Polyvagal theory, developed by Stephen Porges, provides a framework for understanding why some people cannot fall asleep despite exhaustion — and why others sleep for twelve hours and wake unrefreshed. Both patterns are autonomic, driven by which branch of the nervous system is dominant at bedtime.
This article maps the three polyvagal states to sleep presentations, covers the neuroception-of-safety concept that determines state selection, connects the framework to the hyperarousal insomnia literature, and includes regulation exercises matched to each state. It also addresses the scientific debate around polyvagal theory’s neuroanatomical claims. For the broader autonomic picture, see the autonomic sleep disruption pillar.
What Are the Three Polyvagal States and How Do They Affect Sleep?
Porges (2022) describes neuroception as the neural process by which the nervous system evaluates risk without conscious awareness. This continuous background scan of environmental and relational cues determines which level of the autonomic hierarchy is active. When the nervous system detects safety, the ventral vagal complex supports homeostatic functions — including the conditions required for restorative sleep. Chronic perceived danger suppresses ventral vagal engagement, maintaining sympathetic hyperarousal or dorsal vagal freeze states.
Ventral vagal and restorative sleep. When the ventral vagal state is dominant, parasympathetic output increases and heart rate variability rises. A study in healthy newborns found that NREM-precursor sleep — termed “quiet sleep” in neonatal research — is associated with higher amplitude respiratory sinus arrhythmia and longer heart periods, reflecting vagal output from the nucleus ambiguus during quiet sleep (Porges et al., 1999).
Sympathetic state and insomnia. When the sympathetic branch is dominant, heart rate stays elevated, cortisol persists, and the autonomic nervous system remains in an alert mode. This produces sleep onset difficulty, fragmented sleep, and early waking — a nervous system locked in the sympathetic state, unable to transition to ventral vagal for sleep onset.
Dorsal vagal state and unrefreshing hypersomnia. The dorsal vagal response is a conservation and immobilization state. People in dorsal vagal shutdown may sleep for extended periods, but sleep architecture is impaired — they wake exhausted. For a deeper look at this presentation, see the dorsal vagal shutdown and unrefreshing sleep.
What Is Neuroception and Why Does It Determine Whether You Can Sleep?
Neuroception operates below conscious awareness. It evaluates environmental and interoceptive cues — temperature, ambient sound, darkness, the presence or absence of another person — and determines which autonomic state activates. You do not choose to be in sympathetic hyperarousal at bedtime. Neuroception selects that state based on its evaluation of the environment.
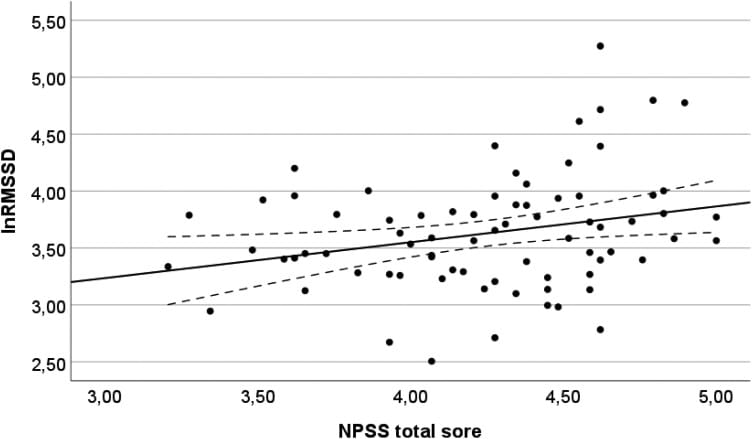
Schwerdtfeger et al. (2025) demonstrated this link using the Neuroception of Psychological Safety Scale and ambulatory heart rate variability monitoring. In Study 1 (N=76), higher self-reported psychological safety perception was associated with higher parasympathetic heart rate variability at rest. In Study 2 (N=245), multi-day ambulatory monitoring showed that safety perception predicted more frequent episodes of HRV increase throughout daily life, even after controlling for physical movement. The authors conclude that “feeling safe and everyday life cardiac autonomic regulation are interrelated.”
Morton et al. (2024) validated the Neuroception of Psychological Safety Scale (NPSS) — the first standardized instrument measuring neuroception as defined by polyvagal theory. The scale has three subscales: compassion, social engagement, and body sensations.
