Vivid dreams and nightmares are among the most frequently discussed but least studied side effects of GLP-1 receptor agonist medications. User descriptions range from “insanely vivid and terrifying” to “woke up crying, didn’t want to go back to sleep.” Despite widespread reports across patient forums, no authoritative sleep-science source has addressed the phenomenon in detail.
This article examines what is known about how GLP-1 drugs interact with the brain’s dream-generating systems, and what remains unknown. The scope is limited to dream-related effects — insomnia and wakefulness on GLP-1 drugs are covered separately. Dream disruption is one of several ways GLP-1 medications affect sleep. Other articles in this series cover the orexin-arousal mechanism, blood sugar crashes during sleep, and appetite-serotonin disruption of sleep chemistry.
How Do Glucagon-Like Peptide-1 Drugs Interact With the Brain’s Dream System
Two distinct neurotransmitter pathways connect GLP-1 receptor agonists to the biology of dreaming. Each operates through a different brain structure and affects a different aspect of dream experience.
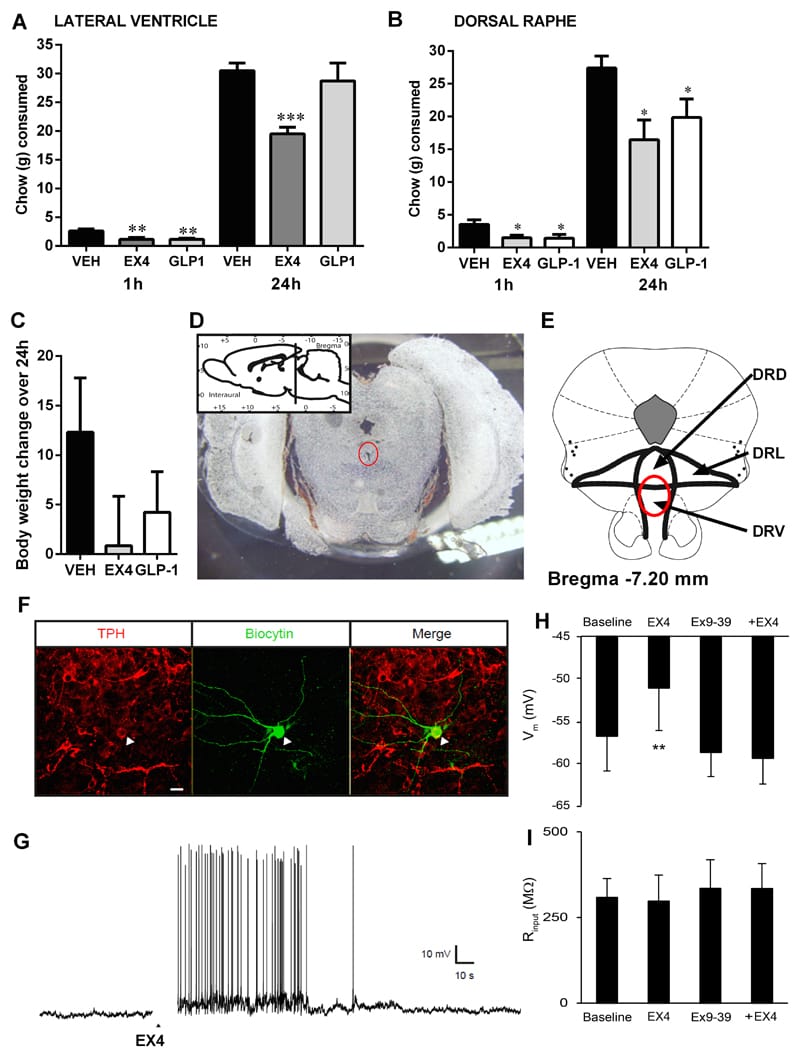
Serotonin and the dorsal raphe nucleus. The dorsal raphe nucleus is the brain’s primary source of serotonin to the forebrain. Serotonin neurons in the dorsal raphe fire actively during wakefulness and go nearly silent at REM sleep onset — their activity tonically suppresses REM. When these neurons stop firing, REM sleep begins. Anderberg et al. (2017) demonstrated that GLP-1 receptor activation in the dorsal raphe increases the electrical activity of serotonin neurons in this region. If GLP-1 receptor agonists alter the firing patterns of dorsal raphe serotonin neurons, the timing and depth of REM sleep entry could shift — producing longer, more intense, or more frequent REM episodes.
