Melatonin is the top-selling sleep supplement in the world. People take it expecting it to knock them out. When it does not work, they take more. The problem is not melatonin — it is a mismatch between what melatonin does (move circadian timing) and what people over 40 need (repair of multiple disrupted sleep mechanisms).
This article covers why melatonin is effective for jet lag, why it does not work for chronic insomnia in adults over 40, and why higher doses make things worse. It does not cover the full circadian architecture. Circadian disruption is one of several causes of sleep disruption in midlife — the full overview is in the parent article on circadian sleep disruption.
How Does Melatonin Reset Your Body Clock During Jet Lag?
The melatonin phase response curve — first mapped by Lewy et al. (1992) — shows that the direction of clock movement depends on when you take melatonin. Evening melatonin advances the clock forward (useful for eastward travel). Morning melatonin delays the clock backward (useful for westward travel). Taking melatonin at the wrong time does not help — it worsens jet lag by pushing the clock in the wrong direction.
The strongest evidence for melatonin in jet lag comes from the Cochrane meta-analysis by Herxheimer and Petrie (2002), which pooled 10 randomized controlled trials. Nine of the 10 trials showed melatonin was effective for travelers crossing 5 or more time zones. Doses ranging from 0.5 to 5 milligrams were effective, though the 5-milligram dose produced faster sleep onset than the 0.5-milligram dose. Doses above 5 milligrams provided no additional benefit.
The gold-standard measurement for confirming whether the clock has moved is dim-light melatonin onset (DLMO) — the time point when endogenous melatonin begins rising under low-light conditions. Pandi-Perumal et al. (2007) established DLMO as the primary marker of SCN phase position. Murray et al. (2024) have since validated at-home salivary DLMO methods, making it possible to measure circadian phase outside a lab.
Jet lag is the ideal use case for melatonin because it involves a single, acute timing mismatch with an otherwise intact clock mechanism. The SCN is healthy, the receptors are functional, the downstream temperature and cortisol rhythms are intact — the clock is set to the wrong time zone. Melatonin fixes that.
Why Is Melatonin a Timing Cue and Not a Sleeping Pill?
Cruz-Sanabria et al. (2023) reviewed the mechanisms by which melatonin promotes sleep, distinguishing the chronobiotic pathway from direct sedation. Melatonin acts through GABA modulation, thermoregulation interaction, and reduction of the circadian alerting drive — the wake-promoting output from the SCN that keeps you awake during daytime hours. None of these mechanisms produce the rapid-onset sedation people expect from a sleeping pill.
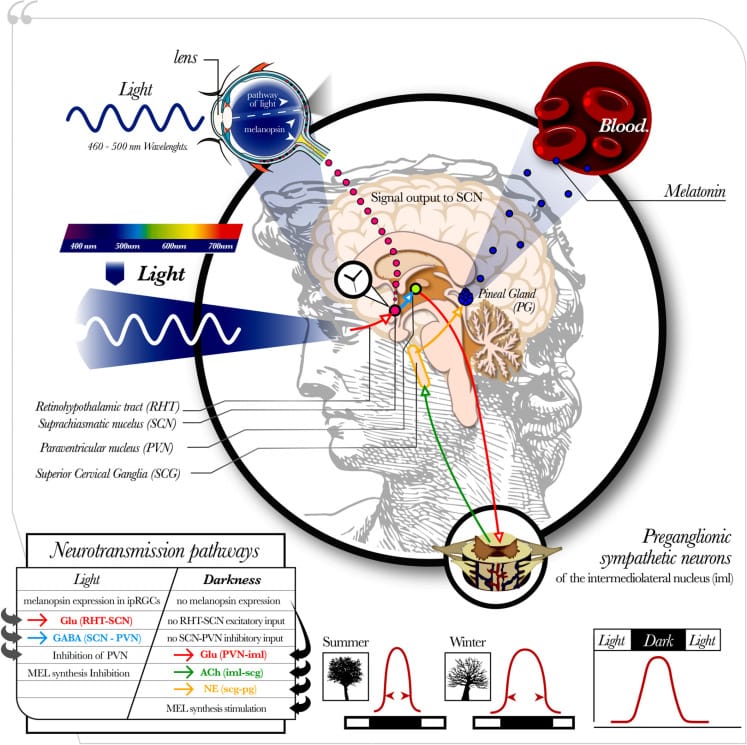
The distinction between melatonin’s two receptor subtypes matters here. Comai and Gobbi (2024) described how MT1 receptors primarily regulate REM sleep while MT2 receptors primarily regulate NREM sleep. MT2 receptors also mediate circadian phase-shifting — the mechanism that moves the clock to a new time zone during jet lag correction. The MT1 pathway contributes to the mild sleep-promoting effect, but it is not sedation in the way benzodiazepines or antihistamines produce sedation.
