Waking at 3am with hunger or food cravings reflects measurable hormonal changes. Leptin (which suppresses hunger) and ghrelin (which drives it) follow circadian rhythms that normally keep appetite suppressed during sleep. When sleep is shortened or fragmented, leptin falls, ghrelin rises, and cortisol amplifies both effects. The 2-4am window is when all three converge.
This article covers how leptin, ghrelin, cortisol, and blood sugar interact during the second half of the night — and what changes when that interaction breaks down. Nocturnal hunger is one of several mechanisms by which metabolic disruption fragments sleep; the full picture is in Metabolic Sleep Disruption. For blood sugar drops specifically, see Can a Blood Sugar Drop Wake You Up at 3am?.
How Do Leptin and Ghrelin Change During Sleep?
Leptin, produced by adipose (fat) cells, acts as a satiety cue — it communicates to the brain that energy stores are adequate and reduces the drive to eat. Ghrelin, produced primarily in the stomach, does the opposite: it drives hunger and food-seeking behavior. During adequate sleep, these two hormones maintain a balance that keeps appetite suppressed from bedtime through morning.
Motivala et al. (2009) measured ghrelin and leptin at three timepoints — 11 PM, 2 AM, and 6 AM — via polysomnography in 14 men with primary insomnia versus 24 age- and weight-matched healthy controls. This is the only published study to evaluate nocturnal appetite hormones at the 2 AM timepoint specifically in participants with primary insomnia during monitored sleep.
Leptin showed time-dependent variation in both groups (p < 0.001), rising across the night as expected. Ghrelin levels across the night were lower in those with insomnia compared to controls (p < 0.0001), with differences reaching statistical significance at 11 PM (p < 0.01) and 6 AM (p < 0.01). The 2 AM comparison showed a trend in the same direction (p = 0.08).
The finding that chronic insomnia produced lower rather than higher nocturnal ghrelin is notable. It suggests that chronic sleep disruption may produce adaptive downregulation of the hunger-drive axis over time — a distinct hormonal pattern from what happens during acute sleep restriction. The acute response (ghrelin rises, hunger spikes) and the chronic adaptation (ghrelin suppresses, metabolic disruption continues) represent two different stages of the same neuroendocrine process.
What Happens to Hunger Hormones When You Lose Sleep?
