Waking at 3am often has metabolic timing. Between 2 and 4am, three biological processes converge at their vulnerable point: blood glucose is at its lowest, the circadian cortisol process is primed for its sharpest rise, and cellular energy reserves from the first sleep cycles are depleted. Repeated 3am waking is not a sleep pattern alone but a metabolic cue — relevant to anyone concerned with brain health, metabolic longevity, and sustained energy.
This article covers the specific 3am timing mechanism — what fires first, why the 2-4am window is uniquely vulnerable, and what the evidence says about prevention. For the complete metabolic cause overview, see Metabolic Sleep Disruption. Mitochondrial energy status is one factor that determines whether your body absorbs these fluctuations unnoticedly or whether they produce a full arousal.
Why Is the 2-4am Window Metabolically Vulnerable?
When does the cortisol clock peak during sleep?
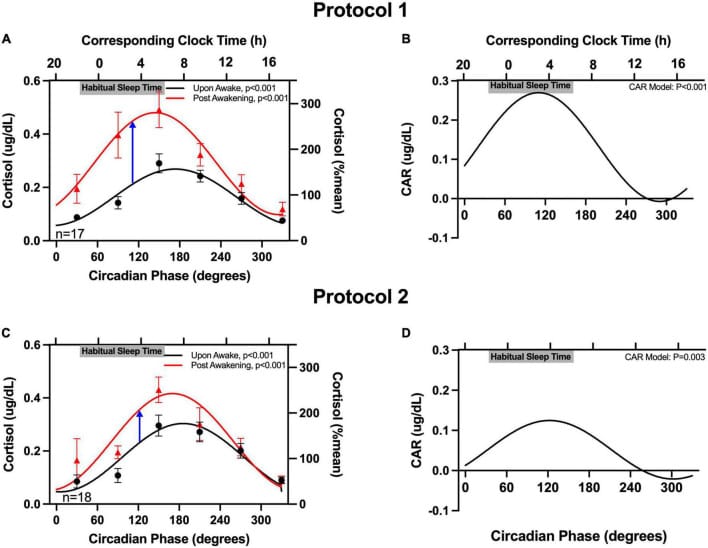
A forced-desynchrony study in 34 adults used two approaches — 5-hour-20-minute and 18-hour sleep/wake cycles — to distribute waking events across every circadian phase. Both approaches produced similar cortisol rhythms, with the cortisol awakening response peaking at approximately 3:40-3:45am biological time. No detectable cortisol awakening response occurred during afternoon circadian phases, even when subjects woke from sleep at those times (Bowles et al., 2022). The cortisol surge at 3am is programmed by the circadian clock itself, not triggered by the act of waking.
When does the brain’s energy recharge run out?
In rat studies, adenosine triphosphate concentrations surged in wake-active brain regions during the initial hours of sleep, correlating directly with non-rapid-eye-movement delta activity. This surge depended on sleep, not time of day — preventing sleep prevented the energy recharge (Dworak et al., 2010). By the end of the sleep period, phosphorylated AMP-activated protein kinase levels showed a tendency to return toward waking levels — consistent with the cell’s energy sensor responding to lower adenosine triphosphate reserves as sleep progressed (Dworak et al., 2010). The brain regions active during wakefulness were the first to run low. While these dynamics have been well-characterized in animal models, direct measurement of adenosine triphosphate changes across human sleep cycles remains technically limited.
Why is late-sleep glucose sensitivity heightened?
In a controlled study of 16 healthy participants, plasma glucose was clamped to 2.2 mmol/L during either early sleep or late sleep (3.5 hours after sleep onset). During late sleep, 100% of subjects awakened from hypoglycemia compared to 63% during early sleep. But the counterregulatory hormonal response — epinephrine, norepinephrine, adrenocorticotropic hormone, cortisol, and growth hormone — was simultaneously weaker during late-sleep hypoglycemia (Jauch-Chara et al., 2007). You are more likely to wake from a glucose drop at 3am and less equipped to hormonally correct it.
When circadian cortisol priming, energy depletion, and glucose vulnerability converge at the same 2-4am window, the margin for unnoticed recovery disappears.
What Fires First at 3am — Blood Sugar Drop, Cortisol Spike, or Both?
Is cortisol the first body-wide response to sleep disruption?
