The claim that gut bacteria affect the brain appears in health media with increasing frequency. The evidence that proves how — through which anatomical pathway, at what speed, and with what degree of dependence — is less commonly discussed. The vagotomy experiments provide the most direct proof available: sever the vagus nerve, and probiotic effects on the brain stop.
This article covers three lines of evidence — the behavioral vagotomy experiments, the electrophysiological recordings showing minute-speed nerve activation, and the 2025 neuropod cell discovery — along with the translation gap from mice to humans, and what the mechanism means for choosing probiotics. For the broader autonomic framework connecting the vagus nerve to sleep, see the autonomic sleep disruption pillar. The vagus nerve is the relay for multiple gut-brain pathways that affect sleep architecture, and the probiotic-vagal connection is one component of that larger picture.
What Happened When Scientists Cut the Vagus Nerve and Gave Mice Probiotics?
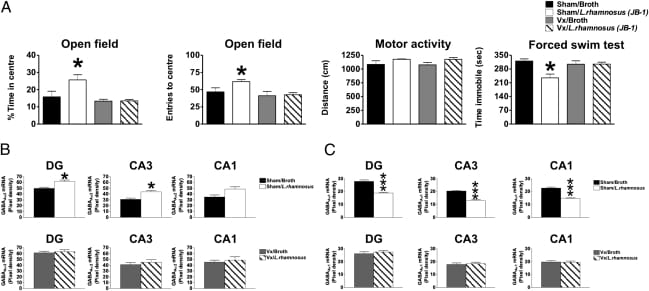
Bravo et al. (2011) administered L. rhamnosus JB-1 to BALB/c mice for 28 days and measured behavioral, neurochemical, and hormonal outcomes. JB-1-treated mice showed reduced anxiety-like behavior on the elevated plus maze and decreased immobility in the forced swim test — both standard assays for anxiety and depression-like behavior in rodents. At the neurochemical level, JB-1 produced region-dependent changes in GABA receptor subunit mRNA expression: GABA(B1b) increased in the cingulate and prelimbic cortices while decreasing in the hippocampus, amygdala, and locus coeruleus. Stress-induced corticosterone was attenuated compared to vehicle controls.
The critical finding: mice that had undergone subdiaphragmatic vagotomy before the probiotic regimen showed no behavioral improvements and no corticosterone attenuation. The anxiety reduction, antidepressant-like effects, and stress hormone suppression were abolished. Most GABA receptor changes were also prevented, though some hippocampal alterations persisted in modified form. The bacteria still reached the gut, but without an intact vagus nerve, the central behavioral and endocrine effects did not occur.
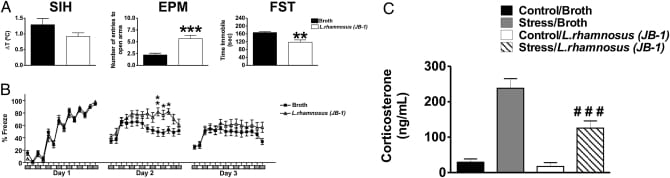
Prior work supports strain specificity for this class of effect. Perez-Burgos et al. (2013) showed that Lactobacillus salivarius — a different species — and sterile growth media produced no vagal nerve response, while JB-1 activated vagal afferents within minutes. The probiotic effect was not from any Lactobacillus. It required a specific strain acting through the vagus nerve.
Liu et al. (2021) extended these findings a decade later. Using the same model — JB-1 in BALB/c mice with and without vagotomy — they measured immune outcomes alongside behavior. In vagally intact mice, JB-1 increased the frequency of splenic regulatory T cells and decreased activated microglia in the hippocampus. Vagotomy abolished both immune effects alongside the behavioral and HPA axis effects.
An additional finding from the 2021 study: vagotomy alone — independent of any probiotic — increased hippocampal microglial activation. Basal vagal tone is required to maintain microglial homeostasis in the brain. The vagus nerve is not a passive conduit waiting for probiotic input. It is an active regulator of neuroimmune status.
This reframed the mechanism from a neural relay model to a neuroimmunoendocrine pathway. The vagus nerve orchestrates behavioral, hormonal, and immune responses to gut bacteria simultaneously. Severing it removes all three.
Can Probiotics Directly Activate Vagal Nerve Fibers?
The vagotomy experiments showed that the vagus nerve was required. The electrophysiological work showed how fast the connection works.
Perez-Burgos et al. (2013) used ex vivo extracellular recordings from the mesenteric nerve bundle of mouse jejunal segments. When live JB-1 was applied to the gut lumen, single-unit and multiunit vagal afferent firing rates increased within minutes. The response was strain-specific and required live bacteria. Lactobacillus salivarius and sterile growth media produced no change.