Why this matters for sleep. Neuroception of danger activates the sympathetic state — producing the hyperarousal that prevents sleep onset. Neuroception of life-danger activates the dorsal vagal state — producing immobilization and unrefreshing hypersomnia. Neuroception of safety supports the ventral vagal state that enables restorative sleep.
The sleep environment feeds neuroception directly. Temperature, ambient sound, darkness, co-regulation (sleeping with a partner versus alone), and familiarity of the room all provide cues that the nervous system evaluates. For how trauma locks neuroception in a persistent danger-detection mode, see trauma, hypervigilance, and insomnia.
What Does the Evidence Say About Autonomic Hyperarousal and Insomnia?
Riemann et al. (2010) published the landmark review synthesizing evidence across five domains — autonomic, neuroendocrine, neuroimmunological, electrophysiological, and neuroimaging — all pointing to elevated arousal as the unifying mechanism of insomnia. Autonomic evidence includes elevated resting heart rate and reduced heart rate variability. Neuroendocrine evidence demonstrates higher cortisol secretion and elevated plasma catecholamines. Neuroimaging data reveal elevated whole-brain metabolic activation during sleep in insomnia.
Wix-Ramos et al. (2024) compared 32 people with chronic insomnia to 19 healthy controls using simultaneous overnight polysomnography and wearable monitoring. Insomnia participants showed elevated heart rate during both sleep and wakefulness. Electrodermal activity — a direct measure of sympathetic sweating — was higher in the insomnia group throughout the sleep period. The wearable data showed that the autonomic state in chronic insomnia resembles sustained sympathetic activation rather than the parasympathetic dominance that characterizes normal sleep.
Zhu and Xue (2025) mapped the structural relationships between hyperarousal subtypes and insomnia in 1,209 participants. Pre-sleep cognitive arousal emerged as the primary bridge connecting the hyperarousal domain to insomnia.
The self-reinforcing cycle. Zhang et al. (2025) conducted a meta-analysis of 11 RCTs (549 participants) and found that sleep deprivation produced a decrease in RMSSD — the primary parasympathetic vagal tone marker — and an increase in the LF/HF ratio, reflecting sympathetic predominance. Poor sleep produces sympathetic dominance, and sympathetic dominance produces poor sleep.
In polyvagal terms: the hyperarousal literature documents what happens (sympathetic overdrive preventing sleep). Polyvagal theory adds a framework for the why — neuroception of danger preventing the state transition that sleep onset requires.
Autonomic state is one factor affecting your sleep. Metabolic disruptions, inflammatory processes, hormonal changes, or circadian misalignment may also be contributing. When multiple causes overlap, identifying which ones are active is a useful next step.
Find out which causes might be driving your 3am wakeups →
Which Nervous System Regulation Exercises Help Each Polyvagal State Before Bed?
One reason standard “relaxation techniques” can produce the opposite of the intended effect: a person in dorsal vagal shutdown who does deep breathing may go deeper into collapse rather than transitioning to the ventral vagal state. Matching the exercise to the state matters.
How to identify which state is dominant before bed:
- Sympathetic activation: Racing or elevated heart rate, inability to stop thinking, jaw clenching, muscle tension, feeling wired despite exhaustion.
- Dorsal vagal shutdown: Fatigue that is not restful, feeling numb or disconnected, heaviness in the body, slowed thinking. You may have slept ten or more hours and still feel unrested.
For sympathetic activation — moving toward ventral vagal:
- Extended exhale breathing. Inhale for four counts, exhale for six to eight counts. The extended exhale activates vagal pathways, promoting parasympathetic dominance. For breathing technique evidence, see TLV’s vagus nerve stimulation article.
- Cold water face immersion. Submerging the face in cold water for 15-30 seconds activates the dive reflex — a rapid parasympathetic response mediated by the trigeminal-vagal pathway.
- Humming or vocal toning. The vagus nerve innervates the pharynx and larynx. Sustained humming stimulates these vagal branches, promoting parasympathetic engagement.
For dorsal vagal shutdown — moving toward ventral vagal:
The goal here is gentle activation, not more relaxation. The dorsal vagal state is already immobilized — adding stillness deepens the shutdown.