Spiegel et al. (2004) ran a randomized crossover trial in 12 healthy young men (mean age 22, BMI 23.6): two nights of approximately 4 hours of sleep versus approximately 10 hours produced 18% lower leptin (p = 0.04), 28% higher ghrelin (p < 0.04), 24% higher subjective hunger (p < 0.01), and 33-45% higher appetite for calorie-dense carbohydrate foods (p = 0.02). Caloric intake and physical activity were controlled throughout the study, establishing a direct neuroendocrine mechanism independent of food availability.
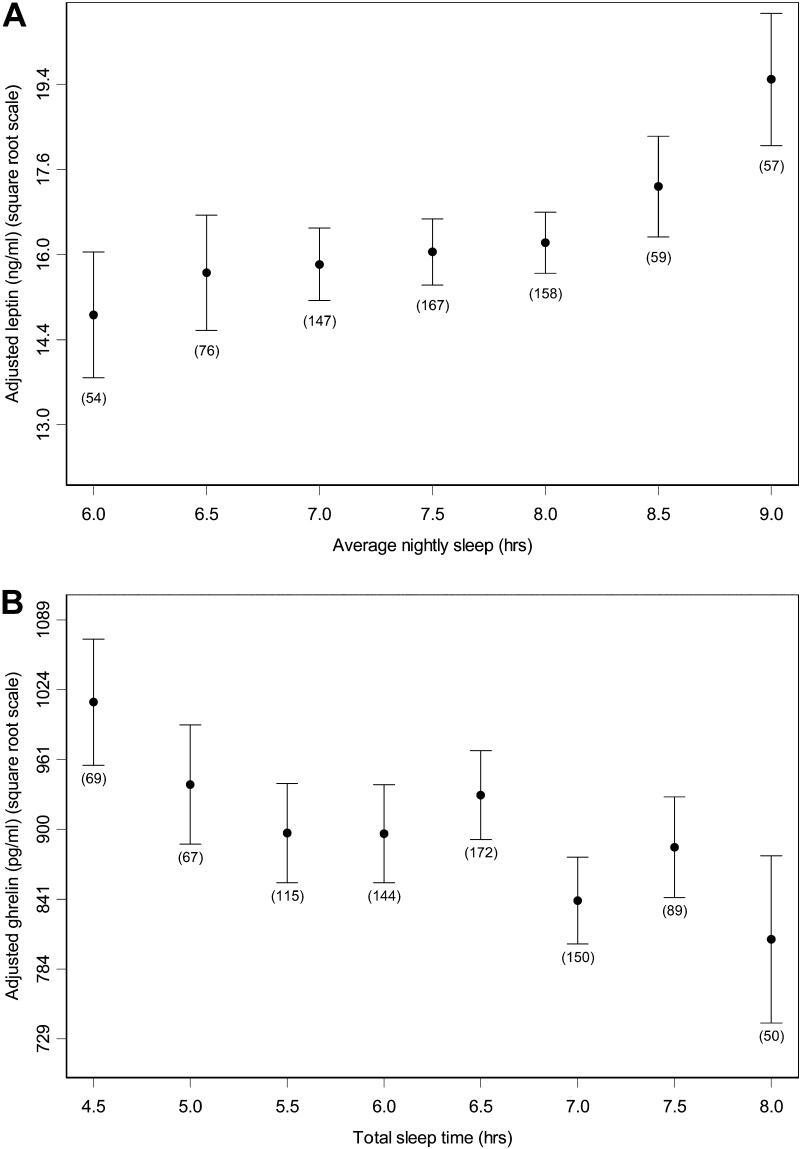
Taheri et al. (2004) showed similar effects at population scale. In 1,024 participants from the Wisconsin Sleep Cohort, 5 hours versus 8 hours of habitual sleep predicted 15.5% lower leptin (p = 0.01) and 14.9% higher ghrelin (p = 0.008), independent of BMI. The hormonal differences remained after controlling for body weight, establishing that sleep duration predicts leptin and ghrelin levels rather than reflecting obesity-related endocrine changes.
The concurrent suppression of satiety-promoting leptin and amplification of hunger-driving ghrelin creates a bidirectional hormonal environment where hunger occurs during sleep instead of staying suppressed. Two hormones moving in opposite directions at the same time — less “you’re full” and more “you’re hungry” — produces a measurable appetite increase after two nights.
Kulkarni et al. (2024), writing in Nature Reviews Endocrinology, add a circadian dimension: ghrelin acts bidirectionally on circadian clocks, meaning nocturnal hunger can become entrained as a conditioned physiological response after repeated 3am eating episodes. Once the ghrelin peak moves to 3am and the response is eating, the circadian clock reinforces that timing. LEAP2, a liver- and gut-derived ghrelin receptor antagonist, may provide a counterbalancing mechanism whose disruption amplifies ghrelin sensitivity under metabolic stress.
How Do Cortisol and Blood Sugar Amplify the 3am Hunger Pattern?
Leproult et al. (1997) measured cortisol across a 32-hour window under three conditions: normal sleep, partial sleep deprivation, and total sleep deprivation. Partial sleep deprivation produced 37% higher evening cortisol (p = 0.03). Total deprivation produced 45% higher (p = 0.003). The onset of the cortisol quiescent period — when cortisol should be at its lowest to support deep sleep — was delayed by at least one hour after sleep loss.
Elevated nocturnal cortisol opposes the normal rise in leptin during sleep. Cortisol promotes gluconeogenesis (glucose production by the liver), contributing to both suppressed satiety and blood sugar instability during the second half of the night. The result is a compounding effect: less leptin, more ghrelin, and elevated cortisol converging at the same time.