JB-1 also augmented vagal afferent discharge in response to intraluminal distension — the nerve became more sensitive to mechanical stimulation while JB-1 was present. Prior subdiaphragmatic vagotomy abolished all electrophysiological effects, confirming that the neural changes occurred through vagal afferents rather than spinal afferents or humoral routes.
The speed matters. Behavioral and neurochemical effects of probiotics emerge over weeks. But the vagal nerve response begins within minutes. The gut-brain connection includes fast neural transmission, not only slow hormonal routes.
In 2025, a study published in Nature identified the physical structure that enables this speed. Liu et al. (2025) discovered that the bacterial structural protein flagellin activates Toll-like receptor 5 (TLR5) on a subset of gut epithelial cells — neuropod cells expressing peptide YY (PYY). These neuropod cells form synaptic connections with vagal nodose neurons. When flagellin reaches the gut lining, TLR5-positive neuropod cells release PYY onto NPY2R-expressing vagal neurons, transmitting a microbial input to the brain through a defined cell-to-neuron synapse.
Calcium imaging of 121 neuropod cells showed that 26% responded to flagellin application. Intracolonic flagellin perfusion activated approximately 43% of NPY2R-positive vagal neurons. The appetite-suppressing effect of this circuit was abolished by pharmacological blockade of either TLR5 or the Y2 receptor, confirming strict pathway dependence. Conditional knockout mice lacking TLR5 in PYY-expressing neuropod cells consumed more food and gained more weight than littermate controls.
Grundeken and El Aidy (2025) synthesized this and related evidence in a review describing enteroendocrine cells as the gatekeepers of microbiome-gut-brain communication. These cells, equipped with neuropod structures, form direct glutamatergic synaptic connections with enteric and vagal neurons. Short-chain fatty acids produced by bacterial fermentation activate free fatty acid receptors (FFAR2, FFAR3) on these cells, triggering release of GLP-1, GLP-2, and PYY that modulate satiety, gut motility, and downstream brain activity through vagal afferents.
Does This Evidence Translate From Mice to Humans?
The direct answer: not with JB-1. The strain that produced the landmark vagotomy data has been tested in humans and did not replicate the mouse findings.
Kelly et al. (2017) conducted a randomized, double-blind, placebo-controlled crossover trial with 29 healthy adult men. Participants consumed JB-1 over an 8-week crossover period. The within-person comparison measured mood, anxiety, sleep quality, cognitive processing, cortisol response to a laboratory stressor, and inflammatory biomarkers (interleukin-6).
None of the outcomes differed between the probiotic and placebo conditions. Self-reported mood and anxiety: no difference. EEG-based cognitive performance: no difference. Cortisol stress response: no difference. Inflammatory markers: no difference. Working memory: no difference. The authors concluded that JB-1 “was not superior to placebo” in modifying stress-related measures in healthy male subjects.
This result does not mean the vagal mechanism is wrong. It means that a strain producing robust effects in an inherently anxious mouse line (BALB/c) did not produce measurable effects in healthy human men. Several factors contribute to the translation gap:
- Mouse and human gut ecology differ in pH, transit time, microbiome composition, and mucosal immune environment
- Dose scaling from mouse to human is non-linear and poorly standardized
- BALB/c mice are bred for high anxiety — a population that may be more responsive to anxiolytic approaches than healthy humans
- Strain colonization in the human gut is variable and often transient
- The Kelly trial enrolled 29 participants — powered for large effects but potentially underpowered for moderate ones
The vagal mechanism itself, however, has human evidence — from a different formulation. Morkl et al. (2025) conducted a randomized, double-blind, placebo-controlled trial enrolling 86 participants — 40 with major depressive disorder and 46 healthy controls — who received either a multi-species probiotic or placebo twice daily for 3 months. The primary endpoint was vagal nerve function measured by 24-hour ECG-derived heart rate variability.
After 3 months, people with depression receiving the probiotic showed improved morning vagal nerve function compared to those with depression receiving placebo. Gut microbiome analysis revealed increased Christensenellales, including Akkermansia muciniphila, in the probiotic-treated group. Sleep quality improved, including reduced sleep medication use and shortened sleep latency.
This is one of the first human trials to use vagal tone as a primary probiotic endpoint — and it confirmed that probiotic-induced microbiome changes can enhance vagal nerve activity in humans. The mechanism is real. The strain that proved it in mice is not the strain that proves it in humans.
