hormones can contribute to 3am waking
- Does Ozempic Give You Vivid Dreams — How GLP-1 drugs may affect dream intensity through arousal and neuropsychiatric pathways
- Does Eating Too Little on Ozempic Cause Insomnia — How appetite suppression can affect tryptophan, serotonin, melatonin, and overnight glucose stability
- Does Your Ozempic Injection Day Disrupt Your Sleep The 24-48 Hour Pattern — Why sleep disruption can follow a weekly pattern tied to injection timing and peak drug exposure
- Does Your Semaglutide Dose Increase Restart Your Insomnia Why Each Titration Can Disrupt Sleep Again — Why each titration step can restart sleep disruption before adaptation catches up
- Can You Take Melatonin, Magnesium, or Sleep Aids While on Ozempic — How melatonin, magnesium, DORAs, and other sleep aids intersect with GLP-1 physiology
- Do GLP-1 Drugs Disrupt Your Body Clock Ozempic, Circadian Rhythms, and Melatonin — How GLP-1 secretion rhythms, food timing, peripheral clocks, and melatonin intersect
- Ozempic Night Sweats: How GLP-1 Drugs May Affect Temperature at Night — How GLP-1 drugs may affect nighttime sweating through autonomic tone and thermoregulation
- Ozempic Nausea and Sleep: How Stomach Side Effects Keep You Awake at Night — How delayed gastric emptying, reflux, and nausea can fragment sleep
- Will Your Sleep Go Back to Normal When You Stop Ozempic — What is known and not known about sleep after semaglutide discontinuation
- Does Ozempic Muscle Loss Affect Your Sleep Sarcopenia, Temperature, and Deep Sleep — How lean-mass loss, thermoregulation, and deep sleep can interact during GLP-1 weight loss
- Could Ozempic Contribute to Restless Legs at Night Iron, B12, and the Dopamine Connection — How iron, B12, dopamine, and rapid weight loss may intersect with restless legs at night
- Does Compounded Semaglutide Affect Sleep Differently Than Brand-Name Ozempic — How dose variability, formulation differences, and adverse-event tracking can affect sleep side effects
- Ozempic and Sleep After 40: Why GLP-1 Side Effects Can Feel Different as You Age — Why GLP-1 sleep effects can land differently after 40 because of muscle, hormones, and glucose regulation
References
- Acuna-Goycolea, C., & van den Pol, A. N. (2004). Glucagon-like peptide 1 excites hypocretin/orexin neurons by direct and indirect mechanisms: Implications for viscera-mediated arousal. The Journal of Neuroscience, 24(37), 8141–8152. https://pubmed.ncbi.nlm.nih.gov/15371515/
- Arillotta, D., Floresta, G., Guirguis, A., Corkery, J. M., Catalani, V., Martinotti, G., Sensi, S. L., & Schifano, F. (2023). GLP-1 receptor agonists and related mental health issues; insights from a range of social media platforms using a mixed-methods approach. Brain Sciences, 13(11), 1503. https://pubmed.ncbi.nlm.nih.gov/38002464/
- Chen, W., Cai, P., Zou, W., & Fu, Z. (2024). Psychiatric adverse events associated with GLP-1 receptor agonists: A real-world pharmacovigilance study based on the FDA Adverse Event Reporting System database. Frontiers in Endocrinology, 15, 1330936. https://pubmed.ncbi.nlm.nih.gov/38390214/
- De Giorgi, R., Koychev, I., Adler, A. I., Cowen, P. J., Harmer, C. J., Harrison, P. J., & Taquet, M. (2024). 12-month neurological and psychiatric outcomes of semaglutide use for type 2 diabetes: A propensity-score matched cohort study. EClinicalMedicine, 74, 102726. https://pubmed.ncbi.nlm.nih.gov/39764175/
- Fortin, S. M., Lipsky, R. K., Lhamo, R., Chen, J., Kim, E., Borner, T., Schmidt, H. D., & Hayes, M. R. (2023). The locus coeruleus contributes to the anorectic, nausea, and autonomic physiological effects of glucagon-like peptide-1. Science Advances, 9(39), eadh0980. https://pubmed.ncbi.nlm.nih.gov/37729419/
