A recurring pattern across GLP-1 drug user communities is that the worst sleep of the week reliably falls on injection day or the night after. This maps directly onto the drug’s pharmacokinetic profile — it is predictable, not coincidental. Medical content on semaglutide side effects rarely connects injection timing to sleep quality.
This article explains why the 24-48 hour post-injection window produces the worst sleep, whether morning versus evening injection changes the outcome, and how long the injection-day pattern persists across the duration of use. It does not cover all GLP-1 sleep mechanisms; for the full overview, see the parent article.
GLP-1 drugs disrupt sleep through several distinct pathways. This article covers the injection-timing pathway — when the drug’s peak concentration collides with your sleep window. Other articles in this series cover the orexin paradox, blood sugar crashes, and dose escalation effects.
Why Is Insomnia Worst 24-48 Hours After Your Ozempic Injection
Semaglutide’s subcutaneous formulation has a half-life of approximately 168 hours (one week), supporting once-weekly dosing. Peak plasma concentration occurs within the first 1-3 days post-injection, and the magnitude of that peak is dose-dependent — higher therapeutic doses produce higher absolute Cmax values. A 2024 review of 17 pharmacokinetic studies documented this dose-dependent Cmax and the 1-3 day peak window for the subcutaneous formulation (Yang & Yang, 2024).
The reason semaglutide lasts so long is structural. Native GLP-1 — the hormone your gut produces after eating — has a plasma half-life of approximately 2 minutes. Semaglutide’s structural modifications — including C-18 fatty diacid conjugation enabling albumin binding — extend the effective half-life from 2 minutes to approximately 168 hours (Min et al., 2025). This engineering is what makes once-weekly dosing possible, but it also means the drug produces sustained concentration peaks rather than rapid clearance.
The side-effect burden during the Cmax window is well-quantified. A 2025 meta-analysis of 54,972 randomized participants at increased cardiovascular risk across 50 trials found that semaglutide increases nausea risk by a relative risk of 3.00 (95% CI 2.63-3.42) and vomiting by RR 4.12 (95% CI 3.47-4.90) compared to placebo (Sillassen et al., 2025). These rates reflect a cardiovascular-risk population and may differ in general obesity contexts; both peak during the Cmax window. Nausea and vomiting during nighttime hours directly disrupt both sleep onset and sleep maintenance.
GLP-1 receptors are present in brain regions directly involved in sleep-wake regulation: the hypothalamus, locus coeruleus, and nucleus tractus solitarius. A 2025 pharmacovigilance analysis of over 2 million individual case safety reports from the WHO VigiBase database identified anxiety (adjusted reporting odds ratio 1.26) and depressed mood (aROR 1.70) as elevated reports for semaglutide (Nishida et al., 2025). Both anxiety and depression share neurobiological mechanisms with insomnia through serotonergic, noradrenergic, and corticotropin-releasing hormone pathways — a connection well-established in psychiatric sleep literature, though not explored in the pharmacovigilance data itself. At Cmax-level drug concentrations, these CNS effects are at their strongest.
The injection-day insomnia pattern follows from this pharmacology: inject in the evening, and peak drug levels coincide with your sleep window on nights 1-2. Inject in the morning, and peak levels occur during daytime hours on days 1-2.