Bharwani et al. (2020) added another dimension. Their brain activation mapping study showed that vagotomy abolished the majority of JB-1-induced c-Fos neuronal activation in mice — but not all. A subset of brain regions remained responsive after vagotomy, revealing parallel vagal-independent communication pathways. The vagus nerve is the primary and fastest route, but short-chain fatty acids crossing the blood-brain barrier and immune mediators like cytokines and regulatory T cells provide complementary, slower routes.
For choosing probiotics: follow the human trial evidence, not the mouse mechanistic data. Strains with human RCT support for sleep and stress outcomes — including strains with human trial evidence for sleep — offer a more reliable basis for selection than the strains that produced the cleanest mouse experiments. The mechanism is the same. The effective strains differ across species.
What Does the Vagotomy Evidence Mean for Choosing Probiotics?
If the vagus nerve is the required relay for probiotic-to-brain communication, then vagal health determines whether probiotics reach their central targets. This has practical implications.
Vagal tone as a prerequisite. Chronic stress reduces vagal tone — measured as decreased heart rate variability. Chronic inflammation suppresses vagal output through the inflammatory reflex. Post-viral conditions, including long COVID, can damage vagal fibers directly. Aging is associated with progressive vagal decline. In any of these contexts, a probiotic may reach the gut and produce local effects without generating the vagal input needed for brain and sleep outcomes.
Multi-strain formulations may engage multiple gut-vagal pathways. The JB-1 story illustrates the risk of depending on a single strain-receptor interaction. The 2025 Morkl et al. trial used a multi-species probiotic — not a single strain — and produced measurable vagal improvements in humans. Multi-strain formulations interact with a broader range of enteroendocrine and neuropod cells, increasing the probability of engaging vagal afferents through more than one receptor pathway.
Supporting vagal function alongside probiotic use. Kurhaluk et al. (2025) mapped the molecular network by which the vagus nerve and gut microbiota jointly regulate the HPA axis, the cholinergic anti-inflammatory reflex, and GABA/serotonin availability. The network is bidirectional: vagal efferents influence gut microbiome composition, and microbiota-derived inputs travel back through vagal afferents. Supporting the vagal side of this circuit — through HRV monitoring to track vagal function, breathing practices that increase parasympathetic output, or vagus nerve stimulation devices — may enhance probiotic efficacy by ensuring the relay is functional.
The vagotomy research also clarifies which outcomes probiotics can and cannot influence through the vagal pathway. Brain effects — GABA receptor expression, anxiety behavior, HPA axis modulation, microglial homeostasis — require vagal integrity. Local gut effects — microbiome composition changes, short-chain fatty acid production, mucosal immune modulation — do not depend on the vagus nerve and will occur regardless. A person with impaired vagal function may still benefit from probiotics for gut health without experiencing the central nervous system effects.
For the gut-vagus-sleep connection and how disrupted sleep itself damages gut microbiome composition, see that article on the bidirectional relationship. And for a practical timeline of when probiotic effects on sleep tend to emerge, see the evidence-based timeline for probiotic sleep improvements.
Gut-vagus-brain pathway impairment may not be the only factor affecting your sleep. Autonomic hyperarousal, metabolic disruptions, inflammatory processes, or hormonal changes may also be contributing. When multiple causes overlap, identifying which ones are active is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Are There Other Pathways Besides the Vagus Nerve for Probiotics to Affect the Brain?
Bharwani et al. (2020) mapped c-Fos immunoreactivity — a marker of neuronal activation — across the mouse brain after oral JB-1 administration. Live JB-1 induced rapid, widespread brain activation at 165 minutes post-gavage. Vagotomy abolished c-Fos activation in the majority of responsive brain regions.
But a subset of brain regions remained c-Fos positive after vagotomy. The vagus nerve accounts for the primary and fastest route of gut-to-brain bacterial communication, but parallel pathways exist. These include:
- Short-chain fatty acids — bacterial fermentation products (butyrate, propionate, acetate) that can cross the blood-brain barrier and modulate neuroinflammation and neurotransmitter production
- Immune mediators — cytokines and regulatory T cells influenced by gut bacteria travel through the bloodstream to affect brain function on a slower timescale
- Enteric nervous system — the gut’s intrinsic nervous system can relay information to the brain through spinal afferents, independently of the vagus
Chronic 28-day JB-1 exposure produced a different regional activation pattern than acute dosing, indicating neural plasticity — the brain adapts to sustained bacterial input over time. Heat-killed bacteria altered vagal activity but did not induce the rapid, widespread brain activation seen with live bacteria — confirming that active bacterial metabolic output is required for the full central response.