- Gabery, S., Salinas, C. G., Paulsen, S. J., Ahnfelt-Rønne, J., Alanentalo, T., Baquero, A. F., Buckley, S. T., Farkas, E., Fekete, C., Frederiksen, K. S., Helms, H. C. C., Jeppesen, J. F., John, L. M., Pyke, C., Nøhr, J., Lu, T. T., Polex-Wolf, J., Prevot, V., Raun, K., … Secher, A. (2020). Semaglutide lowers body weight in rodents via distributed neural pathways. JCI Insight, 5(6), e133429. https://pubmed.ncbi.nlm.nih.gov/32213703/
- Kinzig, K. P., D’Alessio, D. A., Herman, J. P., Sakai, R. R., Vahl, T. P., Figueiredo, H. F., Murphy, E. K., & Seeley, R. J. (2003). CNS glucagon-like peptide-1 receptors mediate endocrine and anxiety responses to interoceptive and psychogenic stressors. The Journal of Neuroscience, 23(15), 6163–6170. https://pubmed.ncbi.nlm.nih.gov/12867498/
- Nishida, K., Chrétien, B., Dolladille, C., Ebina, T., Aleksic, B., Cabé, N., Savey, V., Onoue, T., & Yatsuya, H. (2025). Psychiatric and psychological adverse effects associated with dulaglutide, semaglutide, and liraglutide: A vigibase study. Clinical Nutrition, 51, 252–265. https://pubmed.ncbi.nlm.nih.gov/40617160/
- Sa, B., Maristany, A., Subramaniam, A., Guillen, R., Buonocore, B., Smith, A., Oldak, S. E., & Padilla, V. (2026). Psychiatric effects of GLP-1 receptor agonists: A systematic review of emerging evidence. Diabetes, Obesity & Metabolism, 28(1), 50–59. https://pubmed.ncbi.nlm.nih.gov/41126551/
- Schultes, B., Jauch-Chara, K., Gais, S., Hallschmid, M., Reiprich, E., Kern, W., Oltmanns, K. M., Peters, A., Fehm, H. L., & Born, J. (2007). Defective awakening response to nocturnal hypoglycemia in patients with type 1 diabetes mellitus. PLoS Medicine, 4(2), e69. https://pubmed.ncbi.nlm.nih.gov/17326710/
- Taipale, H., Taylor, M., Lähteenvuo, M., Mittendorfer-Rutz, E., Tanskanen, A., & Tiihonen, J. (2026). Association between GLP-1 receptor agonist use and worsening mental illness in people with depression and anxiety in Sweden: A national cohort study. The Lancet Psychiatry, 13(4), 327–335. https://pubmed.ncbi.nlm.nih.gov/41862258/
- Tobaiqy, M., & Elkout, H. (2024). Psychiatric adverse events associated with semaglutide, liraglutide and tirzepatide: A pharmacovigilance analysis of individual case safety reports submitted to the EudraVigilance database. International Journal of Clinical Pharmacy, 46(2), 488–495. https://pubmed.ncbi.nlm.nih.gov/38265519/
- Wadden, T. A., Brown, G. K., Egebjerg, C., Frenkel, O., Goldman, B., Kushner, R. F., McGowan, B., Overvad, M., & Fink-Jensen, A. (2024). Psychiatric safety of semaglutide for weight management in people without known major psychopathology: Post hoc analysis of the STEP 1, 2, 3, and 5 trials. JAMA Internal Medicine, 184(11), 1290–1300. https://pubmed.ncbi.nlm.nih.gov/39226070/
- Wang, M., Yang, Z., Yan, M., Liu, S., & Xiao, S. (2025). Depression and suicide/self-injury signals for weight loss medications: A disproportionality analysis of semaglutide, liraglutide, and tirzepatide in FAERS database. Journal of Affective Disorders, 389, 119670. https://pubmed.ncbi.nlm.nih.gov/40523410/
- Winrow, C. J., & Renger, J. J. (2014). Discovery and development of orexin receptor antagonists as therapeutics for insomnia. British Journal of Pharmacology, 171(2), 283–293. https://pubmed.ncbi.nlm.nih.gov/23731216/
- Winzeler, B., da Conceição, I., Refardt, J., Sailer, C. O., Dutilh, G., & Christ-Crain, M. (2019). Effects of glucagon-like peptide-1 receptor agonists on hypothalamic-pituitary-adrenal axis in healthy volunteers. The Journal of Clinical Endocrinology and Metabolism, 104(1), 202–208. https://pubmed.ncbi.nlm.nih.gov/30272170/
- Yang, X.-D., & Yang, Y.-Y. (2024). Clinical pharmacokinetics of semaglutide: A systematic review. Drug Design, Development and Therapy, 18, 2555–2570. https://pubmed.ncbi.nlm.nih.gov/38952487/
Written by Kat Fu, M.S., M.S. – Last reviewed: May 2026 – 17 references cited