This is why “I took melatonin and nothing happened” is the expected response when someone uses it as a sleeping pill. Melatonin does not produce a noticeable sensation of drowsiness. It is not supposed to. The people reporting on forums that melatonin “doesn’t knock me out” or that they are “still staring at the ceiling an hour after taking it” are measuring the wrong outcome. Melatonin’s job is to tell the clock what time it is — not to override wakefulness on demand.
Why Does Melatonin Not Work for Chronic Insomnia After 40?
The American Academy of Sleep Medicine (AASM) practice guideline authored by Sateia et al. (2017) recommends against using melatonin for both sleep onset insomnia and sleep maintenance insomnia in adults. The guideline committee reviewed the available randomized trial evidence and concluded that melatonin’s effects were not sufficient to support recommendation.
The meta-analysis by Choi et al. (2022), which pooled 24 randomized controlled trials of melatonin for chronic insomnia, found that melatonin was not significantly effective in improving sleep onset latency, total sleep time, or sleep efficiency in adults. Earlier meta-analyses that pooled across all age groups and sleep disorders found modest overall effects — around 7 minutes of faster sleep onset and 8 minutes of additional sleep time. For someone lying awake for two hours every night, these margins — with no improvement in middle-of-the-night waking — do not solve the problem.
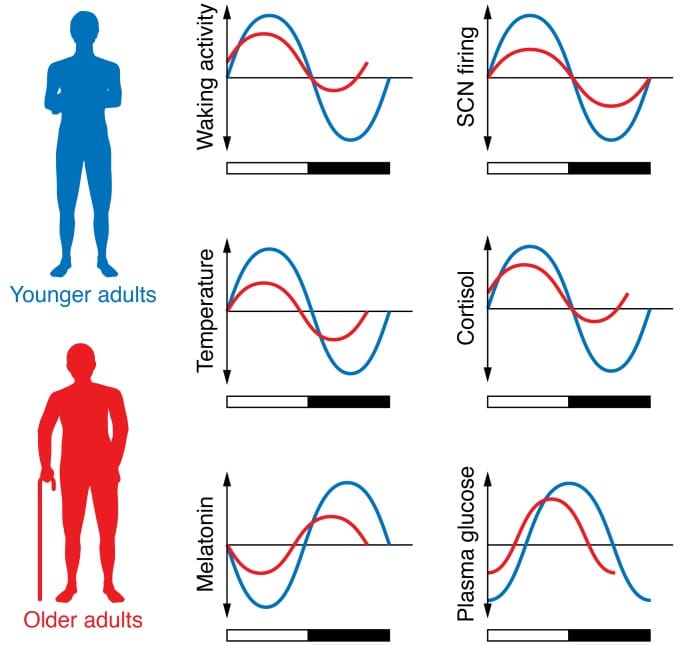
The reason melatonin cannot fix chronic insomnia after 40 is that chronic insomnia is not a timing problem — it is a multi-mechanism degradation problem. Hood and Amir (2017) documented the age-related changes in the SCN that undermine circadian function: reduced expression of the neuropeptides AVP (arginine vasopressin) and VIP (vasoactive intestinal peptide), fewer GABAergic synapses between SCN neurons, and an overall reduction in the amplitude of the SCN firing rhythm. The core body temperature rhythm — a key sleep onset cue — loses 20 to 40 percent of its amplitude in men.
Adding exogenous melatonin to a degraded SCN does not restore what has been lost. The timing cue arrives, but the downstream components that execute sleep — temperature drop, cortisol suppression, consolidated NREM architecture — are too weakened to respond to it with the same force. Endogenous melatonin production declines with age, but replacing it with a supplement does not reverse the receptor changes, the SCN neuronal losses, or the flattened temperature rhythm. This area of research is being mapped, but the direction is consistent: the problem is architectural, not a missing molecule.
That is why “melatonin doesn’t work” for chronic insomnia is not a melatonin problem. It is a mismatch between a single-mechanism tool and a multi-mechanism condition.
Does Taking More Melatonin Help You Sleep Better?
Cruz-Sanabria et al. (2024) conducted a dose-response meta-analysis of 26 randomized melatonin trials and found that the maximum effect peaked at around 4 milligrams. Beyond 4 milligrams, there was no additional benefit. The meta-analysis also found that the optimal administration time was approximately 3 hours before the desired bedtime — not 30 minutes before, which is how people typically take it.