The practical implication: the vagus nerve is the dominant pathway, but probiotic effects on the brain are not exclusively vagal-dependent. A person with partial vagal impairment may still experience some central probiotic effects through these secondary routes, though at reduced magnitude and slower onset.
How Long Until Probiotics Reach the Brain Through the Vagus Nerve?
The timeline has two layers. The neural response is fast — Perez-Burgos et al. (2013) recorded increased vagal afferent firing within minutes of JB-1 contacting the gut lumen, and the 2025 neuropod cell discovery shows that the physical relay works at synaptic speed (seconds from neuropod cell to vagal neuron).
But a single nerve impulse does not change GABA receptor expression or HPA axis calibration. Those downstream effects require sustained input over weeks. In the Bravo et al. (2011) mouse study, behavioral and neurochemical changes were measured after 28 days of daily administration. In the Morkl et al. (2025) human trial, vagal function improvements emerged over 3 months.
The response is fast. The adaptation is slow. For a week-by-week breakdown of when to expect changes in sleep and mood outcomes from probiotics, see the evidence-based timeline for probiotic sleep improvements.
Can Vagus Nerve Damage Reduce Probiotic Effectiveness?
The experimental evidence is clear on this point. Complete vagotomy eliminates probiotic behavioral effects, corticosterone suppression, regulatory T cell expansion, and microglial homeostasis, and prevents most GABA receptor changes (Bravo et al., 2011; Liu et al., 2021). The bacteria still reach the gut. They produce few measurable central effects.
In humans, complete vagotomy is rare outside of historical surgical contexts. But partial vagal impairment is common — associated with chronic stress, chronic inflammation, post-viral conditions including long COVID, autonomic neuropathy, and aging. The degree of vagal impairment may predict the degree of attenuation in probiotic brain effects.
Liu et al. (2021) showed that vagotomy alone increased hippocampal microglial activation — even without any probiotic. Vagal tone is not a passive relay waiting for input. It is an active regulator of neuroinflammation. Impaired vagal function may not only reduce probiotic efficacy but also create the neuroinflammatory conditions that probiotics are often taken to address.
This creates a practical sequence: assess and support vagal function before relying on probiotics for brain and sleep outcomes. HRV monitoring provides an accessible measure of vagal tone. Vagus nerve stimulation devices offer a direct approach to improving vagal output. Breathing practices that extend the exhale phase increase parasympathetic activity. These approaches support the relay that probiotics depend on.
Related Reading
- Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It — full overview of autonomic causes including vagal tone, GABA, cortisol, and hyperarousal
- Does Lion’s Mane Affect Your Sleep? Why It Helps Some People and Keeps Others Awake — lion’s mane’s dual mechanism: neurotrophic activity, cholinergic activity, and sleep architecture
- Why Do Cholinergic Supplements Give You Vivid Dreams? — the acetylcholine-REM pathway behind supplement-related vivid dreams
- Which Choline Supplement Is Least Likely to Disrupt Your Sleep? A Form-by-Form Comparison — how different choline forms reach the brain and affect sleep
- How Do You Take Nootropics Without Disrupting Your Sleep? A Neuroscience-Based Approach — timing and stacking guidance for preserving sleep while using nootropics
- Why Can You Not Sleep After COVID? What Happened to Your Vagus Nerve — how post-viral autonomic disruption can affect sleep continuity
- Why Does Your Heart Race at Night After COVID? What the Vagus Nerve Has to Do With It — why post-COVID autonomic instability can show up as nighttime tachycardia
- Can POTS and Dysautonomia Cause Insomnia? Why Your Autonomic Nervous System Will Not Let You Rest — how orthostatic intolerance and autonomic instability can fragment sleep
- Can a Viral Infection Cause Chronic Insomnia? — how viral infections can disrupt autonomic regulation and sleep
- Why Will Your Nervous System Not Let You Sleep? Polyvagal Theory and Insomnia — how polyvagal states map to insomnia, hyperarousal, and unrefreshing sleep
