The evidence-based daytime minimum is 250 melanopic equivalent daylight illuminance (melanopic EDI) lux at eye level — the wavelengths your circadian clock responds to, centered around 480 nanometers. Standard indoor lighting often delivers under 150 melanopic EDI lux. If morning sunlight is unavailable due to schedule, latitude, or season, a 10,000-lux broad-spectrum light therapy box at eye level for 20 to 30 minutes within the first hour after waking provides a comparable entrainment cue.
Morning sunlight is the top circadian recommendation. How much light is enough? The answer depends on melanopic EDI, and the indoor-outdoor gap is larger than most people assume.
This article covers the melanopic threshold, the indoor-outdoor gap, what to do when morning sunlight is unavailable, and how aging changes light requirements. Light exposure is one of several factors that influence circadian timing; the full circadian disruption overview covers the broader picture.
What Is Melanopic Lux and Why Does It Matter More Than Regular Lux?
Melanopic lux measures the light wavelengths that activate intrinsically photosensitive retinal ganglion cells (ipRGCs) — the cells that send timing information to the circadian clock. Standard photopic lux measures overall brightness for vision. The two metrics diverge because ipRGCs have peak sensitivity at 480 to 483 nanometers, while photopic lux weights the entire visible spectrum. A room can be bright to your eyes (high photopic lux) and dim to your clock (low melanopic EDI).
Your retina contains cells that have nothing to do with seeing images. These are intrinsically photosensitive retinal ganglion cells (ipRGCs), and they contain a photopigment called melanopsin. Their job is to report ambient light conditions to the suprachiasmatic nucleus — the brain’s master circadian pacemaker. Melanopsin peaks at 481 to 483 nanometers, squarely in the short-wavelength (blue) region of the visible spectrum.
St Hilaire et al. (2022) mapped how this spectral sensitivity changes with exposure duration. During short exposures, cones drive the circadian response — the retina uses fast visual photoreceptors as an early detection mechanism. During longer exposures of approximately 1 to 3 hours and beyond, melanopsin takes over as the dominant photopigment. This means the spectral characteristics of morning light exposure during a sustained outdoor walk engage a different photoreceptor weighting than a brief glance out a window.
Melanopic EDI (equivalent daylight illuminance) was developed to capture this distinction. It quantifies light in terms of ipRGC activation, accounting for spectral composition — not just raw brightness. Standard photopic lux weights the entire visible spectrum with a peak near 555 nanometers (green-yellow), matching cone photoreceptor sensitivity for daytime vision.
The practical divergence between these two metrics is large. Schollhorn et al. (2023) demonstrated in a controlled study that sleep latency and melatonin suppression tracked with melanopic irradiance, not photopic lux. Participants exposed to display light with high melanopic content took longer to fall asleep and showed greater melatonin suppression than those exposed to the same photopic brightness with low melanopic content. The circadian clock was responding to the spectral signature of the light, not how bright it looked.
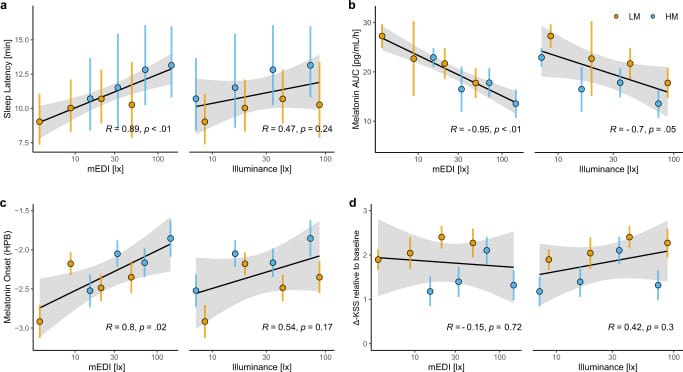
This means a warm-toned LED at 200 photopic lux may deliver under 50 melanopic EDI. A daylight-spectrum LED at the same photopic lux delivers 150 or more melanopic EDI. The room feels equally bright. The circadian input is three to four times higher from the daylight-spectrum source.
How Much Light Does Your Clock Need — and How Much Are You Getting?
An international consensus recommends a minimum of 250 melanopic EDI lux at eye level during daytime hours. Standard indoor office lighting delivers 300 to 500 photopic lux at desk level, but this translates to only 100 to 200 melanopic EDI lux at eye level with typical fluorescent or LED sources. Cloudy outdoor daylight delivers approximately 2,000 photopic lux; direct sunlight exceeds 100,000. The gap between indoor melanopic exposure and the evidence-based minimum is a factor of 2 to 3 for people who spend their mornings indoors.