One pharmacological explanation for the dose ceiling may involve receptor desensitization. Comai and Gobbi (2024) described the distinct roles of MT1 and MT2 receptors in sleep regulation. At supraphysiological doses, melatonin receptors can become less responsive — taking 10 milligrams does not produce twice the effect of 5 milligrams.
This explains a common pattern: someone takes 3 milligrams, feels no sedation (because melatonin is not a sedative), increases to 5 milligrams, then 10, then 20 — and each increase produces diminishing returns or no change at all. The expected pharmacological outcome of supraphysiological melatonin doses is a plateau or diminished response, not deeper sleep.
The physiological dose range — what produces blood melatonin levels comparable to the body’s own nighttime production — is 0.3 to 1.0 milligrams. This range is supported by the phase-response curve data and pharmacokinetic studies. Over-the-counter melatonin products typically sell 5 to 10 milligram doses, which are 10 to 30 times higher than what the evidence supports.
Many people have more than one cause contributing to their sleep disruption. Circadian timing problems — including the melatonin-related mechanisms covered here — may compound with metabolic, hormonal, inflammatory, or autonomic factors. Identifying which causes might be involved is a useful next step.
Find out which causes might be driving your 3 AM wakeups →
Frequently Asked Questions
Can You Become Dependent on Melatonin?
Long-term melatonin studies have not identified withdrawal effects comparable to those seen with prescription sleep medications. Unlike benzodiazepines and Z-drugs, melatonin does not produce rebound insomnia — a worsening of sleep that occurs after stopping a medication. This is consistent with its mechanism of action: a chronobiotic that adjusts timing rather than a sedative that suppresses arousal circuits.
What Time Should You Take Melatonin for Sleep?
The dose-response meta-analysis by Cruz-Sanabria et al. (2024) found that the optimal timing for melatonin administration was approximately 3 hours before the desired sleep onset. This aligns with the DLMO window — the period when endogenous melatonin begins rising under dim-light conditions. Taking melatonin 30 minutes before bed, which is the standard practice, misses the phase-advance window and reduces effectiveness. The melatonin arrives after the body has already begun its own melatonin rise, adding little to the timing cue.
Does Melatonin Make Some People Feel Groggy the Next Morning?
At supraphysiological doses, melatonin’s half-life means circulating levels can remain elevated into the morning, continuing to activate melatonin receptors during a period when the SCN should be sending wake-promoting output. Lower doses (0.3 to 1.0 milligrams) are less likely to produce this carryover effect because they are eliminated faster and do not saturate the receptor pool. Immediate-release formulations also tend to produce less morning grogginess than prolonged-release formulations at equivalent doses.
Is Prolonged-Release Melatonin Better Than Immediate-Release?
The Wade et al. (2007) randomized controlled trial of Circadin — a 2 milligram prolonged-release melatonin formulation — tested whether sustained melatonin delivery throughout the night would improve sleep quality in older adults. The results were modest: 26 percent of the melatonin group met responder criteria versus 15 percent on placebo. Sleep onset improved. This result is consistent with the core thesis of this article: even the best melatonin formulation reaches a ceiling because it addresses timing, not the multi-mechanism architectural degradation that causes chronic insomnia after 40.
