Most coverage of GLP-1 drug side effects focuses on nausea, fatigue, and gastrointestinal symptoms. The connection between eating less and sleeping worse is absent from consumer health media, despite a well-established biochemical chain linking caloric intake to sleep neurotransmitter production.
This article traces the pathway from reduced food intake to lower tryptophan availability to reduced serotonin and melatonin synthesis to disrupted sleep. The focus is specifically on the neurotransmitter mechanism, not the blood sugar mechanism (covered separately in the companion article on Ozempic and 3am blood sugar crashes).
Appetite suppression can disrupt sleep through multiple pathways. This article covers the tryptophan-serotonin-melatonin chain. The companion article covers blood sugar instability from undereating. The parent pillar page covers the broader metabolic picture.
How Does Ozempic’s Appetite Suppression Deplete the Brain’s Sleep Chemistry
The biochemical chain is direct: dietary tryptophan is converted to 5-hydroxytryptophan (5-HTP), then to serotonin, then to melatonin. When total food intake drops, less tryptophan enters the bloodstream, and the entire downstream chain produces less sleep-promoting neurotransmitter.
Strasser et al. (2015) measured this depletion in a randomized controlled trial of 38 overweight adults. Participants eating approximately 600 kcal/day experienced a 21% reduction in plasma tryptophan; those eating approximately 1,200 kcal/day experienced a 15% reduction. The depletion was dose-dependent with deficit severity. These caloric intake levels map directly to what many GLP-1 users report eating when appetite suppression is strongest.
Riemann et al. (2002) confirmed that tryptophan depletion alone is sufficient to worsen sleep. After consuming a tryptophan-free amino acid drink, 15 patients with primary insomnia showed increased light sleep, decreased stage 2 sleep, and elevated REM density — objective markers of impaired sleep continuity. The experimental removal of tryptophan produced the sleep disruption, consistent with a causal role.
A narrative review by Binks et al. (2020) synthesized evidence that carbohydrate intake drives insulin release, and insulin clears competing large neutral amino acids (leucine, isoleucine, valine) from the bloodstream — allowing tryptophan to cross the blood-brain barrier more efficiently. When GLP-1 users eat less overall, and particularly less carbohydrate, both the total tryptophan supply and the efficiency of tryptophan delivery to the brain decline simultaneously.
How Much Tryptophan Do You Lose on Ozempic
Morales-Suarez-Varela et al. (2024) analyzed tryptophan intake and sleep quality in 11,485 Spanish university students. Tryptophan consumption below 526 mg/day was associated with an odds ratio of 2.56 for insomnia in males (95% CI: 1.36-4.82) and 1.47 in females (95% CI: 1.10-2.05). Because this study used a young, university-aged population, the threshold may differ in older adults or clinical populations — but the dose-response relationship between tryptophan intake and insomnia risk is consistent with the broader literature.
In dietary terms: 100 grams of chicken breast contains approximately 300 mg of tryptophan. A person eating two full meals with adequate protein can easily exceed 526 mg/day. A person on semaglutide who has dropped to one small meal and a snack may fall well below that threshold. The foods people tend to eat less of when appetite is suppressed — turkey, chicken, eggs, dairy, oats — are the foods highest in tryptophan.
Kautz et al. (2024), in a study of 326 pregnant women, reported that dietary tryptophan intake was associated with reduced sleep-onset latency in unadjusted analyses, though this association did not survive full adjustment for demographic and lifestyle confounders.
Sutanto et al. (2024) confirmed the pathway is responsive to intervention: in a single-blinded randomized controlled trial, 30 older adults were assigned to receive 100 mg/day of 5-HTP or no supplement for 12 weeks. Those receiving 5-HTP showed improved Pittsburgh Sleep Quality Index scores and increased serum serotonin compared to the control group, with benefits most prominent in poor sleepers. Restoring the serotonin precursor improved the measurable sleep outcome.