A temporal-cascade study in mice measured hormonal and immune responses at 1, 2, 6, 12, and 24 hours after acute sleep fragmentation. Serum corticosterone (the rodent equivalent of cortisol) was elevated within 1 hour — the first measurable body-wide event. Interleukin-6 appeared later at 6-24 hours. Heart tissue showed pro-inflammatory gene expression at 1 hour, while hypothalamic expression was paradoxically suppressed before becoming elevated at 6 hours (Nguyen et al., 2023). These animal data suggest that hypothalamic-pituitary-adrenal axis activation may be the initiating body-wide response, with inflammation following hours later. Whether the same temporal sequence holds in humans has not been directly tested.
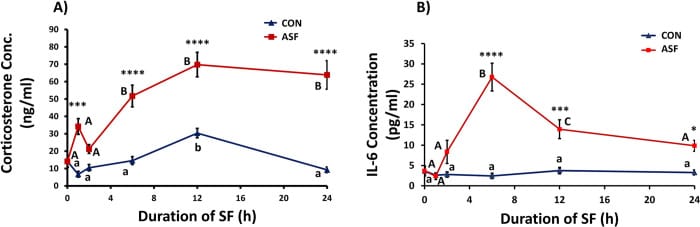
Does sleep fragmentation raise glucose and cortisol at the same time?
A randomized crossover trial in 31 individuals with obstructive sleep apnea compared nights with CPAP use versus CPAP withdrawal. When sleep fragmentation recurred, nocturnal free fatty acids, blood glucose, and cortisol all rose dynamically in patterns paralleling arousal events. participants with comorbid diabetes showed greater glucose elevations than normoglycemic participants (Chopra et al., 2017). The mechanism: simultaneous sympathetic nervous activity and hypothalamic-pituitary-adrenal axis activation from repetitive arousals.
Is the cortisol rise already happening before you wake?
A review of in vivo microdialysis data from 201 healthy individuals showed no statistically significant cortisol acceleration upon awakening compared to the preceding hour of sleep (Velazquez Sanchez & Dalley, 2025). The cortisol increase measured after waking is a continuation of the underlying circadian rise, not an awakening-triggered event. The glucose drop may determine whether you wake into the cortisol surge that is already underway.
How do glucose-sensing channels connect blood sugar to arousal?
In mouse studies, adenosine-triphosphate-sensitive potassium channels sense the intracellular adenosine-triphosphate-to-adenosine-diphosphate ratio and couple metabolic state to neuronal membrane potential. Under normal conditions, these channels modulate glycolytic flux, influencing cortical excitability and creating metabolic conditions permissive for sleep. Elevated interstitial lactate marks wakefulness; decreased interstitial lactate is required for sleep initiation (Constantino et al., 2025). When glucose metabolism is disrupted, these channels not work to suppress cortical excitability, and arousal persists. Whether this mechanism works identically in human neurons has not been showed, but the channel proteins are conserved across mammals.
Does the Dawn Phenomenon Cause 3am Waking in Non-Diabetics?
Are healthy young adults protected from glucose-driven wakeups?
A prospective study equipped 119 healthy university students with continuous glucose monitors and Oura Ring wearables for up to 14 days. Nocturnal sleep quality did not influence following-day glucose profiles, and experimental sleep restriction did not produce significant glucose or insulin changes (Ng et al., 2025). This is the baseline of metabolic health. When you are age 35 and older, perimenopausal, prediabetic, or carrying inflammatory load — this glucose resilience is what has reduced.
Does metabolic vulnerability amplify the 3am cascade?
The same sleep disruption produces different metabolic consequences depending on baseline metabolic health. Obstructive sleep apnea participants with comorbid diabetes showed greater nocturnal glucose elevations during sleep fragmentation than normoglycemic participants experiencing identical fragmentation (Chopra et al., 2017). The 3am cascade is dose-dependent on insulin sensitivity.
Why is late-sleep counterregulation weaker?
During late sleep, the hormonal defense against falling glucose is distinctly weaker — epinephrine, cortisol, adrenocorticotropic hormone, and growth hormone responses are all blunted compared to early-sleep hypoglycemia (Jauch-Chara et al., 2007). In someone with early insulin resistance, the initial glucose drop is deeper and the hormonal correction is both delayed and overshooting.
How do energy-pathway activity pathways depend on glucose stability?
Both adenosine-dependent and P2-receptor-dependent adenosine triphosphate pathways govern the sleep-wake switch. Adenosine triphosphate hydrolysis to adenosine drives classical sleep pressure, while direct P2 receptor pathway activity provides brain-region-specific arousal modulation (Gao et al., 2024). When glucose metabolism is disrupted, both pathways are affected. The mitochondrial sleep switch depends on stable substrate delivery, and glucose instability undermines that delivery at the cellular level.