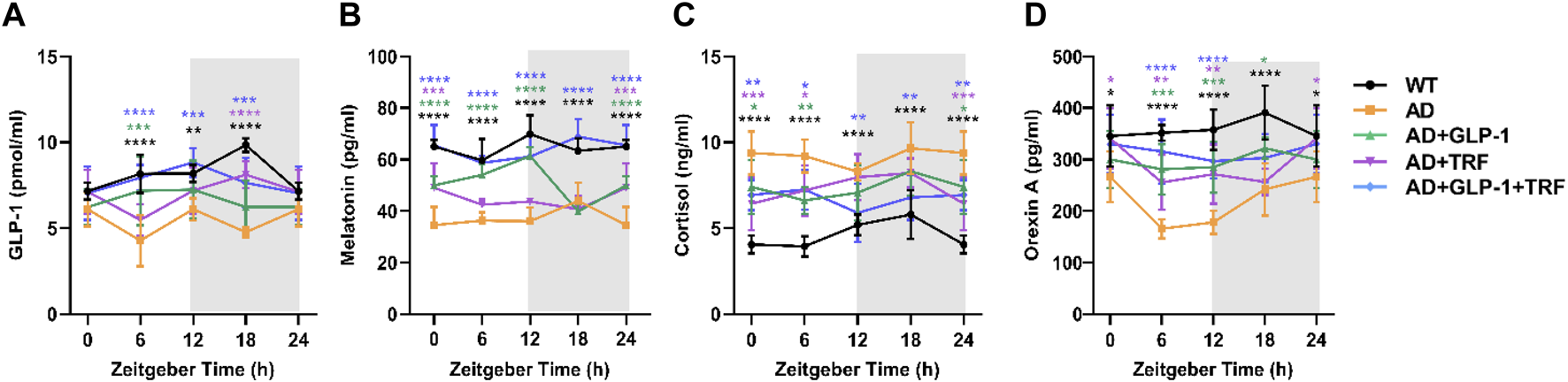
Does Injecting Ozempic in the Morning Instead of at Night Improve Sleep
The pharmacokinetic reasoning is direct. Morning injection places Cmax at hours 24-72 (daytime on days 2-3). Evening injection places Cmax at hours 24-72 (nighttime on nights 1-2). The total side-effect burden is the same in both scenarios — nausea, GI disruption, and CNS arousal are dose-determined, not timing-determined. What changes is whether peak drug exposure overlaps with your sleep window.
A 2022 preclinical study provided evidence that GLP-1 injection timing is not pharmacologically neutral with respect to circadian rhythms. Dong et al. (2022) demonstrated in mouse models that timed GLP-1 injection combined with time-restricted feeding improved hypothalamic clock gene expression — specifically Bmal1, Clock, and Dbp — partially restoring activity-rest cycles, core body temperature rhythms, and hormone secretion timing, though treated mice did not fully reach wild-type levels. The combination of timed GLP-1 injection with time-restricted feeding was more effective than either intervention alone.
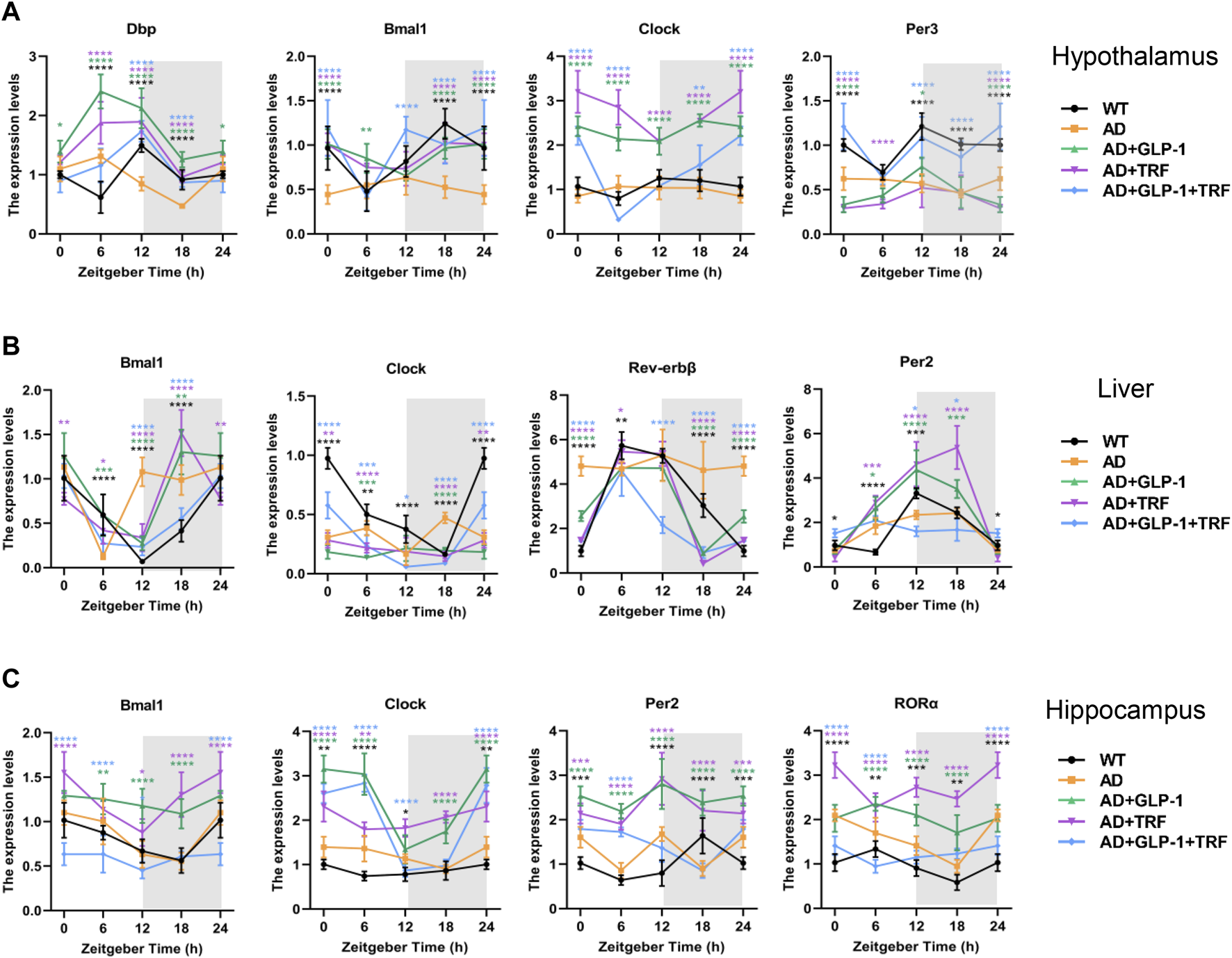
The circadian dimension extends beyond injection timing. Gil-Lozano et al. (2014) identified an intrinsic circadian clock within intestinal L cells — the cells that produce endogenous GLP-1 — driving diurnal GLP-1 secretory rhythms. The highest GLP-1 release occurs just before the normal feeding period. This means your body’s own GLP-1 production peaks before daytime meals. Brubaker and Gil-Lozano (2016) built on this finding, proposing GLP-1 as “a functional component of the peripheral metabolic clock.” Aligning exogenous semaglutide with this endogenous GLP-1 circadian peak — by injecting in the morning — may reduce circadian disruption while keeping the pharmacodynamic response consistent.
One important distinction: the injection-timing question applies only to subcutaneous formulations (Ozempic, Wegovy). Oral semaglutide (Rybelsus) must be taken in the morning in the fasting state with up to 120 mL of water, followed by a 30-minute eating delay — a regulatory requirement established by pharmacokinetic studies showing that food substantially reduces or eliminates oral semaglutide absorption (Baekdal et al., 2021; van Hout et al., 2023). For oral semaglutide, there is no timing flexibility.