- Why Does Trauma Cause Insomnia? How Your Nervous System Stays on Guard at Night — how trauma can keep sleep shaped by hyperarousal and vigilance
- Why Are You Sleeping 12 Hours and Still Exhausted? The Dorsal Vagal Shutdown Explained — the dorsal vagal pattern behind long sleep that still feels unrefreshing
- Which Probiotic Strains Improve Sleep? What the Vagus Nerve Evidence Shows — which probiotic strains have human sleep evidence and how the vagus nerve may be involved
- Does Poor Sleep Damage Your Gut Microbiome? — the relationship between sleep fragmentation, microbiome disruption, and inflammatory load
- How Long Do Probiotics Take to Improve Sleep? What the Research Shows Week by Week — what probiotic sleep studies suggest about timing across weeks
References
- Bharwani, A., West, C., Champagne-Jorgensen, K., McVey Neufeld, K.-A., Ruberto, J., Kunze, W. A., Bienenstock, J., & Forsythe, P. (2020). The vagus nerve is necessary for the rapid and widespread neuronal activation in the brain following oral administration of psychoactive bacteria. Neuropharmacology, 170, 108067. https://pubmed.ncbi.nlm.nih.gov/32224131/
- Bravo, J. A., Forsythe, P., Chew, M. V., Escaravage, E., Savignac, H. M., Dinan, T. G., Bienenstock, J., & Cryan, J. F. (2011). Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proceedings of the National Academy of Sciences of the United States of America, 108(38), 16050–16055. https://pubmed.ncbi.nlm.nih.gov/21876150/
- Grundeken, E., & El Aidy, S. (2025). Enteroendocrine cells: the gatekeepers of microbiome-gut-brain communication. NPJ Biofilms and Microbiomes, 11(1), 179. https://pubmed.ncbi.nlm.nih.gov/40890205/
- Kelly, J. R., Allen, A. P., Temko, A., Hutch, W., Kennedy, P. J., Farid, N., Murphy, E., Boylan, G., Bienenstock, J., Cryan, J. F., Clarke, G., & Dinan, T. G. (2017). Lost in translation? The potential psychobiotic Lactobacillus rhamnosus (JB-1) fails to modulate stress or cognitive performance in healthy male subjects. Brain, Behavior, and Immunity, 61, 50–59. https://pubmed.ncbi.nlm.nih.gov/27865949/
- Kurhaluk, N., Kolodziejska, R., Kaminski, P., & Tkaczenko, H. (2025). Integrative neuroimmune role of the parasympathetic nervous system, vagus nerve and gut microbiota in stress modulation: A narrative review. International Journal of Molecular Sciences, 26(23), 11706. https://pubmed.ncbi.nlm.nih.gov/41373850/
- Liu, W. W., Reicher, N., Alway, E., Rupprecht, L. E., Weng, P., Schaefgen, C., Klein, M. E., Villalobos, J. A., Puerto-Hernandez, C., Kiesling Altun, Y. G., Carbajal, A., Aguayo-Guerrero, J. A., Coss, A., Sahasrabudhe, A., Anikeeva, P., de Araujo, A., Bali, A., de Lartigue, G., Gil-Lievana, E., Gutierrez, R., Miao, E. A., Rawls, J. F., Kaelberer, M. M., & Bohorquez, D. V. (2025). A gut sense for a microbial pattern regulates feeding. Nature, 645(8081), 729–736. https://pubmed.ncbi.nlm.nih.gov/40702192/
- Liu, Y., Sanderson, D., Mian, M. F., McVey Neufeld, K.-A., & Forsythe, P. (2021). Loss of vagal integrity disrupts immune components of the microbiota-gut-brain axis and inhibits the effect of Lactobacillus rhamnosus on behavior and the corticosterone stress response. Neuropharmacology, 195, 108682. https://pubmed.ncbi.nlm.nih.gov/34175326/
- Morkl, S., Narrath, M., Schlotmann, D., Sallmutter, M. T., Putz, J., Lang, J., Brandstatter, A., Pilz, R., Karl Lackner, H., Goswami, N., Steuber, B., Tatzer, J., Lackner, S., Holasek, S., Painold, A., Jauk, E., Wenninger, J., Horvath, A., Spicher, N., Barth, A., Butler, M. I., & Wagner-Skacel, J. (2025). Multi-species probiotic supplement enhances vagal nerve function — results of a randomized controlled trial in patients with depression and healthy controls. Gut Microbes, 17(1), 2492377. https://pubmed.ncbi.nlm.nih.gov/40298641/
- Perez-Burgos, A., Wang, B., Mao, Y.-K., Mistry, B., McVey Neufeld, K.-A., Bienenstock, J., & Kunze, W. (2013). Psychoactive bacteria Lactobacillus rhamnosus (JB-1) elicits rapid frequency facilitation in vagal afferents. American Journal of Physiology. Gastrointestinal and Liver Physiology, 304(2), G211–G220. https://pubmed.ncbi.nlm.nih.gov/23139216/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 9 references cited