Brown et al. (2022) published the first international consensus recommendations for melanopic light exposure. The thresholds: a minimum of 250 melanopic EDI lux during daytime hours, a maximum of 10 melanopic EDI lux in the evening (within 3 hours of habitual sleep), and a maximum of 1 melanopic EDI lux during sleep. These targets were derived from dose-response curves for melatonin suppression, circadian phase resetting, and subjective alertness.
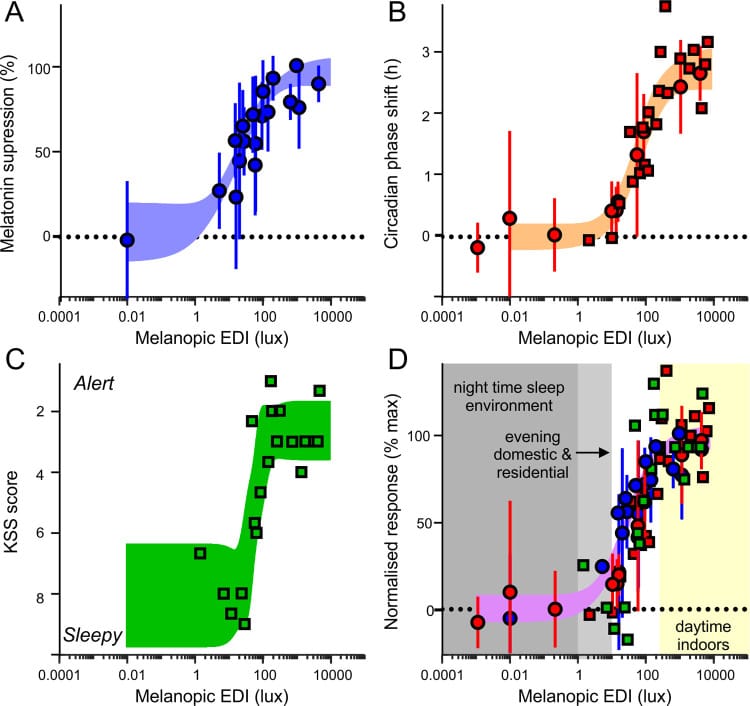
How do real-world exposures compare to these targets? Didikoglu et al. (2023) answered this by fitting 59 UK adults with wrist-worn melanopic light loggers that recorded continuous exposure over multiple days. Participants often fell below the 250 melanopic EDI minimum during daytime hours, spending much of the waking day well below threshold. The study also found a dose-response relationship between melanopic light and alertness: each 1 log-lux increase in melanopic EDI was associated with a 0.5-point reduction in subjective sleepiness (KSS). More melanopic light during the day meant less daytime drowsiness.
Windred et al. (2024) extended this to population-level health outcomes. In an analysis of more than 88,000 UK Biobank participants with wrist-worn light sensors, high daytime light exposure was associated with a 17 to 34 percent reduction in all-cause mortality. High nighttime light exposure was associated with a 21 to 34 percent increase in all-cause mortality. This is the largest dataset linking personal light exposure patterns to mortality risk.
The gap for indoor workers is not marginal. A standard office under fluorescent lighting meets visual standards at 300 to 500 photopic lux at desk level, but delivers only 100 to 200 melanopic EDI at eye level. Stepping outside on a cloudy day delivers approximately 2,000 photopic lux. Direct morning sunlight exceeds 100,000. The circadian clock is calibrated for outdoor light levels, and indoor environments fall short by an order of magnitude.
What Happens When You Cannot Get Morning Sunlight?
Seasonal light loss delays circadian timing. In Seattle (47 degrees N), students fell asleep 35 minutes later and woke 27 minutes later in winter compared to summer — and daytime light exposure was the strongest predictor, not temperature or social schedule. For people who cannot get outdoor morning light, a 10,000-lux broad-spectrum light therapy box at eye level for 20 to 30 minutes provides a comparable phase-advance cue. Dawn simulation devices — gradual light ramps before waking — can produce measurable circadian phase advance.