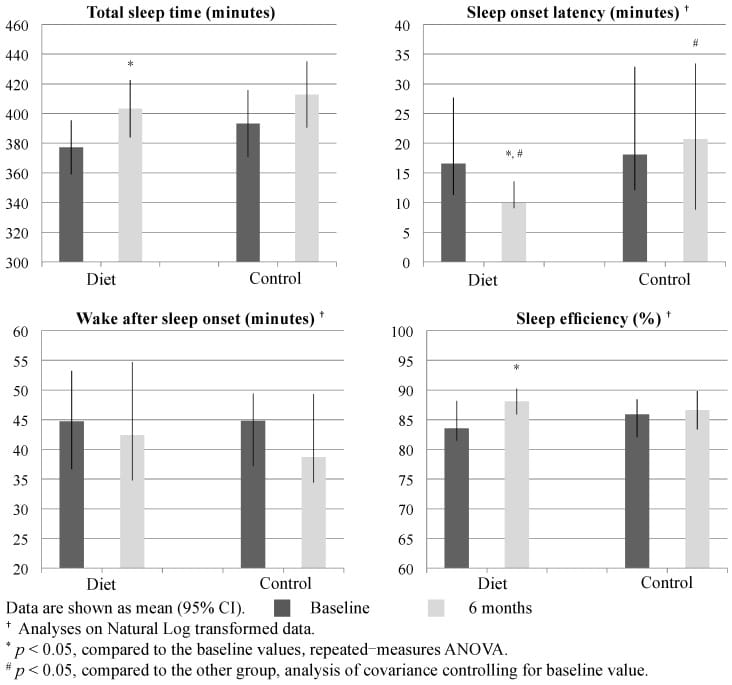
Does Ozempic Directly Affect Serotonin and Sleep Beyond Appetite Suppression
Kolling et al. (2024), using a preclinical mouse model, demonstrated that GLP-1 receptors are co-expressed on serotonin-producing neurons in the dorsal raphe nucleus and other serotonergic regions. Using spatial transcriptomic mapping, the researchers quantified GLP-1 receptor expression on tryptophan hydroxylase 2-positive neurons — the cells that synthesize serotonin in the brainstem. When GLP-1 receptor expression on these neurons was reduced (in the context of a chronic social isolation stress model), serotonin signaling was disrupted — though the contribution of GLP-1 receptor loss versus chronic stress itself cannot be fully separated in this design.
Two separate pathways connect GLP-1 drugs to serotonin-dependent sleep disruption. The dietary pathway: appetite suppression reduces tryptophan intake, reducing the raw material for serotonin synthesis. The pharmacological pathway: semaglutide directly activates GLP-1 receptors on serotonin neurons, potentially altering their firing patterns regardless of what the user eats. This second pathway has been demonstrated in mice but not yet confirmed in humans.
Attenburrow et al. (2003) added a further layer: in a double-blind trial, 50 women on a 1,000 kcal/day diet for three weeks were randomized to receive tryptophan supplementation or placebo. The placebo group showed enhanced prolactin response to serotonin drug challenge — a functional marker suggesting increased 5-HT sensitivity during caloric restriction. The tryptophan-supplemented group did not show this enhanced response, indicating that restoring tryptophan during dieting prevented the serotonergic compensation. GLP-1 users who do not maintain adequate tryptophan intake may experience similar serotonergic shifts alongside the direct pharmacological modulation, which may amplify the disruption beyond what either mechanism produces alone.
The dietary pathway is addressable by changing what you eat. The pharmacological pathway is an inherent effect of the drug.
Can Changing What You Eat on Ozempic Help You Sleep Better
Tan et al. (2016) demonstrated this in a six-month randomized controlled trial: a 300-500 kcal/day deficit with nutrient-adjusted macronutrient composition improved sleep onset latency, total sleep time, sleep efficiency, and nocturnal awakenings in overweight men with chronic insomnia. The distinction is between severe, nutrient-poor restriction (which depletes tryptophan and worsens sleep) and moderate, nutrient-aware restriction (which can preserve sleep).
For GLP-1 users experiencing appetite suppression, the principle applies directly. The goal is not to fight the appetite suppression but to adjust the nutritional quality of what is consumed:
- Tryptophan-rich foods at the evening meal: turkey, eggs, dairy, oats, and nuts are among the highest dietary sources of tryptophan. Prioritizing these foods when appetite is limited concentrates tryptophan intake in fewer calories.
- Including carbohydrate with the evening meal: carbohydrate intake triggers insulin release, which clears competing amino acids from the bloodstream and allows tryptophan to cross the blood-brain barrier more efficiently. Evening timing matters most because serotonin-to-melatonin conversion occurs at night.
- Evening intake matters disproportionately: melatonin synthesis depends on serotonin availability during the hours before and during sleep. Skipping dinner or eating only a small, protein-poor evening snack removes the substrate the brain needs for overnight melatonin production.
A 2025 meta-analysis by Mei et al., synthesizing 28 randomized controlled trials of dietary supplements (including amino acids, vitamins, minerals, and natural extracts), confirmed that supplementation improved sleep quality scores, reduced sleep latency, and increased sleep efficiency. Within the analysis, tryptophan specifically was associated with reduced wake after sleep onset. These are mechanisms worth discussing with a healthcare provider or dietitian, not supplementation recommendations — but they confirm the pathway responds measurably to intervention.