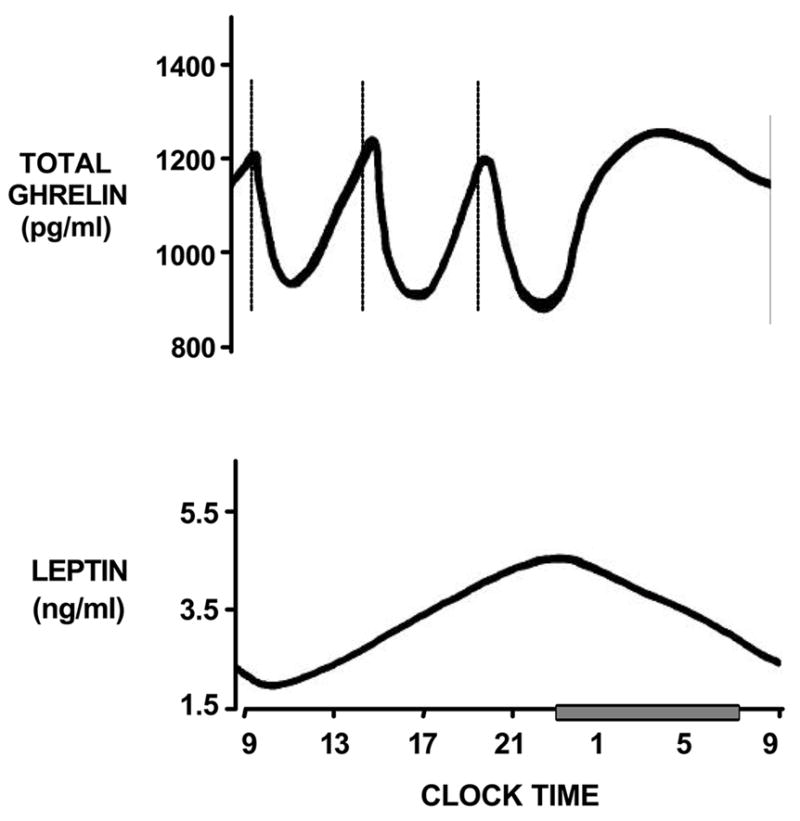
Velazquez Sanchez and Dalley (2025) provide a reframing of the cortisol-waking relationship. Their in-home microdialysis study of 201 volunteers — a large-scale home-setting cortisol study — found that the rate of cortisol increase did not differ between the hour after waking and the preceding hour of sleep. This suggests both the cortisol rise and the awakening may be driven by an upstream circadian cue rather than one causing the other. The practical focus, then, is circadian timing itself — sleep timing, light exposure, feeding windows — rather than cortisol suppression directly.
Souza et al. (2024) showed that 4 nights of restricted sleep (sleeping only from 1-5am) decreased the insulin sensitivity index (p = 0.02) and increased insulin area under the curve (p < 0.01) in healthy men. Accumulated sleep restriction creates insulin resistance, which creates glycemic instability, which drives nocturnal blood sugar fluctuations that activate the cortisol and adrenaline release triggering 3am arousal. The cycle is self-reinforcing: sleep restriction drives insulin resistance, insulin resistance drives glycemic instability, and glycemic instability drives further sleep disruption.
One important caveat: Ng et al. (2025) tracked 119 healthy university students with continuous glucose monitors and Oura Rings and found that longer sleep was associated with lower same-night glucose variability (p < 0.001), but night-to-night sleep variations did not affect glucose profiles or insulin markers in this young, healthy cohort. The blood sugar vulnerability from sleep restriction is more pronounced in older adults, those with insulin resistance, pre-diabetes, or metabolic impairment — not uniform across all sleep-disrupted individuals.
The 3am hunger pattern involves multiple hormonal axes converging at the same time of night. For some people, the primary driver is the leptin-ghrelin reversal from insufficient sleep. For others, blood sugar instability or cortisol timing plays the larger role. And for many, all three interact.
If you wake at 3am hungry on a regular basis, the pattern might be driven by hormonal changes from sleep restriction, circadian ghrelin entrainment from repeated nighttime eating, blood sugar instability from developing insulin resistance, or a combination of these.