- Gentle orienting. Slowly look around the room. Name five things you can see. This activates the social engagement circuitry — cranial nerves involved in vision, hearing, and facial expression — coordinated by the ventral vagal complex.
- Bilateral stimulation (butterfly tap). Alternately tapping the left and right sides of the chest can facilitate a state transition without demanding high energy.
- Gentle movement. Rocking, swaying, or slow side-to-side head movement provides rhythmic input that is accessible from dorsal vagal shutdown.
Frequently Asked Questions
Is Polyvagal Theory Scientifically Validated?
Polyvagal theory is widely used in trauma-informed care and somatic therapy, but it has drawn scientific criticism that is worth taking seriously. Grossman (2023) and Taylor and colleagues have published detailed critiques arguing that the phylogenetic hierarchy Porges describes — dorsal vagal developing first, then sympathetic, then ventral vagal — is not consistently supported by comparative vertebrate anatomy. Their objections are specific: the distinction between unmyelinated dorsal vagal fibers and myelinated ventral vagal fibers does not map neatly onto all vertebrate lineages, and some fish and reptiles show vagal complexity that the hierarchical model does not account for. These are substantive anatomical challenges, not peripheral objections. Porges (2025) addresses several of these points by reframing the debate and citing additional neurophysiological data, though the debate remains unresolved in the comparative anatomy literature.
What is not in dispute: the vagus nerve has functionally distinct branches, autonomic state affects behavior and physiological capacity, and the nervous system evaluates safety below conscious awareness. The disagreement centers on whether the evolutionary sequence is as orderly as the original theory proposed. The empirical predictions the theory generates — such as the safety-HRV link demonstrated by Schwerdtfeger et al. (2025) — continue to receive support.
For sleep, identifying which autonomic state is dominant and matching the approach to that state has practical utility regardless of whether every evolutionary claim in the original formulation is correct.
How Is Polyvagal Insomnia Different From Regular Insomnia?
The distinction is in the lens, not the condition. What polyvagal theory adds is a reason for the state (neuroception of danger), a name for the opposite extreme (dorsal vagal shutdown producing unrefreshing hypersomnia), and a principle for selecting exercises — match the exercise to the autonomic state, not to “insomnia” as a broad category.
Can You Measure Which Polyvagal State You Are In?
Wearable devices that track overnight HRV provide a reasonable approximation. A pattern of low HRV with elevated resting heart rate during sleep suggests sympathetic dominance. Improving HRV trends — particularly in the RMSSD metric — suggest increasing ventral vagal influence. TLV covers how to interpret overnight HRV data in the HRV and vagal tone article.
Related Reading
- Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It — full overview of autonomic causes including vagal tone, GABA, cortisol, and hyperarousal
- Does Lion’s Mane Affect Your Sleep? Why It Helps Some People and Keeps Others Awake — lion’s mane’s dual mechanism: neurotrophic activity, cholinergic activity, and sleep architecture
- Why Do Cholinergic Supplements Give You Vivid Dreams? — the acetylcholine-REM pathway behind supplement-related vivid dreams
- Which Choline Supplement Is Least Likely to Disrupt Your Sleep? A Form-by-Form Comparison — how different choline forms reach the brain and affect sleep
- How Do You Take Nootropics Without Disrupting Your Sleep? A Neuroscience-Based Approach — timing and stacking guidance for preserving sleep while using nootropics
- Why Can You Not Sleep After COVID? What Happened to Your Vagus Nerve — how post-viral autonomic disruption can affect sleep continuity
- Why Does Your Heart Race at Night After COVID? What the Vagus Nerve Has to Do With It — why post-COVID autonomic instability can show up as nighttime tachycardia
- Can POTS and Dysautonomia Cause Insomnia? Why Your Autonomic Nervous System Will Not Let You Rest — how orthostatic intolerance and autonomic instability can fragment sleep