Appetite-driven changes to sleep neurotransmitter production are one of several metabolic pathways that can disrupt sleep. The same caloric deficit that depletes tryptophan can also destabilize blood sugar, alter cortisol rhythms, and interact with hormonal changes — each affecting sleep through a different mechanism. Identifying which causes might be active in your case is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Can Low Blood Sugar From Appetite Suppression Wake You at 3am
The blood sugar mechanism is separate from the tryptophan-serotonin mechanism covered in this article. Both can result from the same root cause — reduced food intake on GLP-1 drugs — but they disrupt sleep through different biochemical chains. Strasser et al. (2015) documented the caloric restriction levels at which tryptophan depletion occurs; the same degree of undereating can simultaneously deplete glycogen stores and trigger nocturnal blood sugar instability. The companion article on Ozempic and 3am blood sugar crashes covers the glucose-cortisol pathway in detail.
Does Ozempic Reduce the Urge to Eat at Night Specifically
Semaglutide reduces hunger broadly, not selectively at night — but serotonin-to-melatonin conversion occurs during the hours before and during sleep, so reduced evening intake disproportionately affects overnight neurotransmitter production even if total daily intake is adequate.
Does Protein Deficiency on Ozempic Affect Sleep
When appetite is suppressed, many people gravitate toward small, easy-to-eat foods — crackers, toast, fruit, broth — that are low in protein and therefore low in tryptophan. The tryptophan threshold of approximately 526 mg/day identified by Morales-Suarez-Varela et al. (2024) is easily breached by protein-poor eating patterns on GLP-1 drugs.
Does Ozempic Affect Melatonin Production Directly
Sutanto et al. (2024) showed that supplementing 5-HTP raised serum serotonin in older adults, with benefits most prominent in poor sleepers. When the precursor is restored, downstream neurotransmitter output increases.
Is Ozempic-Related Insomnia Worse for People Already Eating Low-Carbohydrate or Ketogenic Diets
Low-carbohydrate diets reduce insulin secretion, which means fewer competing large neutral amino acids are cleared from the blood, and tryptophan competes less successfully for blood-brain barrier transport (Binks et al., 2020). Adding GLP-1-driven appetite suppression on top of an already low-carbohydrate baseline reduces both the total tryptophan supply and the efficiency of tryptophan delivery to the brain. This double reduction has not been studied directly in GLP-1 users, but both mechanisms are individually well-established.
Related Reading
- Metabolic Sleep Disruption — The full metabolic sleep framework covering glucose regulation, insulin resistance, mitochondrial decline, NAD+ loss, and fat metabolism
- Does Ozempic Cause Insomnia What GLP-1 Drugs Do to Your Sleep — The broad overview of GLP-1 sleep side effects and mechanisms
- Why Does Ozempic Make You Tired But Keep You Awake — How GLP-1 drugs activate orexin/wakefulness circuitry while appetite suppression can create fatigue
- Why Does Ozempic Wake You Up at 3am — How lower overnight fuel availability and counter-regulatory hormones can contribute to 3am waking
- Does Ozempic Give You Vivid Dreams — How GLP-1 drugs may affect dream intensity through arousal and neuropsychiatric pathways
- Does Your Ozempic Injection Day Disrupt Your Sleep The 24-48 Hour Pattern — Why sleep disruption can follow a weekly pattern tied to injection timing and peak drug exposure
- Does Your Semaglutide Dose Increase Restart Your Insomnia Why Each Titration Can Disrupt Sleep Again — Why each titration step can restart sleep disruption before adaptation catches up
- Can You Take Melatonin, Magnesium, or Sleep Aids While on Ozempic — How melatonin, magnesium, DORAs, and other sleep aids intersect with GLP-1 physiology
- Does Ozempic Give You Anxiety at Night Why GLP-1 Drugs May Contribute to Nighttime Panic — How orexin activation, HPA-axis signaling, and glucose instability may contribute to nighttime anxiety
- Do GLP-1 Drugs Disrupt Your Body Clock Ozempic, Circadian Rhythms, and Melatonin — How GLP-1 secretion rhythms, food timing, peripheral clocks, and melatonin intersect