Caveat: No randomized controlled trial has compared morning versus evening injection timing for sleep outcomes. The reasoning above is based on pharmacokinetic data and preclinical circadian research, not trial evidence.
How Long Does Injection-Day Insomnia Last on Ozempic
The within-week pattern follows the drug’s plasma concentration curve. Cmax occurs at days 1-3 post-injection, followed by a gradual decline toward the trough concentration before the next weekly injection. Sleep disruption tracks this curve: worst on nights 1-2, improving through days 4-7 as drug levels decrease.
Across multiple weeks at the same dose, adaptation occurs. The Sillassen et al. (2025) meta-analysis of 54,972 participants documented that gastrointestinal adverse events — nausea, vomiting, diarrhea, constipation — are significantly elevated with semaglutide use. Clinical experience and prescribing information indicate these effects are typically most pronounced during dose-escalation phases and tend to attenuate at stable doses. Since gastrointestinal effects — nausea, reflux, gastric discomfort — are a primary driver of injection-day sleep disruption, their resolution predicts improved sleep at stable doses.
The distinction between gastrointestinal and CNS contributions matters for predicting duration. Mifsud et al. (2025) reviewed GLP-1 receptor agonist effects on sleep and distinguished between weight-mediated sleep improvements (progressive, developing over months as body weight decreases) and direct CNS or respiratory effects. The injection-day pattern reflects the acute CNS and GI component — not the slow, chronic weight-loss component. GI-driven sleep disruption resolves faster as tolerance develops. Brain-level effects on arousal circuits may adapt more slowly because orexin neuron activation is a direct pharmacological effect, not mediated by nausea or GI discomfort.