- Can a Viral Infection Cause Chronic Insomnia? — how viral infections can disrupt autonomic regulation and sleep
- Why Does Trauma Cause Insomnia? How Your Nervous System Stays on Guard at Night — how trauma can keep sleep shaped by hyperarousal and vigilance
- Why Are You Sleeping 12 Hours and Still Exhausted? The Dorsal Vagal Shutdown Explained — the dorsal vagal pattern behind long sleep that still feels unrefreshing
- Which Probiotic Strains Improve Sleep? What the Vagus Nerve Evidence Shows — which probiotic strains have human sleep evidence and how the vagus nerve may be involved
- How Do Probiotics Reach Your Brain? The Vagotomy Evidence — what vagotomy studies show about gut-brain communication
- Does Poor Sleep Damage Your Gut Microbiome? — the relationship between sleep fragmentation, microbiome disruption, and inflammatory load
- How Long Do Probiotics Take to Improve Sleep? What the Research Shows Week by Week — what probiotic sleep studies suggest about timing across weeks
References
Dressle, R. J., & Riemann, D. (2023). Hyperarousal in insomnia disorder: Current evidence and potential mechanisms. Journal of Sleep Research, 32(6), e13928. https://pubmed.ncbi.nlm.nih.gov/37183177/
Grossman, P. (2023). Fundamental challenges and likely refutations of the five basic premises of the polyvagal theory. Biological Psychology, 180, 108589. https://pubmed.ncbi.nlm.nih.gov/37230290/
Morton, L., Cogan, N., Kolacz, J., Calderwood, C., Nikolic, M., Bacon, T., Pathe, E., Williams, D., & Porges, S. W. (2024). A new measure of feeling safe: Developing psychometric properties of the Neuroception of Psychological Safety Scale (NPSS). Psychological Trauma: Theory, Research, Practice and Policy, 16(4), 701-708. https://pubmed.ncbi.nlm.nih.gov/35849369/
Porges, S. W. (2022). Polyvagal Theory: A Science of Safety. Frontiers in Integrative Neuroscience, 16, 871227. https://pubmed.ncbi.nlm.nih.gov/35645742/
Porges, S. W. (2025). Polyvagal theory: A journey from physiological observation to neural innervation and clinical insight. Frontiers in Behavioral Neuroscience, 19, 1659083. https://pubmed.ncbi.nlm.nih.gov/41035859/
Porges, S. W., Doussard-Roosevelt, J. A., Stifter, C. A., McClenny, B. D., & Riniolo, T. C. (1999). Sleep state and vagal regulation of heart period patterns in the human newborn: An extension of the polyvagal theory. Psychophysiology, 36(1), 14-21. https://pubmed.ncbi.nlm.nih.gov/10098376/
Riemann, D., Spiegelhalder, K., Feige, B., Voderholzer, U., Berger, M., Perlis, M., & Nissen, C. (2010). The hyperarousal model of insomnia: A review of the concept and its evidence. Sleep Medicine Reviews, 14(1), 19-31. https://pubmed.ncbi.nlm.nih.gov/19481481/
Schwerdtfeger, A. R., Wekenborg, M., Tatschl, J. M., & Rominger, C. (2025). Neuroception of safety is associated with elevated heart rate variability in the laboratory and more frequent heart rate variability increases in everyday life. Annals of Behavioral Medicine, 59(1). https://pubmed.ncbi.nlm.nih.gov/40165438/
Taylor, E. W., Wang, T., & Leite, C. A. C. (2022). An overview of the phylogeny of cardiorespiratory control in vertebrates with some reflections on the ‘Polyvagal Theory’. Biological Psychology, 172, 108382. https://pubmed.ncbi.nlm.nih.gov/35777519/
Wix-Ramos, R., Galvez-Goicuria, J., Verona-Almeida, M., Ayala, J. L., Lopez-Vinas, L., Rocio-Martin, E., Luque-Cardenas, C., Quintas, S., Gago-Veiga, A., & Pagan, J. (2024). Monitoring differences in the function of the autonomic nervous system in patients with chronic insomnia using a wearable device. Sleep Medicine, 115, 122-130. https://pubmed.ncbi.nlm.nih.gov/38359591/
Zhang, S., Niu, X., Ma, J., Wei, X., Zhang, J., & Du, W. (2025). Effects of sleep deprivation on heart rate variability: A systematic review and meta-analysis. Frontiers in Neurology, 16, 1556784. https://pubmed.ncbi.nlm.nih.gov/40895095/
Zhu, K., & Xue, S. (2025). Identifying the association of hyperarousal and insomnia symptoms: A network perspective. Journal of Psychiatric Research, 189, 216-222. https://pubmed.ncbi.nlm.nih.gov/40527108/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 12 references cited