Can Eating Before Bed Prevent the 3am Cascade?
How does a pre-bed meal prevent glucose-triggered arousal?
If late-sleep hypoglycemia triggers 100% awakening (Jauch-Chara et al., 2007), preventing the glucose drop below the arousal threshold is the direct approach. Slow-digesting protein and fat — not carbohydrates, which can produce a ROS-related hypoglycemic rebound — extend the glucose runway through the vulnerable window. Maintaining adequate glucose substrate keeps adenosine-triphosphate-sensitive potassium channels in a sleep-permissive configuration (Constantino et al., 2025).
How does continuous glucose monitoring identify the driver?
Continuous glucose monitoring combined with sleep tracking allows individuals to correlate wakeup timestamps with glucose traces. The informative pattern to look for: a glucose nadir within 30-60 minutes before the recorded wakeup event suggests the blood sugar arm is active. Flat glucose through a 3am wakeup suggests cortisol rhythm or mitochondrial drivers instead (Ng et al., 2025). Two weeks of concurrent monitoring gives sufficient data to identify the pattern.
When is food timing not enough?
If the 3am wakeup is driven by mitochondrial electron-flux imbalance in sleep-control neurons, food timing will not address the pattern. A 2025 Nature study in Drosophila identified mitochondrial adenosine triphosphate dynamics as a molecular driver of sleep pressure (Sarnataro et al., 2025). Whether these findings translate directly to humans is not yet established, but the mitochondrial pathways involved are conserved across species. Mitochondrial support approaches are covered in the supplement evidence review, and the wired-but-tired pattern addresses cortisol rhythm inversion specifically.
The 3am wakeup rarely has a single cause. Blood sugar instability, circadian cortisol programming, and mitochondrial energy deficits each create their own version of early-morning arousal — and in many people, more than one of these processes is involved. Your wakeup pattern might be primarily glucose-driven, primarily cortisol-driven, or a overlapping combination that changes depending on what you ate, how stressed you are, and how much sleep debt you are carrying.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
What Hormone Surges at 3am?
When blood sugar drops below the counterregulatory threshold during the 3am window, additional hormones fire: epinephrine, norepinephrine, glucagon, and growth hormone all activate to raise glucose. But cortisol has the longest duration and the strongest arousal-maintaining effect and can sustain alertness long enough that returning to sleep after a 3am wakeup is often difficult (Bowles et al., 2022; Velazquez Sanchez & Dalley, 2025).
Can Low Blood Sugar Wake You Up at Night?
The paradox: the hormonal defense is simultaneously weaker during late sleep — epinephrine, cortisol, and adrenocorticotropic hormone responses are all blunted compared to early-sleep hypoglycemia (Jauch-Chara et al., 2007). A modest glucose dip that would be unnoticedly corrected at midnight produces a fully conscious wakeup at 3am, and the weakened counterregulatory response means glucose recovers more slowly, extending the arousal window.
Can a Continuous Glucose Monitor Show Why You Wake at 3am?
Both prescription continuous glucose monitors (Libre, Dexcom) and direct-to-consumer options (Levels, Nutrisense) provide the overnight glucose data needed. Two weeks of concurrent continuous glucose monitoring and sleep tracking gives sufficient data to identify whether the glucose nadir precedes the arousal (Ng et al., 2025).
Is a 3am Wakeup a Sign of Insulin Resistance or Prediabetes?
Continuous glucose monitoring distinguishes metabolic-driver wakeups from cortisol-only wakeups. A glucose dip preceding 3am wakeups on nights warrants metabolic testing — fasting insulin, hemoglobin A1c, oral glucose tolerance test — to evaluate subclinical insulin resistance. Stable glucose through a 3am wakeup points to the cortisol rhythm arm or mitochondrial energy depletion instead (Chopra et al., 2017).