Each dose escalation resets the adaptation. Moving from 0.25 mg to 0.5 mg to 1.0 mg to 2.0 mg (Ozempic) or 2.4 mg (Wegovy) produces a new, higher Cmax at each step that restarts the side-effect adaptation process. Users at a stable maintenance dose for several months typically report reduced injection-day disruption, though this is community-reported and not trial-measured.
Does Injection Timing Affect Sleep Differently on Mounjaro or Zepbound
Tirzepatide shares the structural engineering that produces sustained plasma peaks. Like semaglutide, tirzepatide uses albumin-binding half-life extension to achieve once-weekly dosing. Min et al. (2025) documented Tmax ranges for both drugs — semaglutide 36-60 hours, tirzepatide 8-72 hours — both broadly within the first 1-3 days post-injection. Tirzepatide’s half-life is approximately 5 days compared to semaglutide’s approximately 7 days, but the Cmax timing is similar.
The same injection-timing logic applies: morning injection moves peak drug exposure to daytime regardless of which GLP-1 receptor agonist is used. Tirzepatide has the same nausea and vomiting burden during the Cmax window, so the strategy of injecting in the morning to keep peak side effects away from the sleep window applies equally.
Tirzepatide’s dual GIP/GLP-1 agonism adds one unknown variable. GIP receptors have a partially overlapping but distinct brain distribution from GLP-1 receptors. Whether GIP receptor activation modifies the injection-day sleep pattern is not yet established in published studies.
Mifsud et al. (2025) highlighted a landmark development: tirzepatide’s FDA approval as the first medication for obstructive sleep apnea. This represents the first time a metabolic medication was approved for a sleep disorder. The authors proposed reframing OSA “as a chronic metabolic disease rather than solely a mechanical airway disorder” — a reconceptualization that underscores how directly GLP-1 class drugs interact with sleep physiology. If these drugs carry FDA approval for a sleep condition, their ability to disrupt sleep through the same receptor pathways is pharmacologically expected.
Injection-day insomnia is one pattern within a larger picture of metabolic sleep disruption. GLP-1 drugs also affect orexin-driven arousal, blood sugar stability, tryptophan availability, and circadian timing — each of which may compound the injection-day pattern. Sleep disruption from metabolic causes often overlaps with hormonal, circadian, or autonomic factors that amplify each other.
Find out which causes might be driving your 3am wakeups →
Does the Insomnia Pattern Restart With Every Weekly Ozempic Injection
The weekly rhythm in concrete terms: night 1 (injection day) and night 2 tend to carry the worst sleep quality, as Cmax-driven nausea and CNS arousal are at their peak. By nights 4-5, plasma levels have declined toward mid-week values, and sleep quality approaches pre-injection baseline. Night 7 (the night before the next injection) is typically the best sleep night of the cycle, as drug levels are at their weekly trough.
This pattern tends to be strongest in the first 4-8 weeks at each dose level, based on community reports. The Sillassen et al. (2025) meta-analysis data — showing significantly elevated gastrointestinal adverse event rates — combined with clinical experience indicating GI tolerance development, supports the expectation that the weekly cycle attenuates over time. The CNS arousal component may persist longer, but community reports indicate that many users notice reduced injection-day disruption after several weeks at the same dose.
Can Splitting Your Semaglutide Dose Reduce Injection-Night Insomnia
The pharmacokinetic logic: if a single weekly injection of 1.0 mg produces a given Cmax, two injections of 0.5 mg separated by 3-4 days would each produce a lower individual Cmax while maintaining a similar average steady-state concentration. Lower peaks mean less intense nausea, less acute CNS arousal, and less GI disruption during each peak window (Yang & Yang, 2024).
No published trial has tested split dosing for sleep outcomes. The concept parallels flexible titration strategies — slower dose increases have achieved better tolerability in dose-escalation studies. Split dosing requires physician approval because it modifies the drug’s pharmacokinetic profile in ways that may affect efficacy, and because the injection pen is calibrated for specific single doses.