Dunster et al. (2023) tracked over 500 students at the University of Washington in Seattle (47 degrees N latitude) across winter and summer quarters. In winter, students fell asleep 35 minutes later and woke 27 minutes later compared to summer. The researchers measured multiple candidate predictors — temperature, photoperiod, social schedules, academic workload — and found that daytime light exposure was the strongest single predictor of the seasonal change in sleep timing. The effect was driven by reduced daytime melanopic input, not by later sunsets or social schedule changes.
This finding matters for anyone who commutes in darkness, works in windowless environments, or lives at latitudes where winter sunrise comes after the workday begins.
Light therapy boxes. A 10,000-lux broad-spectrum light therapy box positioned at approximately 40 to 60 centimeters from the face delivers melanopic input above the 250 melanopic EDI threshold. Broad-spectrum sources (covering the full visible range with enrichment in the 460 to 490 nanometer region) are preferred over narrow-band blue LEDs, which can cause discomfort and do not engage the full cone-melanopsin interaction that characterizes sustained light exposure. Timing matters: the phase-advance effect is strongest when exposure occurs within the first hour after waking. The recommended duration is 20 to 30 minutes.
Dawn simulation. Terman and Terman (2010) tested this in a randomized controlled trial. A gradual dawn ramp administered while participants were still asleep produced circadian phase advance. Light penetrates closed eyelids at sufficient intensity to activate ipRGCs, particularly as the ramp approaches its peak. Dawn simulation requires no active effort — the device runs while you sleep.
Practical hierarchy. Outdoor morning sunlight delivers the highest melanopic EDI and remains the first-choice option. A light therapy box is the next best alternative when outdoor exposure is not feasible. Dawn simulation provides a passive option with documented phase-advance effects. Bright indoor lighting (daylight-spectrum bulbs, 5000K or above, positioned near eye level) is the lowest tier — better than warm-toned ambient lighting but often insufficient to reach the 250 melanopic EDI target on its own.
Does Aging Change How Much Light You Need?
Yes. The crystalline lens yellows with age, progressively reducing transmission of short-wavelength light — including the 480-nanometer range that melanopsin depends on. By late adulthood, blue light reaching the retina is substantially reduced compared to a young eye — lens transmission declines by approximately 0.6 to 0.7 percentage points per year. In a study of 970 adults, sleep disturbance risk increased with reduced blue light transmission (p < 0.0001). Older adults need brighter or more spectrally enriched morning light to achieve the same circadian entrainment as younger adults.
The crystalline lens of the eye is not static. It accumulates yellow chromophores throughout life — a progressive optical filter that preferentially absorbs short-wavelength light. Kessel et al. (2011) quantified this filtration and its relationship to sleep in 970 adults aged 30 to 60. The study found that the risk of sleep disturbances increased when blue light transmission to the retina was low (p < 0.0001), even after correction for age and other confounding factors. The same wavelengths that the lens progressively blocks are the wavelengths the circadian clock depends on.
Eto and Higuchi (2023) reviewed the downstream consequences of this age-related filtration. Two optical changes reduce the short-wavelength light reaching the retina in older adults: progressive lens opacity and age-related pupil constriction (senile miosis). Both reduce ipRGC stimulation, meaning less melanopic input reaches the circadian clock despite the same ambient light conditions. Some evidence suggests compensatory mechanisms may partially offset this reduced input, but the net effect is that older adults receive less circadian-relevant light per unit of ambient illuminance.
Zeitzer et al. (2000) established the dose-response framework for circadian photoreception. The relationship between light intensity and circadian phase resetting follows a sigmoid curve — half-maximal circadian response occurs at approximately 80 to 160 lux. Below this point, small increases in light produce large gains in circadian response. Above it, additional light produces diminishing returns. For a young adult, typical morning outdoor light sits well above the half-maximal point. For an older adult with decades of lens yellowing, the effective retinal illuminance may be at or near the half-maximal threshold — on the steep part of the curve where reductions in light have disproportionate consequences for circadian function.
A 40-year-old receiving 30 minutes of morning sunlight may be getting adequate melanopic input with room to spare. A 65-year-old doing the same routine may be near or below the functional threshold due to cumulative lens filtration. For older adults, longer outdoor exposure, higher-output light therapy devices, or daylight-spectrum bulbs enriched in the 460 to 490 nanometer range can compensate.
Many people have more than one cause contributing to their sleep disruption. Circadian light-related disruption may compound with metabolic, hormonal, inflammatory, or autonomic factors. Identifying which causes might be involved is a useful next step.