Related Reading
- Circadian Rhythm Disruption: How a Misaligned Internal Clock Fragments Sleep, Drives 3AM Wakeups, and Reduces Sleep Depth — the full overview of circadian sleep disruption, including melatonin, cortisol, body temperature, peripheral clocks, and early-morning waking
- Is Your Pineal Gland Calcifying? How Age-Related Melatonin Production Loss Differs From Receptor Decline — how pineal calcification, melatonin output, and receptor decline diverge with age
- How Many Melanopic Lux Do You Need for Circadian Entrainment? — how melanopic light exposure sets the clock more directly than ordinary lux
- How Much Blue Light From Your Phone Suppresses Melatonin? The Dose-Duration Relationship — how screen brightness, duration, and melanopic irradiance affect melatonin timing
- Do Blue Light Glasses Help You Sleep? Amber vs Standard Lenses — What Studies Show — how amber lenses, clear lenses, and evening screens differ in sleep studies
- How Long Does It Take to Reset Your Circadian Rhythm? — why the reset timeline differs for the SCN, peripheral clocks, and behavioral cues
- How Does Caffeine Delay Your Circadian Clock by 40 Minutes? — how caffeine delays circadian phase through adenosine and clock-gene timing
References
1. Herxheimer, A., & Petrie, K. J. (2002). Melatonin for the prevention and treatment of jet lag. Cochrane Database of Systematic Reviews, (2), CD001520. https://pubmed.ncbi.nlm.nih.gov/12076414/
2. Lewy, A. J., Ahmed, S., Jackson, J. M., & Sack, R. L. (1992). Melatonin shifts human circadian rhythms according to a phase-response curve. Chronobiology International, 9(5), 380-392. https://pubmed.ncbi.nlm.nih.gov/1394610/
3. Pandi-Perumal, S. R., Smits, M., Spence, W., Srinivasan, V., Cardinali, D. P., Lowe, A. D., & Kayumov, L. (2007). Dim light melatonin onset (DLMO): A tool for the analysis of circadian phase in human sleep and chronobiological disorders. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 31(1), 1-11. https://pubmed.ncbi.nlm.nih.gov/16884842/
4. Murray, J. M., Stone, J. E., Abbott, S. M., Bjorvatn, B., Burgess, H. J., Cajochen, C., Dekker, J. J., Duffy, J. F., Epstein, L. J., Garbazza, C., Harsh, J., Klerman, E. B., Lane, J. M., Lockley, S. W., Pavlova, M. K., Quan, S. F., Reid, K. J., Scheer, F. A. J. L., Sletten, T. L., Wright, K. P., Jr., Zee, P. C., Phillips, A. J. K., Czeisler, C. A., & Rajaratnam, S. M. W. (2024). A protocol to determine circadian phase by at-home salivary dim light melatonin onset assessment. Journal of Pineal Research, 76(5), e12994. https://pubmed.ncbi.nlm.nih.gov/39158010/
5. Cruz-Sanabria, F., Carmassi, C., Bruno, S., Bazzani, A., Carli, M., Scarselli, M., & Faraguna, U. (2023). Melatonin as a chronobiotic with sleep-promoting properties. Current Neuropharmacology, 21(4), 951-987. https://pubmed.ncbi.nlm.nih.gov/35176989/
6. Comai, S., & Gobbi, G. (2024). Melatonin, melatonin receptors and sleep: Moving beyond traditional views. Journal of Pineal Research, 76(7), e13011. https://pubmed.ncbi.nlm.nih.gov/39400423/
7. Sateia, M. J., Buysse, D. J., Krystal, A. D., Neubauer, D. N., & Heald, J. L. (2017). Clinical practice guideline for the pharmacologic treatment of chronic insomnia in adults: An American Academy of Sleep Medicine clinical practice guideline. Journal of Clinical Sleep Medicine, 13(2), 307-349. https://pubmed.ncbi.nlm.nih.gov/27998379/
8. Choi, K., Lee, Y. J., Park, S., Je, N. K., & Suh, H. S. (2022). Efficacy of melatonin for chronic insomnia: Systematic reviews and meta-analyses. Sleep Medicine Reviews, 66, 101692. https://pubmed.ncbi.nlm.nih.gov/36179487/
9. Hood, S., & Amir, S. (2017). The aging clock: Circadian rhythms and later life. Journal of Clinical Investigation, 127(2), 437-446. https://pubmed.ncbi.nlm.nih.gov/28145903/
10. Cruz-Sanabria, F., Bruno, S., Crippa, A., Frumento, P., Scarselli, M., Skene, D. J., & Faraguna, U. (2024). Optimizing the time and dose of melatonin as a sleep-promoting drug: A systematic review of randomized controlled trials and dose-response meta-analysis. Journal of Pineal Research, 76(5), e12985. https://pubmed.ncbi.nlm.nih.gov/38888087/
11. Wade, A. G., Ford, I., Crawford, G., McMahon, A. D., Nir, T., Laudon, M., & Zisapel, N. (2007). Efficacy of prolonged release melatonin in insomnia patients aged 55-80 years: Quality of sleep and next-day alertness outcomes. Current Medical Research and Opinion, 23(10), 2597-2605. https://pubmed.ncbi.nlm.nih.gov/17875243/
12. Ferracioli-Oda, E., Qawasmi, A., & Bloch, M. H. (2013). Meta-analysis: Melatonin for the treatment of primary sleep disorders. PLoS ONE, 8(5), e63773. https://pubmed.ncbi.nlm.nih.gov/23691095/
Written by Kat Fu, M.S., M.S. ? Last reviewed: May 2026 ? 12 references cited