- Ozempic Night Sweats: How GLP-1 Drugs May Affect Temperature at Night — How GLP-1 drugs may affect nighttime sweating through autonomic tone and thermoregulation
- Ozempic Nausea and Sleep: How Stomach Side Effects Keep You Awake at Night — How delayed gastric emptying, reflux, and nausea can fragment sleep
- Will Your Sleep Go Back to Normal When You Stop Ozempic — What is known and not known about sleep after semaglutide discontinuation
- Does Ozempic Muscle Loss Affect Your Sleep Sarcopenia, Temperature, and Deep Sleep — How lean-mass loss, thermoregulation, and deep sleep can interact during GLP-1 weight loss
- Could Ozempic Contribute to Restless Legs at Night Iron, B12, and the Dopamine Connection — How iron, B12, dopamine, and rapid weight loss may intersect with restless legs at night
- Does Compounded Semaglutide Affect Sleep Differently Than Brand-Name Ozempic — How dose variability, formulation differences, and adverse-event tracking can affect sleep side effects
- Ozempic and Sleep After 40: Why GLP-1 Side Effects Can Feel Different as You Age — Why GLP-1 sleep effects can land differently after 40 because of muscle, hormones, and glucose regulation
References
- Attenburrow, M. J., Williams, C., Odontiadis, J., Powell, J., Van de Ouderaa, F., Williams, M., & Cowen, P. J. (2003). The effect of a nutritional source of tryptophan on dieting-induced changes in brain 5-HT function. Psychological Medicine, 33(8), 1381-1386. https://pubmed.ncbi.nlm.nih.gov/14672246/
- Binks, H., E Vincent, G., Gupta, C., Irwin, C., & Khalesi, S. (2020). Effects of Diet on Sleep: A Narrative Review. Nutrients, 12(4), 936. https://pubmed.ncbi.nlm.nih.gov/32230944/
- Kautz, A., Meng, Y., Yeh, K. L., Peck, R., Brunner, J., Best, M., Fernandez, I. D., Miller, R. K., Barrett, E. S., Groth, S. W., & O’Connor, T. G. (2024). Dietary Intake of Nutrients Involved in Serotonin and Melatonin Synthesis and Prenatal Maternal Sleep Quality and Affective Symptoms. Journal of Nutrition and Metabolism, 2024, 6611169. https://pubmed.ncbi.nlm.nih.gov/39015539/
- Kolling, L. J., Khan, K., Wang, R., Pierson, S. R., Hartman, B. D., Balasubramanian, N., Guo, D. F., Rahmouni, K., & Marcinkiewcz, C. A. (2024). Interaction of serotonin/GLP-1 circuitry in a dual preclinical model for psychiatric disorders and metabolic dysfunction. Psychiatry Research, 337, 115951. https://pubmed.ncbi.nlm.nih.gov/38735240/
- Mei, M., Zhou, Q., Gu, W., Li, F., Yang, R., Lei, H., & Liu, C. (2025). Dietary Supplement Interventions and Sleep Quality Improvement: A Systematic Review and Meta-Analysis. Nutrients, 17(24), 3952. https://pubmed.ncbi.nlm.nih.gov/41470897/
- Morales-Suarez-Varela, M., Amezcua-Prieto, C., Peraita-Costa, I., Mateos-Campos, R., Ayan, C., Ortiz-Moncada, R., Fernandez-Villa, T., & Grupo de Investigacion UniHcos (2024). Sleep Patterns and Tryptophan Consumption among Students at Spanish Universities: The Unihcos Project. Nutrients, 16(14), 2376. https://pubmed.ncbi.nlm.nih.gov/39064819/
- Riemann, D., Feige, B., Hornyak, M., Koch, S., Hohagen, F., & Voderholzer, U. (2002). The tryptophan depletion test: impact on sleep in primary insomnia – a pilot study. Psychiatry Research, 109(2), 129-135. https://pubmed.ncbi.nlm.nih.gov/11927137/
- Strasser, B., Berger, K., & Fuchs, D. (2015). Effects of a caloric restriction weight loss diet on tryptophan metabolism and inflammatory biomarkers in overweight adults. European Journal of Nutrition, 54(1), 101-107. https://pubmed.ncbi.nlm.nih.gov/24687684/
- Sutanto, C. N., Xia, X., Heng, C. W., Tan, Y. S., Lee, D. P. S., Fam, J., & Kim, J. E. (2024). The impact of 5-hydroxytryptophan supplementation on sleep quality and gut microbiota composition in older adults: A randomized controlled trial. Clinical Nutrition, 43(3), 593-602. https://pubmed.ncbi.nlm.nih.gov/38309227/
- Tan, X., Alen, M., Wang, K., Tenhunen, J., Wiklund, P., Partinen, M., & Cheng, S. (2016). Effect of Six-Month Diet Intervention on Sleep among Overweight and Obese Men with Chronic Insomnia Symptoms: A Randomized Controlled Trial. Nutrients, 8(11), 751. https://pubmed.ncbi.nlm.nih.gov/27886073/
Written by Kat Fu, M.S., M.S. – Last reviewed: May 2026 – 10 references cited