Find out which causes might be driving your 3 AM wakeups →
Frequently Asked Questions
Does Morning Sunlight Through a Window Count?
Partially. Standard window glass transmits visible light including 480-nanometer wavelengths, but blocks UV and reduces total irradiance by 25 to 50 percent depending on glass type, tinting, and angle. A seat by a large, uncoated window may deliver adequate melanopic input. A seat away from the window in a fluorescent-lit room does not.
Glass does not selectively block 480-nanometer light the way the aging lens does — standard uncoated window glass has relatively flat transmission across the visible spectrum. The issue is total irradiance reduction. Double-glazed, low-emissivity, or tinted windows can reduce total light transmission by 50 percent or more. Distance from the window compounds the loss: illuminance drops as the inverse square of distance.
Using the Brown et al. (2022) benchmark of 250 melanopic EDI lux, a position within 1 meter of a large, uncoated window on an overcast day may deliver adequate levels. A position 3 to 4 meters from the same window, under overhead fluorescent lighting, will fall well short. The answer depends on window size, glass type, and distance — not on whether glass “blocks blue light,” which is a misconception for standard residential glass.
How Long Do You Need to Be Outside for the Circadian Benefit?
The circadian pacemaker responds in a sigmoid dose-response — half-maximal phase change occurs at approximately 80 to 160 lux under controlled nocturnal conditions. In practical terms, 20 to 30 minutes of outdoor morning light on a cloudy day (approximately 2,000 lux) is well above the half-maximal threshold. On sunny days, 10 to 15 minutes may be sufficient.
Zeitzer et al. (2000) showed that the relationship between light intensity and circadian phase resetting is not linear — it follows a sigmoid function with a steep middle range and plateau at both ends. The half-maximal response occurred at approximately 80 to 160 lux in controlled nocturnal conditions. Outdoor morning light on an overcast day delivers roughly 2,000 lux — roughly 15 to 25 times above the half-maximal point. Direct sunlight exceeds 100,000 lux.
Based on the Brown et al. (2022) duration guidance and the dose-response data, 20 to 30 minutes of outdoor exposure on a cloudy morning provides a strong circadian entrainment cue. On sunny mornings, 10 to 15 minutes may be sufficient. The important factor is consistency — a single bright morning does not compensate for a week of dim indoor mornings. Daily exposure maintains the phase relationship between your circadian clock and the light-dark cycle.
Are Blue Light Glasses and Night Mode Helping or Hurting?
Blue light glasses and phone night mode reduce melanopic stimulation — which is appropriate in the evening when you want melatonin onset to proceed undisturbed, but counterproductive in the morning or daytime when you need melanopic input to entrain the clock. The question is timing, not the glasses themselves.
St Hilaire et al. (2022) showed that the spectral sensitivity of circadian phase resetting peaks at 481 to 483 nanometers — the same wavelength range that blue light glasses are designed to attenuate. During evening hours, reducing this wavelength input is beneficial: it allows melatonin onset to proceed on schedule. The Brown et al. (2022) recommendation of less than 10 melanopic EDI lux in the evening supports this approach.
During morning and daytime hours, the goal reverses. The circadian clock needs melanopic input to maintain its phase alignment with the external light-dark cycle. Wearing blue light glasses in a daytime office environment — where melanopic EDI is already below the 250-lux minimum — further reduces an input that is already insufficient. Night mode on screens has the same directional effect: appropriate after sunset, counterproductive at 9am.
