Leptin is the satiety hormone produced by adipocytes (fat cells) that tells the hypothalamus energy stores are sufficient. In leptin resistance, the brain stops receiving that message despite high circulating leptin levels. This state is common in obesity, driven by impaired blood-brain barrier transport, JAK2-STAT3 transduction breakdown, and chronic hypothalamic inflammation. The sleep connection is underreported: leptin receptors sit in the same hypothalamic nuclei that regulate arousal and sleep-wake transitions.
This article covers the evidence linking leptin resistance to insomnia, the mechanisms driving the bidirectional loop, and what breaks the cycle. For the full overview of how metabolic impairment drives middle-of-the-night wakeups, see the parent pillar: Metabolic Sleep Disruption.
How Does Sleep Loss Change Leptin and Ghrelin?
The first large-scale evidence came from the Wisconsin Sleep Cohort (n = 1,024). Those sleeping 5 hours versus 8 hours had 15.5% lower leptin (p = 0.01) and 14.9% higher ghrelin, independent of BMI. A U-shaped relationship between sleep duration and BMI emerged in the 74.4% sleeping under 8 hours (Taheri et al., 2004).
The causal direction was established the same year. A randomized crossover study in 12 healthy young men showed that two days of sleep restriction (4 hours per night) produced an 18% leptin decrease (p = 0.04), a 28% ghrelin increase (p < 0.04), a 24% increase in hunger (p < 0.01), and a 33-45% increase in appetite for high-carbohydrate foods (p = 0.02). The within-subject design eliminates body composition as a confounder (Spiegel et al., 2004).
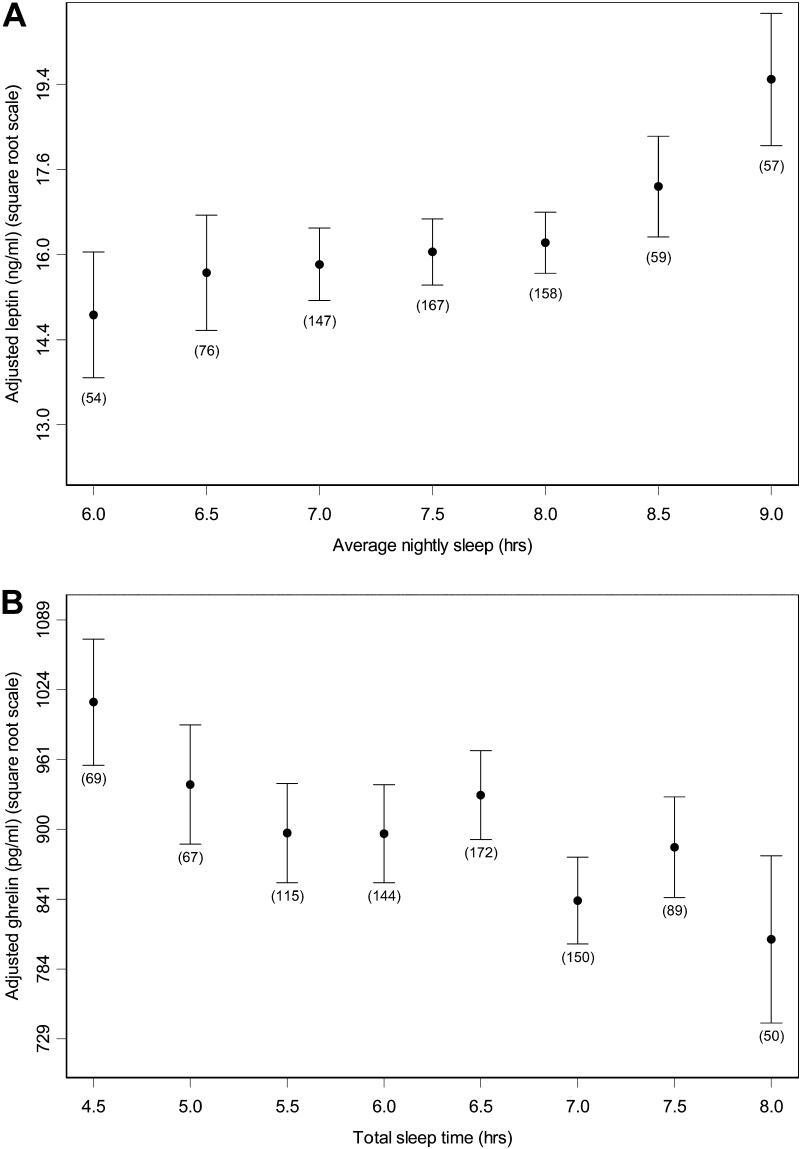
A 2020 meta-analysis of 21 studies (n = 2,250) supported the pattern: short sleep elevated ghrelin (SMD = 0.14, p = 0.01). Total sleep deprivation raised both leptin (SMD = 0.24, p = 0.001) and ghrelin (SMD = 0.18, p = 0.01). The paradoxical acute leptin increase under total deprivation likely reflects stress-driven counter-regulation rather than the chronic suppression seen in habitual short sleepers (Lin et al., 2020).