Kolling et al. (2024) provided supporting evidence for this serotonin/GLP-1 interaction in a mouse model, showing that socially isolated mice had reduced GLP-1 receptor expression on serotonin neurons across raphe nuclei and altered serotonergic function. Their study also measured sleep (finding reduced sleep in isolated mice during the light phase) and dorsal raphe neuronal activity (finding increased action potential frequency in isolated mice), adding behavioral and electrophysiological evidence to the pathway Anderberg identified.
Dopamine and the nucleus accumbens. The second pathway involves mesolimbic dopamine. Aranas et al. (2025) showed in mice and rats that semaglutide attenuates cocaine-evoked dopamine elevation in the nucleus accumbens — the central node of the brain’s reward circuit. The nucleus accumbens is involved in reward and emotional processing during both wakefulness and sleep. Whether semaglutide affects baseline dopamine levels in this region — outside a drug-challenge context — has not been tested. If it does, altered dopamine tone during REM sleep could affect dream recall and perceived vividness through changed reward processing during sleep, though this remains speculative.
These are two separate pathways — serotonin gating of REM onset and dopamine modulation of dream emotional content — not a single mechanism. Both are affected by GLP-1 receptor agonists, and both converge on dream experience.
Why Do Ozempic Dreams Get Worse When You Increase the Dose
The dose-escalation pattern is the most consistent detail in anecdotal user reports on patient forums and social media. Descriptions like “going from 1 mg to 2 mg the dreams got WILD,” “every single night since starting,” and “woke up not knowing where I am 2-3 times a week” appear repeatedly. No clinical trial has formally tracked dream content or intensity across dose escalation levels.
The pharmacokinetic basis is straightforward. Higher semaglutide doses produce higher peak plasma concentrations, and semaglutide accesses the brain through circumventricular organs — brain regions that lack a complete blood-brain barrier. Gabery et al. (2020) mapped semaglutide’s brain distribution in rodents, showing drug exposure in regions including the hypothalamus, with dose-dependent effects on body weight and food intake. Greater drug exposure in the brain means greater activation of GLP-1 receptors on dorsal raphe serotonin neurons — the pathway most directly linked to dream-relevant neurotransmitter changes.
Fang et al. (2023) used EEG recordings in rats treated with liraglutide and found that liraglutide dose-dependently increased NREM sleep at all but the lowest dose tested, with effects occurring primarily during the active period whether administered at light or dark onset. REM sleep was not separately analyzed. If NREM expands at the expense of REM, the remaining REM episodes may become compressed and more intense — though whether this produces vivid dreams specifically has not been tested. Kolling et al. (2024) also measured sleep in their mouse model, finding reduced sleep during the light phase in socially isolated mice with disrupted GLP-1 receptor expression.
Badulescu et al. (2024), in a systematic review restricted to human studies, reported that GLP-1 receptor agonists may modulate dopaminergic signalling and reward circuits — the same circuits that overlap with dream emotional processing. The dose-dependent nature of reward-circuit modulation is consistent with the dose-dependent dream intensification that users describe.
Based on anecdotal forum reports, many users describe a 2-4 week adaptation period at each stable dose, during which dreams gradually normalize. Each dose increase then resets the cycle — a pattern consistent with the brain’s serotonin and dopamine systems partially adjusting to a given level of GLP-1 receptor activation, then being overwhelmed by a higher dose. No clinical study has measured this adaptation timeline.
Do Ozempic Dreams Go Away Over Time
The adaptation pattern described in user forums follows a reported curve: dream intensity peaks in the first 1-2 weeks after starting or increasing a dose, then gradually decreases over the following 2-4 weeks. When the dose is escalated, the pattern reportedly repeats. Some users describe full resolution of vivid dreams at a maintenance dose, while others report persistent effects throughout treatment. These timelines are based on self-reports, not controlled measurement.
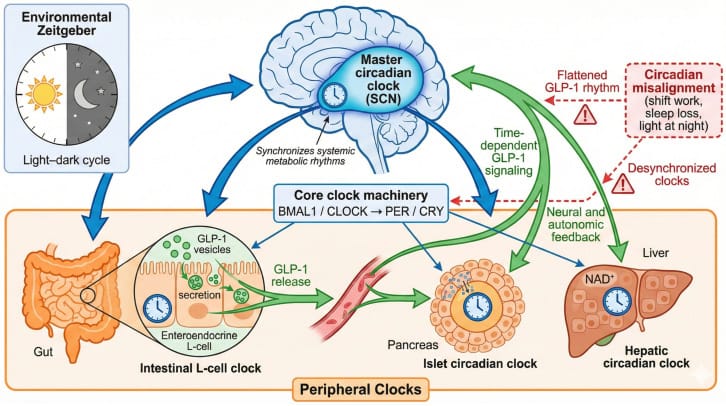
Gandhi et al. (2026) framed GLP-1 receptor agonists as “chronometabolic modulators” — drugs that operate at the intersection of metabolism, circadian biology, and sleep-wake regulation. No prospective clinical trial has examined dream phenomenology on GLP-1 drugs. The adaptation question therefore cannot be definitively answered with current clinical evidence. The user-reported patterns are consistent with neurochemical adaptation but have not been validated in controlled settings.