Related Reading
- Circadian Rhythm Disruption: How a Misaligned Internal Clock Fragments Sleep, Drives 3AM Wakeups, and Reduces Sleep Depth — the full overview of circadian sleep disruption, including melatonin, cortisol, body temperature, peripheral clocks, and early-morning waking
- Why Does Melatonin Work for Jet Lag but Not Chronic Insomnia? — why melatonin works as a timing cue, not a general sleep-depth fix
- Is Your Pineal Gland Calcifying? How Age-Related Melatonin Production Loss Differs From Receptor Decline — how pineal calcification, melatonin output, and receptor decline diverge with age
- How Much Blue Light From Your Phone Suppresses Melatonin? The Dose-Duration Relationship — how screen brightness, duration, and melanopic irradiance affect melatonin timing
- Do Blue Light Glasses Help You Sleep? Amber vs Standard Lenses — What Studies Show — how amber lenses, clear lenses, and evening screens differ in sleep studies
- How Long Does It Take to Reset Your Circadian Rhythm? — why the reset timeline differs for the SCN, peripheral clocks, and behavioral cues
- How Does Caffeine Delay Your Circadian Clock by 40 Minutes? — how caffeine delays circadian phase through adenosine and clock-gene timing
References
1. St Hilaire, M. A., Ámundadóttir, M. L., Rahman, S. A., Rajaratnam, S. M. W., Rüger, M., Brainard, G. C., Czeisler, C. A., Andersen, M., Gooley, J. J., & Lockley, S. W. (2022). The spectral sensitivity of human circadian phase resetting and melatonin suppression to light changes dynamically with light duration. Proceedings of the National Academy of Sciences, 119(51), e2205301119. https://pubmed.ncbi.nlm.nih.gov/36508661/
2. Schöllhorn, I., Stefani, O., Lucas, R. J., Spitschan, M., Slawik, H. C., & Cajochen, C. (2023). Melanopic irradiance defines the impact of evening display light on sleep latency, melatonin and alertness. Communications Biology, 6(1), 228. https://pubmed.ncbi.nlm.nih.gov/36854795/
3. Brown, T. M., Brainard, G. C., Cajochen, C., Czeisler, C. A., Hanifin, J. P., Lockley, S. W., Lucas, R. J., Münch, M., O’Hagan, J. B., Peirson, S. N., Price, L. L. A., Roenneberg, T., Schlangen, L. J. M., Skene, D. J., Spitschan, M., Vetter, C., Zee, P. C., & Wright, K. P., Jr. (2022). Recommendations for daytime, evening, and nighttime indoor light exposure to best support physiology, sleep, and wakefulness in healthy adults. PLOS Biology, 20(3), e3001571. https://pubmed.ncbi.nlm.nih.gov/35298459/
4. Didikoglu, A., Mohammadian, N., Johnson, S., van Tongeren, M., Wright, P., Casson, A. J., Brown, T. M., & Lucas, R. J. (2023). Associations between light exposure and sleep timing and sleepiness while awake in a sample of UK adults in everyday life. Proceedings of the National Academy of Sciences, 120(42), e2301608120. https://pubmed.ncbi.nlm.nih.gov/37812713/
5. Windred, D. P., Burns, A. C., Lane, J. M., Olivier, P., Rutter, M. K., Saxena, R., Cain, S. W., & Phillips, A. J. K. (2024). Brighter nights and darker days predict higher mortality risk: A prospective analysis of personal light exposure in >88,000 individuals. Proceedings of the National Academy of Sciences, 121(43), e2405924121. https://pubmed.ncbi.nlm.nih.gov/39405349/
6. Dunster, G. P., Hua, I., Grahe, A., Fleischer, J. G., Panda, S., Wright, K. P., Jr., Vetter, C., Doherty, J. H., & de la Iglesia, H. O. (2023). Daytime light exposure is a strong predictor of seasonal variation in sleep and circadian timing of university students. Journal of Pineal Research, 74(2), e12843. https://pubmed.ncbi.nlm.nih.gov/36404490/
7. Terman, M., & Terman, J. S. (2010). Circadian rhythm phase advance with dawn simulation treatment for winter depression. Journal of Biological Rhythms, 25(4), 297–301. https://pubmed.ncbi.nlm.nih.gov/20679499/
8. Kessel, L., Siganos, G., Jørgensen, T., & Larsen, M. (2011). Sleep disturbances are related to decreased transmission of blue light to the retina caused by lens yellowing. Sleep, 34(9), 1215–1219. https://pubmed.ncbi.nlm.nih.gov/21886359/
9. Eto, T., & Higuchi, S. (2023). Review on age-related differences in non-visual effects of light: melatonin suppression, circadian phase shift and pupillary light reflex in children to older adults. Journal of Physiological Anthropology, 42(1), 11. https://pubmed.ncbi.nlm.nih.gov/37355647/
10. Zeitzer, J. M., Dijk, D.-J., Kronauer, R. E., Brown, E. N., & Czeisler, C. A. (2000). Sensitivity of the human circadian pacemaker to nocturnal light: Melatonin phase resetting and suppression. Journal of Physiology, 526(Pt 3), 695–702. https://pubmed.ncbi.nlm.nih.gov/10922269/
Written by Kat Fu, M.S., M.S. ? Last reviewed: May 2026 ? 10 references cited