The combined effect — suppressed satiety messaging plus amplified hunger messaging — creates a dual appetite-stimulating state that persists as long as sleep remains restricted. The downstream weight gain from increased caloric intake feeds into leptin resistance mechanisms.
What Causes Leptin Resistance — and Why Does It Affect Sleep?
Multiple convergent pathways produce leptin resistance. A 2024 mechanistic review catalogued the primary routes: chronic receptor downregulation from sustained hyperleptinemia (high leptin from excess fat mass), impaired JAK2-STAT3 intracellular transduction, reduced blood-brain barrier transport (hypertriglyceridemia inhibits leptin’s passage to the brain), SOCS3 upregulation (which dampens receptor response), increased mTOR activity, and endoplasmic reticulum stress. Each pathway creates a self-reinforcing block where abundant circulating leptin does not reach or activate satiety centers (Engin, 2024).
A September 2025 synthesis adds further convergent mechanisms: defective hypothalamic autophagy (the cell’s self-cleaning process), chronic low-grade inflammation, decreased leptin receptor expression, and elevated PTP1B (protein tyrosine phosphatase 1B). No single established therapy reliably reverses leptin resistance in common obesity — the resistance involves multiple concurrent blocks and is self-sustaining (Hu et al., 2025).

The connection to sleep is anatomical. Leptin receptor-expressing neurons in the dorsomedial hypothalamus (DMH) and the lateral hypothalamic area (LHA) regulate both energy balance and arousal state. When leptin can no longer activate these neurons, the brain maintains wakefulness as if the body is energy-depleted — a starvation-alert response that persists despite excess body fat.
The result: a person with obesity who is simultaneously resistant to satiety messaging and unable to achieve consolidated sleep. Sleep loss then further suppresses leptin and promotes weight gain, deepening the resistance and closing the loop.
Does Sleep Architecture Change Leptin Regulation?
Total sleep time is not the full picture. A polysomnography study (n = 58) found that greater REM sleep percentage was associated with more pronounced overnight leptin reduction, independent of total sleep time, age, gender, and body fat percentage. REM-specific processes modulate nocturnal leptin secretion independently of how many hours a person sleeps (Olson et al., 2016).
This matters for factors that suppress REM: alcohol, certain antidepressants (particularly SSRIs), and aging all reduce REM percentage and may alter leptin regulation through a pathway that total sleep duration alone would not capture.
Chronic insomnia produces a different hormonal pattern. A study comparing 14 men with primary insomnia to 24 matched controls found suppressed nighttime ghrelin at all three timepoints (11 PM, 2 AM, 6 AM; p < 0.0001), while leptin did not differ. Those with insomnia also showed less stage 2 and REM sleep and decreased sleep efficiency (Motivala et al., 2009).
This ghrelin suppression is the opposite of experimental sleep deprivation, where ghrelin rises. Insomnia appears to represent a hyperarousal-driven neuroendocrine state rather than accumulated sleep loss, and approaches to improvement may need to address the underlying hyperarousal rather than sleep duration alone.
A 2024 population study (n = 881) added another dimension: higher leptin may intensify the relationship between poor sleep and memory complaints. Individuals with elevated leptin levels may experience greater cognitive consequences from poor quality sleep (Mosini et al., 2024).
What Breaks the Leptin-Insomnia Cycle?
A 2025 bidirectional review framed sleep disorder management as a metabolic step. Sleep deprivation disrupts leptin and ghrelin regulation, increases caloric intake, and reduces physical activity, driving fat accumulation. Simultaneously, obesity worsens sleep quality through mechanical, hormonal, and inflammatory pathways. The review recommends integrating sleep quality assessment into metabolic health work and positions mental health, physical health, and sleep quality as co-equal targets (Figorilli et al., 2025).
CPAP therapy for obstructive sleep apnea demonstrates this: restoring normal breathing reduces circulating leptin over weeks of use, supporting the position that improved sleep modifies the hormonal environment without requiring prior weight loss (Figorilli et al., 2025).
Exogenous leptin works in energy-deficient states — congenital leptin deficiency and lipodystrophy — where the body produces insufficient leptin. In common obesity and type 2 diabetes, leptin administration does not produce meaningful metabolic improvement. The problem is reception, not supply. Additional leptin does not overcome the receptor downregulation, blood-brain barrier transport breakdown, and intracellular transduction blocks that define resistance (Perakakis & Mantzoros, 2024).
The evidence supports addressing sleep quality rather than waiting for weight loss to fix sleep. Both arms of the loop reinforce each other, and addressing both simultaneously is more effective than targeting either alone.
Leptin resistance rarely acts alone. In many adults, blood sugar instability, cortisol rhythm disruptions, inflammation, and hormonal changes might be compounding alongside disrupted leptin communication — and each pathway that might be active responds to different approaches. Identifying which causes might be contributing is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Can Leptin Resistance Cause Insomnia?