Why individual variation exists is a matter of neurobiology. Serotonin receptor density in the dorsal raphe, baseline dopamine tone in the mesolimbic system, and GLP-1 receptor expression levels all differ between individuals. People whose serotonin systems are more sensitive to GLP-1 receptor activation would be expected to experience stronger and longer-lasting dream effects. Prior sleep architecture also matters — someone with pre-existing REM fragmentation may respond differently than someone with consolidated REM periods.
Mifsud et al. (2025), in a review from the Mayo Clinic’s Center for Sleep Medicine, examined the impact of GLP-1 agonists on sleep disorders with a focus on sleep apnea. The review raises questions about whether GLP-1 RA benefits extend beyond weight-mediated effects to direct respiratory or neurological mechanisms, but does not address dream-related outcomes. Until prospective studies measure dream content and intensity alongside polysomnography in GLP-1 drug users, the question of whether adaptation is reliable remains open.
What Is the Difference Between Vivid Dreams and Nightmares on Ozempic
The underlying mechanism — increased REM intensity and dream recall — is the same for vivid dreams and nightmares. The distinction is emotional content. A vivid dream becomes a nightmare when its content is distressing, and the emotional tone of dreams is shaped by dopamine and amygdala activity during REM sleep.
User reports reflect both ends of this spectrum. Some describe dreams that are merely strange or unusually detailed: “I could smell and taste things in my dream, it felt completely real.” Others describe emotional distress: “insanely vivid and terrifying,” “woke up crying, didn’t want to go back to sleep,” and “feel exhausted every morning because of the nightmares.”
As described in the mechanism section above, Aranas et al. (2025) demonstrated in mice and rats that semaglutide attenuates cocaine-evoked dopamine elevation in the nucleus accumbens. Dopamine in this region modulates reward and aversion processing during wakefulness; whether semaglutide affects dopamine in this region during sleep has not been tested. If dopamine modulation extends to sleep states, altered emotional valence of dream content is plausible — shifting experiences that might otherwise register as neutral or positive toward negative or threatening emotional tones. Users with pre-existing anxiety, high stress levels, or trauma history may be particularly susceptible to this shift.
Night terrors and sleep paralysis are separate phenomena from vivid dreams and nightmares. Night terrors occur during deep NREM sleep, not during REM, and sleep paralysis occurs during transitions between REM and wakefulness — both are addressed in the FAQ below. Since Fang et al. (2023) showed that liraglutide alters NREM sleep duration, changes to the NREM-REM transition boundary are mechanistically plausible — but no clinical data confirms a direct link between GLP-1 drugs and either condition.
The practical consequence of vivid nightmares extends beyond the dreams themselves. Emotionally distressing dreams fragment sleep and reduce sleep quality even when total sleep time is adequate. Users describing morning exhaustion despite a full night in bed may be experiencing this effect — the subjective experience of sleeping without resting.
Dream disruption is one way metabolic changes from GLP-1 drugs can affect sleep quality. The same medications may also affect blood sugar stability, the brain’s arousal system, and neurotransmitter production — each potentially contributing to different types of sleep disruption. Identifying which causes might be involved in your specific pattern is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Are Vivid Dreams a Listed Side Effect of Ozempic
Adverse event reporting in clinical trials relies on spontaneous patient reports and predefined categories. Dream content, vividness, and emotional distress are not standard categories in pharmaceutical trial protocols. No prospective trial has evaluated dream phenomenology on GLP-1 receptor agonists. User forums and social media discussions suggest sleep disturbance is a frequent non-gastrointestinal complaint among GLP-1 drug users — a signal that trial-measured adverse events may undercount subjective sleep-related side effects.
Can Ozempic Affect Memory Consolidation Through Dream Disruption
During REM sleep, the brain replays and consolidates emotionally significant memories while reducing the emotional charge attached to them. Disrupted REM — whether through fragmentation, compression, or altered neurotransmitter balance — can impair this process. Anderberg et al. (2017) established that GLP-1 receptor activation modulates dorsal raphe serotonin neurons, which are the primary regulators of REM onset. If semaglutide alters the structure of REM episodes, downstream effects on memory processing are plausible but unconfirmed. This remains a research question, not a demonstrated effect.