Does Injection-Day Insomnia Get Better Over Time on a Stable Dose
The two components of injection-day sleep disruption adapt on different timelines. GI-driven disruption (nausea, reflux, gastric discomfort keeping you awake) resolves as pharmacological tolerance to gastrointestinal effects develops — a pattern well-documented in clinical practice though not specifically quantified in the Sillassen meta-analysis. CNS-driven arousal (orexin neuron activation, heightened alertness, anxiety-like restlessness) is a direct pharmacological effect of GLP-1 receptor binding in the hypothalamus and brainstem, and may persist as long as drug concentrations reach those brain regions.
Community reports suggest 4-8 weeks at a stable dose before injection-day sleep quality improves. This timeline aligns with the GI adaptation curve but does not necessarily reflect CNS adaptation.
Should You Avoid Caffeine on Your Ozempic Injection Day
The additive arousal effect is pharmacologically predictable. Semaglutide activates orexin neurons through GLP-1 receptors in the lateral hypothalamus, producing wakefulness output. Caffeine blocks adenosine A1 and A2A receptors, preventing the sleep-pressure molecule adenosine from promoting drowsiness. Both inputs converge on the same outcome — heightened cortical arousal — through independent mechanisms. On injection day, when semaglutide-driven arousal is at its Cmax peak, adding caffeine creates a compounding wakefulness effect.
This is logical inference from established pharmacology, not direct evidence from a controlled study. No study has measured the interaction between caffeine timing and GLP-1 receptor agonist injection timing on sleep outcomes.
Does the Injection Site Affect Ozempic Sleep Side Effects
Subcutaneous absorption rate can vary between sites — abdominal fat and thigh tissue have different blood flow characteristics — but semaglutide’s albumin-binding half-life extension mechanism dominates the pharmacokinetic profile regardless of initial absorption rate differences. The approximately 168-hour half-life means that small differences in the speed of initial absorption from the injection site are pharmacokinetically minor compared to the drug’s prolonged circulation time (Min et al., 2025). By the time Cmax is reached at 1-3 days post-injection, the drug has fully entered general circulation regardless of where it was injected.
Related Reading
- Metabolic Sleep Disruption — The full metabolic sleep framework covering glucose regulation, insulin resistance, mitochondrial decline, NAD+ loss, and fat metabolism
- Does Ozempic Cause Insomnia What GLP-1 Drugs Do to Your Sleep — The broad overview of GLP-1 sleep side effects and mechanisms
- Why Does Ozempic Make You Tired But Keep You Awake — How GLP-1 drugs activate orexin/wakefulness circuitry while appetite suppression can create fatigue
- Why Does Ozempic Wake You Up at 3am — How lower overnight fuel availability and counter-regulatory hormones can contribute to 3am waking
- Does Ozempic Give You Vivid Dreams — How GLP-1 drugs may affect dream intensity through arousal and neuropsychiatric pathways
- Does Eating Too Little on Ozempic Cause Insomnia — How appetite suppression can affect tryptophan, serotonin, melatonin, and overnight glucose stability
- Does Your Semaglutide Dose Increase Restart Your Insomnia Why Each Titration Can Disrupt Sleep Again — Why each titration step can restart sleep disruption before adaptation catches up
- Can You Take Melatonin, Magnesium, or Sleep Aids While on Ozempic — How melatonin, magnesium, DORAs, and other sleep aids intersect with GLP-1 physiology
- Does Ozempic Give You Anxiety at Night Why GLP-1 Drugs May Contribute to Nighttime Panic — How orexin activation, HPA-axis signaling, and glucose instability may contribute to nighttime anxiety
- Do GLP-1 Drugs Disrupt Your Body Clock Ozempic, Circadian Rhythms, and Melatonin — How GLP-1 secretion rhythms, food timing, peripheral clocks, and melatonin intersect