Find out which causes might be driving your 3am wakeups →
Should You Eat If You Wake Up Hungry at Night?
Two distinct scenarios require different responses.
Scenario 1: Blood sugar drop. When overnight glucose drops low enough to trigger counterregulatory hormones (epinephrine and cortisol), the body enters a stress-response state that produces arousal. Eating a small amount of food — particularly protein and fat rather than pure carbohydrate — can stabilize glucose and reduce the cortisol and adrenaline release that keeps you awake. A tablespoon of nut butter or a few bites of cheese addresses the glucose drop without spiking insulin.
Scenario 2: Conditioned circadian entrainment. If the 3am hunger reflects ghrelin secretion that has become entrained to habitual eating times — the mechanism Kulkarni et al. (2024) describe, where ghrelin and circadian clocks interact bidirectionally — then eating reinforces the cycle. The circadian entrainment strengthens and the 3am hunger becomes more persistent with each episode.
Birketvedt et al. (1999) documented the neuroendocrine profile closest to the 3am hunger pattern in a JAMA study: night eaters averaged 3.6 nocturnal awakenings per night versus 0.3 in controls (p < 0.001), with 52% of awakenings involving food intake averaging 1,134 kJ per episode. Night eaters showed attenuated nocturnal leptin elevation (p < 0.001) and elevated circadian cortisol (p = 0.001) — the combination of suppressed leptin and elevated cortisol defines the hormonal environment for waking at 3am with hunger.
The Jiao et al. (2025) review underscores that sleep disorders disrupt multiple hormonal axes — cortisol, leptin, ghrelin, and insulin regulation — through overlapping mechanisms. Maintaining consistent sleep schedules and adequate sleep duration supports hormonal regulation across all of these pathways.
Sleep extension — getting more total sleep — is the primary approach for nocturnal hunger driven by hormonal disruption. When total sleep duration reaches 7-8 hours consistently, leptin and ghrelin normalize, cortisol timing recalibrates, and the appetite-disrupting cycle reverses.
Frequently Asked Questions
Does Sleep Deprivation Make You Crave Sugar and Carbs?
Elevated evening ghrelin during sleep restriction predicts greater consumption of sweet foods. The 3am sugar craving is a downstream effect of ghrelin elevation. Ghrelin does not increase hunger uniformly — it increases hunger for specific food categories: sweets and other calorie-dense carbohydrate foods scored 33-45% higher in the Spiegel et al. (2004) sleep restriction condition compared to the sleep extension condition.
Kulkarni et al. (2024) describe how ghrelin activates reward-related neural circuits that make high-calorie foods disproportionately appealing. When this ghrelin elevation coincides with the 3am window, the craving for carbohydrates is a hormonally driven preference. Addressing it requires addressing the sleep restriction that elevated ghrelin in the first place. See What Happens to Hunger Hormones When You Lose Sleep? above for the full hormonal mechanism.
Can Improving Sleep Reduce Nighttime Hunger?
Sleep extension is the primary approach. When habitual sleep duration reaches 7-8 hours, the hormonal environment that drives nocturnal hunger — low leptin, high ghrelin, elevated cortisol — returns toward baseline values.
For people whose 3am hunger reflects conditioned circadian entrainment (repeated nighttime eating has trained ghrelin to peak at 3am), consistent meal timing during the day helps recalibrate the circadian ghrelin rhythm. The distinction matters: if the problem is acute glucose-driven hunger, a small protein-and-fat snack may help in the short term. If the problem is a conditioned rhythm, sleep consistency resolves the pattern over weeks as the ghrelin peak returns to daytime hours.
Can Waking Up Hungry at Night Be a Sign of Insulin Resistance?
Sleep restriction drives insulin resistance, and insulin resistance worsens nocturnal blood sugar fluctuations that trigger hunger-driven waking. The relationship runs in both directions.
In the 2025 CGM study (Ng et al.), young healthy adults showed resilient glucose regulation despite sleep variability. But this resilience declines with age, metabolic impairment, and developing insulin resistance. People with pre-diabetes or metabolic impairment are more vulnerable to nocturnal blood sugar fluctuations that trigger the counterregulatory hormone response (epinephrine and cortisol release) leading to 3am arousal.