Does Ozempic Affect Sleep Differently Than Mounjaro for Dreams
The GLP-1 receptor component is the likely driver of dream effects, given the established connections between GLP-1 receptors, dorsal raphe serotonin neurons, and mesolimbic dopamine. Tirzepatide’s additional GIP receptor agonism may or may not add further modulation of dream-related circuits — GIP receptor distribution in the brain is less well characterized than GLP-1 receptor distribution. Li et al. (2025) conducted a meta-analysis of GLP-1 receptor agonists for sleep apnea, with a subgroup comparison finding tirzepatide more effective than liraglutide at reducing apnea-hypopnea index, but did not address dream-related effects. Until head-to-head studies examine dream content on semaglutide versus tirzepatide, the question remains open.
Can Ozempic Cause Sleep Paralysis or Night Terrors
Night terrors and nightmares are frequently confused but are distinct sleep phenomena. Nightmares occur during REM sleep and are remembered upon waking. Night terrors occur during deep NREM sleep and are typically not remembered. Sleep paralysis occurs when the muscle atonia of REM persists into the waking transition. Fang et al. (2023) showed that liraglutide alters NREM sleep duration and bout length. If GLP-1 drugs shift the NREM-REM boundary, transition-state phenomena like sleep paralysis become more plausible — but no clinical report has systematically documented these events in GLP-1 drug users.
Why Do Some People Get Ozempic Dreams and Others Do Not
The GLP-1 receptor is not uniformly expressed across all brains. Anderberg et al. (2017) identified GLP-1 receptors on dorsal raphe serotonin neurons, but receptor density varies between individuals. Someone with higher GLP-1 receptor expression in the dorsal raphe would experience stronger serotonergic effects from the same semaglutide dose. Kolling et al. (2024) demonstrated that GLP-1 receptor expression on serotonin neurons varies across conditions (social isolation reduced Glp1r expression across raphe nuclei), supporting the principle that GLP-1 receptor levels on serotonin neurons are variable and functionally consequential. Baseline sleep architecture also plays a role — a person who already has long, consolidated REM periods and high dream recall is starting from a different baseline than someone who rarely remembers dreams.
Related Reading
- Metabolic Sleep Disruption — The full metabolic sleep framework covering glucose regulation, insulin resistance, mitochondrial decline, NAD+ loss, and fat metabolism
- Does Ozempic Cause Insomnia What GLP-1 Drugs Do to Your Sleep — The broad overview of GLP-1 sleep side effects and mechanisms
- Why Does Ozempic Make You Tired But Keep You Awake — How GLP-1 drugs activate orexin/wakefulness circuitry while appetite suppression can create fatigue
- Why Does Ozempic Wake You Up at 3am — How lower overnight fuel availability and counter-regulatory hormones can contribute to 3am waking
- Does Eating Too Little on Ozempic Cause Insomnia — How appetite suppression can affect tryptophan, serotonin, melatonin, and overnight glucose stability
- Does Your Ozempic Injection Day Disrupt Your Sleep The 24-48 Hour Pattern — Why sleep disruption can follow a weekly pattern tied to injection timing and peak drug exposure
- Does Your Semaglutide Dose Increase Restart Your Insomnia Why Each Titration Can Disrupt Sleep Again — Why each titration step can restart sleep disruption before adaptation catches up
- Can You Take Melatonin, Magnesium, or Sleep Aids While on Ozempic — How melatonin, magnesium, DORAs, and other sleep aids intersect with GLP-1 physiology
- Does Ozempic Give You Anxiety at Night Why GLP-1 Drugs May Contribute to Nighttime Panic — How orexin activation, HPA-axis signaling, and glucose instability may contribute to nighttime anxiety
- Do GLP-1 Drugs Disrupt Your Body Clock Ozempic, Circadian Rhythms, and Melatonin — How GLP-1 secretion rhythms, food timing, peripheral clocks, and melatonin intersect