Leptin does not act as a sedative. It permissively supports sleep by communicating that energy stores are sufficient and food-seeking is unnecessary. When that communication breaks down — whether from receptor downregulation, blood-brain barrier transport breakdown, or SOCS3 upregulation — the default state is sustained arousal. The brain responds as it would during starvation: stay awake, find food (Engin, 2024).
Does Lack of Sleep Make Leptin Resistance Worse?
The Spiegel et al. crossover data demonstrated the acute arm: within two days of sleeping 4 hours per night, leptin dropped 18% and ghrelin rose 28%. The chronic arm follows from the downstream effects — increased caloric intake drives weight gain, which raises circulating leptin, which accelerates receptor desensitization. Both pathways feed resistance (Spiegel et al., 2004).
How Does Sleep Deprivation Affect Leptin and Ghrelin Together?
The Taheri et al. data showed a dose-response pattern: each hour of sleep loss corresponded to proportionally lower leptin and higher ghrelin (Taheri et al., 2004). The Lin et al. meta-analysis found ghrelin elevation across 21 studies, but also found a paradoxical leptin increase under acute total sleep deprivation. Chronic short sleep suppresses leptin; acute total deprivation produces a stress-driven leptin spike. The two patterns reflect different physiological states (Lin et al., 2020).
Does CPAP for Sleep Apnea Improve Leptin Levels?
CPAP restores normal breathing during sleep, eliminating the intermittent hypoxia and sleep fragmentation that characterize obstructive sleep apnea. The leptin reduction after CPAP use supports the “address sleep first” approach — improving sleep alters the hormonal environment that drives metabolic impairment (Figorilli et al., 2025).
Related Reading
- Metabolic Sleep Disruption
- Why Do You Wake Up Hungry at 3am?
- Does Ozempic Cause Insomnia?
- Why Does Menopause Cause Insomnia?
- How Do Blood Sugar, Cortisol, and Leptin Control Whether You Sleep?
References
Engin, A. (2024). The mechanism of leptin resistance in obesity and therapeutic perspective. Advances in Experimental Medicine and Biology, 1460, 463-487. https://pubmed.ncbi.nlm.nih.gov/39287862/
Figorilli, M., Velluzzi, F., & Redolfi, S. (2025). Obesity and sleep disorders: A bidirectional relationship. Nutrition, Metabolism, and Cardiovascular Diseases, 35(6), 104014. https://pubmed.ncbi.nlm.nih.gov/40180826/
Hu, W., Zhu, H., & Gong, F. (2025). Leptin and leptin resistance in obesity: Current evidence, mechanisms and future directions. Endocrine Connections, 14(9), e250521. https://pubmed.ncbi.nlm.nih.gov/40932169/
Lin, J., Jiang, Y., Wang, G., Meng, M., Zhu, Q., Mei, H., Liu, S., & Jiang, F. (2020). Associations of short sleep duration with appetite-regulating hormones and adipokines: A systematic review and meta-analysis. Obesity Reviews, 21(11), e13051. https://pubmed.ncbi.nlm.nih.gov/32537891/
Mosini, A. C., Adami, L. N. G., da Silva Vallim, J. R., Moyses-Oliveira, M., Poyares, D., Andersen, M. L., & Tufik, S. (2024). Leptin moderates the relationship between sleep quality and memory function: A population-based study. Sleep Medicine, 117, 146-151. https://pubmed.ncbi.nlm.nih.gov/38537522/
Motivala, S. J., Tomiyama, A. J., Ziegler, M., Khandrika, S., & Irwin, M. R. (2009). Nocturnal levels of ghrelin and leptin and sleep in chronic insomnia. Psychoneuroendocrinology, 34(4), 540-545. https://pubmed.ncbi.nlm.nih.gov/19059729/
Olson, C. A., Hamilton, N. A., & Somers, V. K. (2016). Percentage of REM sleep is associated with overnight change in leptin. Journal of Sleep Research, 25(4), 419-425. https://pubmed.ncbi.nlm.nih.gov/26919408/
Perakakis, N., & Mantzoros, C. S. (2024). Evidence from clinical studies of leptin: Current and future clinical applications in humans. Metabolism: Clinical and Experimental, 161, 156053. https://pubmed.ncbi.nlm.nih.gov/39490439/
Spiegel, K., Tasali, E., Penev, P., & Van Cauter, E. (2004). Brief communication: Sleep curtailment in healthy young men is associated with decreased leptin levels, elevated ghrelin levels, and increased hunger and appetite. Annals of Internal Medicine, 141(11), 846-850. https://pubmed.ncbi.nlm.nih.gov/15583226/
Taheri, S., Lin, L., Austin, D., Young, T., & Mignot, E. (2004). Short sleep duration is associated with reduced leptin, elevated ghrelin, and increased body mass index. PLoS Medicine, 1(3), e62. https://pubmed.ncbi.nlm.nih.gov/15602591/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 10 references cited