If nocturnal hunger is a recurring pattern alongside daytime fatigue, increased thirst, or difficulty maintaining weight, fasting glucose and HOMA-IR testing can identify whether insulin resistance is contributing. For the full bidirectional relationship, see Does Insulin Resistance Affect Sleep Quality?.
Related Reading
- Metabolic Sleep Disruption
- Does Leptin Resistance Cause Insomnia?
- Does Ozempic Cause Insomnia?
- Why Does Menopause Cause Insomnia?
- How Do Blood Sugar, Cortisol, and Leptin Control Whether You Sleep?
References
- Spiegel, K., Tasali, E., Penev, P., & Van Cauter, E. (2004). Brief communication: Sleep curtailment in healthy young men is associated with decreased leptin levels, elevated ghrelin levels, and increased hunger and appetite. Annals of Internal Medicine, 141(11), 846-850. https://pubmed.ncbi.nlm.nih.gov/15583226/
- Taheri, S., Lin, L., Austin, D., Young, T., & Mignot, E. (2004). Short sleep duration is associated with reduced leptin, elevated ghrelin, and increased body mass index. PLoS Medicine, 1(3), e62. https://pubmed.ncbi.nlm.nih.gov/15602591/
- Birketvedt, G. S., Florholmen, J., Sundsfjord, J., Osterud, B., Dinges, D., Bilker, W., & Stunkard, A. (1999). Behavioral and neuroendocrine characteristics of the night-eating syndrome. JAMA, 282(7), 657-663. https://pubmed.ncbi.nlm.nih.gov/10517719/
- Motivala, S. J., Tomiyama, A. J., Ziegler, M., Khandrika, S., & Irwin, M. R. (2009). Nocturnal levels of ghrelin and leptin and sleep in chronic insomnia. Psychoneuroendocrinology, 34(4), 540-545. https://pubmed.ncbi.nlm.nih.gov/19059729/
- Leproult, R., Copinschi, G., Buxton, O., & Van Cauter, E. (1997). Sleep loss results in an elevation of cortisol levels the next evening. Sleep, 20(10), 865-870. https://pubmed.ncbi.nlm.nih.gov/9415946/
- Souza, J. F. T., Monico-Neto, M., Tufik, S., & Antunes, H. K. M. (2024). Sleep debt and insulin resistance: What’s worse, sleep deprivation or sleep restriction? Sleep Science, 17(3), e272-e280. https://pubmed.ncbi.nlm.nih.gov/39268336/
- Kulkarni, S. S., Singh, O., & Zigman, J. M. (2024). The intersection between ghrelin, metabolism and circadian rhythms. Nature Reviews Endocrinology, 20(4), 228-238. https://pubmed.ncbi.nlm.nih.gov/38123819/
- Ng, A. S. C., Tai, E. S., & Chee, M. W. L. (2025). Effects of night-to-night variations in objectively measured sleep on blood glucose in healthy university students. Sleep, 48(2). https://pubmed.ncbi.nlm.nih.gov/39325824/
- Velazquez Sanchez, C., & Dalley, J. W. (2025). The cortisol awakening response: Fact or fiction? Brain and Neuroscience Advances, 9, 23982128251327712. https://pubmed.ncbi.nlm.nih.gov/40297522/
- Jiao, Y., Butoyi, C., Zhang, Q., Intchasso Adotey, S. A. A., Chen, M., Shen, W., Wang, D., Yuan, G., & Jia, J. (2025). Sleep disorders impact hormonal regulation: Unravelling the relationship among sleep disorders, hormones and metabolic diseases. Diabetology & Metabolic Syndrome, 17(1), 305. https://pubmed.ncbi.nlm.nih.gov/40750881/
- Knutson, K. L. (2007). Impact of sleep and sleep loss on glucose homeostasis and appetite regulation. Sleep Medicine Clinics, 2(2), 187-197. https://pubmed.ncbi.nlm.nih.gov/18516218/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 11 references cited