- Ozempic Night Sweats: How GLP-1 Drugs May Affect Temperature at Night — How GLP-1 drugs may affect nighttime sweating through autonomic tone and thermoregulation
- Ozempic Nausea and Sleep: How Stomach Side Effects Keep You Awake at Night — How delayed gastric emptying, reflux, and nausea can fragment sleep
- Will Your Sleep Go Back to Normal When You Stop Ozempic — What is known and not known about sleep after semaglutide discontinuation
- Does Ozempic Muscle Loss Affect Your Sleep Sarcopenia, Temperature, and Deep Sleep — How lean-mass loss, thermoregulation, and deep sleep can interact during GLP-1 weight loss
- Could Ozempic Contribute to Restless Legs at Night Iron, B12, and the Dopamine Connection — How iron, B12, dopamine, and rapid weight loss may intersect with restless legs at night
- Does Compounded Semaglutide Affect Sleep Differently Than Brand-Name Ozempic — How dose variability, formulation differences, and adverse-event tracking can affect sleep side effects
- Ozempic and Sleep After 40: Why GLP-1 Side Effects Can Feel Different as You Age — Why GLP-1 sleep effects can land differently after 40 because of muscle, hormones, and glucose regulation
References
- Anderberg, R. H., Richard, J. E., Eerola, K., López-Ferreras, L., Banke, E., Hansson, C., Nissbrandt, H., Berqquist, F., Gribble, F. M., Reimann, F., Wernstedt Asterholm, I., Lamy, C. M., & Skibicka, K. P. (2017). Glucagon-Like Peptide 1 and Its Analogs Act in the Dorsal Raphe and Modulate Central Serotonin to Reduce Appetite and Body Weight. Diabetes, 66(4), 1062-1073. https://pubmed.ncbi.nlm.nih.gov/28057699/
- Aranas, C., Caffrey, A., Edvardsson, C. E., Schmidt, H. D., & Jerlhag, E. (2025). Semaglutide suppresses cocaine taking, seeking, and cocaine-evoked dopamine levels in the nucleus accumbens. European Neuropsychopharmacology, 98, 1-10. https://pubmed.ncbi.nlm.nih.gov/40644799/
- Badulescu, S., Tabassum, A., Le, G. H., Wong, S., Phan, L., Gill, H., Llach, C. D., McIntyre, R. S., Rosenblat, J., & Mansur, R. (2024). Glucagon-like peptide 1 agonist and effects on reward behaviour: A systematic review. Physiology & Behavior, 283, 114622. https://pubmed.ncbi.nlm.nih.gov/38945189/
- Fang, J., Miller, P., & Grigson, P. S. (2023). Sleep is increased by liraglutide, a glucagon-like peptide-1 receptor agonist, in rats. Brain Research Bulletin, 192, 142-155. https://pubmed.ncbi.nlm.nih.gov/36410565/
- Gabery, S., Salinas, C. G., Paulsen, S. J., Ahnfelt-Ronne, J., Alanentalo, T., Baquero, A. F., Buckley, S. T., Farkas, E., Fekete, C., Frederiksen, K. S., Helms, H. C. C., Jeppesen, J. F., John, L. M., Pyke, C., Nohr, J., Lu, T. T., Polex-Wolf, J., Prevot, V., Raun, K., … Hogendorf, W. F. J. (2020). Semaglutide lowers body weight in rodents via distributed neural pathways. JCI Insight, 5(6), e133429. https://pubmed.ncbi.nlm.nih.gov/32213703/
- Gandhi, A., Phyu, E. M., Koom-Dadzie, K., Dickson, K. B., & Halm, J. (2026). GLP-1 Receptor Agonists at the Crossroads of Circadian Biology, Sleep, and Metabolic Disease. International Journal of Molecular Sciences, 27(6), 2853. https://pubmed.ncbi.nlm.nih.gov/41898712/
- Kolling, L. J., Khan, K., Wang, R., Pierson, S. R., Hartman, B. D., Balasubramanian, N., Guo, D.-F., Rahmouni, K., & Marcinkiewcz, C. A. (2024). Interaction of serotonin/GLP-1 circuitry in a dual preclinical model for psychiatric disorders and metabolic dysfunction. Psychiatry Research, 337, 115951. https://pubmed.ncbi.nlm.nih.gov/38735240/
- Li, M., Lin, H., Yang, Q., Zhang, X., Zhou, Q., Shi, J., & Ge, F. (2025). Glucagon-like peptide-1 receptor agonists for the treatment of obstructive sleep apnea: a meta-analysis. Sleep, 48(4), zsae280. https://pubmed.ncbi.nlm.nih.gov/39626095/
- Mifsud, C. S., Kolla, B. P., Rushlow, D. R., & Mansukhani, M. P. (2025). The impact of GLP-1 agonists on sleep disorders: spotlight on sleep apnea. Expert Opinion on Pharmacotherapy, 26(14-15), 1529-1538. https://pubmed.ncbi.nlm.nih.gov/41114602/
Written by Kat Fu, M.S., M.S. – Last reviewed: May 2026 – 9 references cited