Related Reading
- Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep — the pillar article for metabolic sleep disruption, covering glucose regulation, cortisol rhythm, mitochondrial stress, NAD+ reduction, and fat metabolism
- Why Are You Exhausted But Can’t Sleep? Is It a Mitochondrial Energy Paradox? — mitochondrial fatigue, unrefreshing sleep, ATP production, and why rest may not restore cellular energy
- Do Your Mitochondria Control When You Sleep? and Why Does It Break Down After 40? — ROS sleep pressure, deep sleep reduction after 40, mitochondrial DNA aging, and sleep timing
- Can CoQ10 and PQQ Supplements Improve Your Sleep? — CoQ10, PQQ, magnesium, supplement timing, and the evidence for mitochondrial sleep support
- Why Are You Wired but Tired, and What Role Do Mitochondria Play? — hyperarousal, cortisol rhythm changes, stress physiology, and mitochondrial power-down reduction
- How Does Exercise Rebuild Your Mitochondria and Fix Your Sleep? — exercise type, timing, mitochondrial biogenesis, PGC-1?, and sleep architecture
- What Is the Measured Cellular Impact of Sleep Fragmentation? — fragmented sleep, oxidative stress, vascular injury, inflammation, and cellular aging impact
References
Bowles, N. P., Thosar, S. S., Butler, M. P., Clemons, N. A., Robinson, L. D., Ordaz, O. H., Herzig, M. X., McHill, A. W., Rice, S. P. M., Emens, J., & Shea, S. A. (2022). The circadian process modulates the cortisol awakening response in humans. Frontiers in Neuroscience, 16, 995452. https://pubmed.ncbi.nlm.nih.gov/36408390/
Chopra, S., Rathore, A., Younas, H., Pham, L. V., Gu, C., Beselman, A., Kim, I. Y., Wolfe, R. R., Perin, J., Polotsky, V. Y., & Jun, J. C. (2017). Obstructive sleep apnea dynamically increases nocturnal plasma free fatty acids, glucose, and cortisol during sleep. The Journal of Clinical Endocrinology and Metabolism, 102(9), 3172-3181. https://pubmed.ncbi.nlm.nih.gov/28595341/
Constantino, N. J., Carroll, C. M., Williams, H. C., Vekaria, H. J., Yuede, C. M., Saito, K., Sheehan, P. W., Snipes, J. A., Raichle, M. E., Musiek, E. S., Sullivan, P. G., Morganti, J. M., Johnson, L. A., & Macauley, S. L. (2025). ATP-sensitive potassium channels alter glycolytic flux to modulate cortical activity and sleep. Proceedings of the National Academy of Sciences of the United States of America, 122(8), e2416578122. https://pubmed.ncbi.nlm.nih.gov/39964713/
Dworak, M., McCarley, R. W., Kim, T., Kalinchuk, A. V., & Basheer, R. (2010). Sleep and brain energy levels: ATP changes during sleep. The Journal of Neuroscience, 30(26), 9007-9016. https://pubmed.ncbi.nlm.nih.gov/20592221/
Gao, Z., Guan, J., Yin, S., & Liu, F. (2024). The role of ATP in sleep-wake regulation: In adenosine-dependent and -independent manner. Sleep Medicine, 119, 147-154. https://pubmed.ncbi.nlm.nih.gov/38678758/
Jauch-Chara, K., Hallschmid, M., Gais, S., Oltmanns, K. M., Peters, A., Born, J., & Schultes, B. (2007). Awakening and counterregulatory response to hypoglycemia during early and late sleep. Diabetes, 56(7), 1938-1942. https://pubmed.ncbi.nlm.nih.gov/17400929/
Ng, A. S. C., Tai, E. S., & Chee, M. W. L. (2025). Effects of night-to-night variations in objectively measured sleep on blood glucose in healthy university students. Sleep, 48(2), zsae224. https://pubmed.ncbi.nlm.nih.gov/39325824/
Nguyen, V. T., Fields, C. J., & Ashley, N. T. (2023). Temporal dynamics of pro-inflammatory cytokines and serum corticosterone following acute sleep fragmentation in male mice. PLoS ONE, 18(12), e0288889. https://pubmed.ncbi.nlm.nih.gov/38096187/
Sarnataro, R., Velasco, C. D., Monaco, N., Kempf, A., & Miesenboeck, G. (2025). Mitochondrial origins of the pressure to sleep. Nature, 645(8081), 722-728. https://pubmed.ncbi.nlm.nih.gov/40670797/
Velazquez Sanchez, C., & Dalley, J. W. (2025). The cortisol awakening response: Fact or fiction? Brain and Neuroscience Advances, 9, 23982128251327712. https://pubmed.ncbi.nlm.nih.gov/40297522/
Written by Kat Fu, M.S., M.S.? Last reviewed: May 2026? 10 references cited