- Ozempic Night Sweats: How GLP-1 Drugs May Affect Temperature at Night — How GLP-1 drugs may affect nighttime sweating through autonomic tone and thermoregulation
- Ozempic Nausea and Sleep: How Stomach Side Effects Keep You Awake at Night — How delayed gastric emptying, reflux, and nausea can fragment sleep
- Will Your Sleep Go Back to Normal When You Stop Ozempic — What is known and not known about sleep after semaglutide discontinuation
- Does Ozempic Muscle Loss Affect Your Sleep Sarcopenia, Temperature, and Deep Sleep — How lean-mass loss, thermoregulation, and deep sleep can interact during GLP-1 weight loss
- Could Ozempic Contribute to Restless Legs at Night Iron, B12, and the Dopamine Connection — How iron, B12, dopamine, and rapid weight loss may intersect with restless legs at night
- Does Compounded Semaglutide Affect Sleep Differently Than Brand-Name Ozempic — How dose variability, formulation differences, and adverse-event tracking can affect sleep side effects
- Ozempic and Sleep After 40: Why GLP-1 Side Effects Can Feel Different as You Age — Why GLP-1 sleep effects can land differently after 40 because of muscle, hormones, and glucose regulation
References
- Baekdal, T. A., Breitschaft, A., Donsmark, M., Maarbjerg, S. J., Sondergaard, F. L., & Borregaard, J. (2021). Effect of various dosing conditions on the pharmacokinetics of oral semaglutide, a human glucagon-like peptide-1 analogue in a tablet formulation. Diabetes Therapy, 12(7), 1915-1927. https://pubmed.ncbi.nlm.nih.gov/34080123/
- Brubaker, P. L., & Gil-Lozano, M. (2016). Glucagon-like peptide-1: The missing link in the metabolic clock? Journal of Diabetes Investigation, 7(Suppl 1), 70-75. https://pubmed.ncbi.nlm.nih.gov/27186359/
- Dong, Y., Cheng, L., & Zhao, Y. (2022). Resetting the circadian clock of Alzheimer’s mice via GLP-1 injection combined with time-restricted feeding. Frontiers in Physiology, 13, 911437. https://pubmed.ncbi.nlm.nih.gov/36148311/
- Gil-Lozano, M., Mingomataj, E. L., Wu, W. K., Ridout, S. A., & Brubaker, P. L. (2014). Circadian secretion of the intestinal hormone GLP-1 by the rodent L cell. Diabetes, 63(11), 3674-3685. https://pubmed.ncbi.nlm.nih.gov/24789917/
- Mifsud, C. S., Kolla, B. P., Rushlow, D. R., & Mansukhani, M. P. (2025). The impact of GLP-1 agonists on sleep disorders: Spotlight on sleep apnea. Expert Opinion on Pharmacotherapy, 26(14-15), 1529-1538. https://pubmed.ncbi.nlm.nih.gov/41114602/
- Min, J. S., Jo, S. J., Lee, S., Kim, D. Y., Kim, D. H., Lee, C. B., & Bae, S. K. (2025). A comprehensive review on the pharmacokinetics and drug-drug interactions of approved GLP-1 receptor agonists and a dual GLP-1/GIP receptor agonist. Drug Design, Development and Therapy, 19, 3509-3537. https://pubmed.ncbi.nlm.nih.gov/40330819/
- Nishida, K., Chretien, B., Dolladille, C., Ebina, T., Aleksic, B., Cabe, N., Savey, V., Onoue, T., & Yatsuya, H. (2025). Psychiatric and psychological adverse effects associated with dulaglutide, semaglutide, and liraglutide: A VigiBase study. Clinical Nutrition, 51, 252-265. https://pubmed.ncbi.nlm.nih.gov/40617160/
- Sillassen, C. D. B., Petersen, J. J., Faltermeier, P., Yucel, D., Siddiqui, F., Andersen, R. K., Graever, L., Bjerg, J. L., Kamp, C. B., Grand, J., Dominguez, H., Frolich, A., Gaede, P., Gluud, C., Mathiesen, O., & Jakobsen, J. C. (2025). The adverse effects associated with semaglutide use in patients at increased risk of cardiovascular events: A systematic review with meta-analysis and Trial Sequential Analysis. BMC Medicine, 23(1), 654. https://pubmed.ncbi.nlm.nih.gov/41286875/
- van Hout, M., Forte, P., Jensen, T. B., Boschini, C., & Baekdal, T. A. (2023). Effect of various dosing schedules on the pharmacokinetics of oral semaglutide: A randomised trial in healthy subjects. Clinical Pharmacokinetics, 62(4), 635-644. https://pubmed.ncbi.nlm.nih.gov/36932262/
- Yang, X. D., & Yang, Y. Y. (2024). Clinical pharmacokinetics of semaglutide: A systematic review. Drug Design, Development and Therapy, 18, 2555-2570. https://pubmed.ncbi.nlm.nih.gov/38952487/
Written by Kat Fu, M.S., M.S. – Last reviewed: May 2026 – 10 references cited
