Caffeine delays your circadian clock by approximately 40 minutes through the adenosine-cAMP pathway. When caffeine blocks adenosine A1 and A2A receptors, it disrupts the input that normally feeds into the molecular clock’s core feedback loop — the Per1 and Per2 genes. This is a circadian phase delay, not wakefulness alone. The effect size varies based on your CYP1A2 genotype (how fast you metabolize caffeine) and ADORA2A genotype (how sensitive your receptors are to the caffeine that remains).
Caffeine keeps you awake. But it also delays your internal clock — a circadian phase delay that persists even after the stimulant effect wears off. This is why a late-afternoon coffee can still be disrupting your sleep architecture days later, even when you fall asleep at your normal time.
This article covers the molecular mechanism through which caffeine enters the circadian clock, the two genetic factors that determine why some people are more affected than others, pharmacokinetic variability across age and sex, and the dose-timing evidence that contradicts the blanket “stop coffee by 2pm” advice.
Caffeine is one of several factors that can disrupt circadian timing. The full circadian disruption overview covers the broader picture.
How Does Caffeine Enter the Molecular Clock?
Caffeine blocks adenosine A1 and A2A receptors. Normally, adenosine builds during wakefulness and routes sleep-pressure information into the molecular clock via calcium-ERK and CREB/CRTC1 pathways, which regulate the clock genes Per1 and Per2. Caffeine disrupts this input, delaying the molecular feedback loop that governs circadian timing. In a controlled inpatient study, a double espresso consumed 3 hours before habitual bedtime delayed dim-light melatonin onset by approximately 40 minutes.
Adenosine accumulates in your brain across every hour you spend awake. This buildup is the biological basis of sleep pressure — the longer you have been awake, the more adenosine you have, and the stronger the drive to sleep. But adenosine does more than make you drowsy. It feeds timing information into the molecular clock itself.
Jagannath et al. (2021) mapped this pathway in detail. Adenosine activates A1 and A2A receptors, which trigger calcium-ERK-AP-1 and CREB/CRTC1-CRE molecular cascades. These cascades converge on the same downstream targets — the clock genes Per1 and Per2 — that light uses to entrain your circadian rhythm. In other words, sleep pressure and light exposure regulate circadian timing through overlapping molecular machinery.
Caffeine is a competitive antagonist at both A1 and A2A receptors. When caffeine occupies these receptors, adenosine cannot deliver its timing input to the clock. The result is a delayed phase in the core transcription-translation feedback loop that sets your circadian rhythm.
Burke et al. (2015) demonstrated this in a controlled inpatient study. Participants received a double espresso (equivalent to approximately 200 mg caffeine) 3 hours before their habitual bedtime under dim-light conditions. Caffeine delayed dim-light melatonin onset (DLMO) by approximately 40 minutes — roughly half the magnitude of a 3-hour bright-light exposure administered in the same study. In vitro, the same group showed that caffeine lengthened the circadian period of human osteosarcoma cells in an adenosine-receptor- and cAMP-dependent manner, confirming the mechanism at the cellular level.
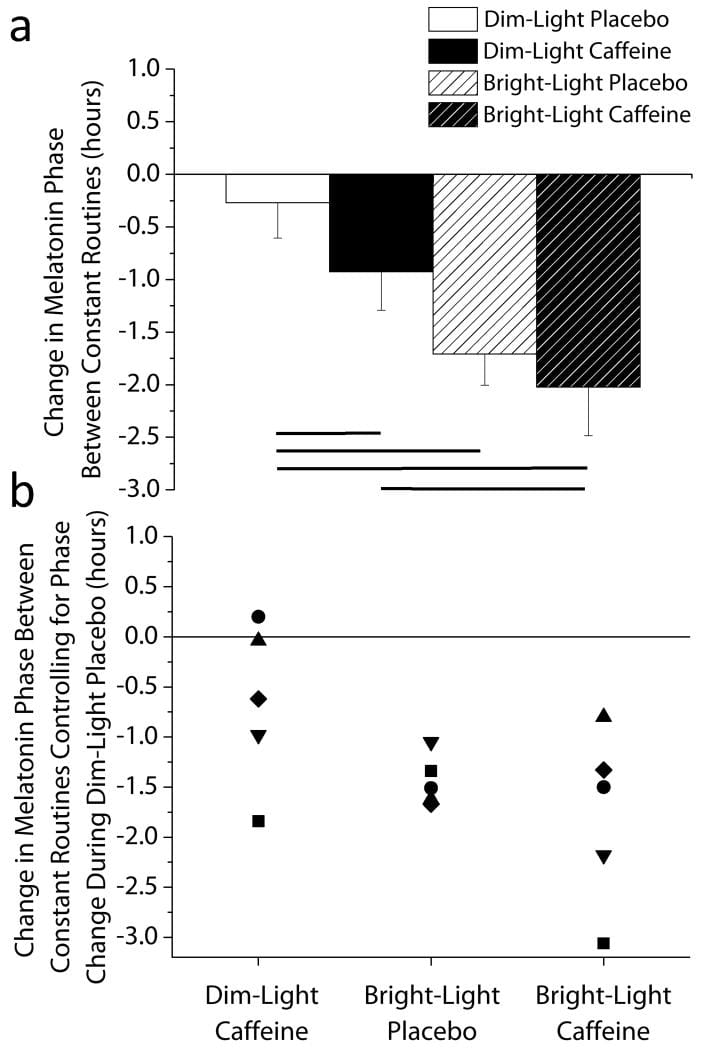
Reichert et al. (2022) further clarified that caffeine acts through two parallel mechanisms: it blocks homeostatic sleep pressure (the drowsiness pathway) and disrupts circadian gating (the clock-timing pathway) at the same time. These are not the same effect. You can feel alert — caffeine doing its job on sleep pressure — while your clock is being delayed without any perceptible cue.
Why Can Some People Drink Coffee at 4pm and Sleep Fine?
Two genes explain the majority of the variation. CYP1A2 controls how fast your liver metabolizes caffeine — fast metabolizers eliminate it more quickly, while slow metabolizers can take several-fold longer. ADORA2A determines how sensitive your adenosine receptors are to the caffeine that remains. A genetic variant (rs5751876) makes some people less sensitive to caffeine’s sleep-disrupting effects regardless of how long it stays in their body.
The question of individual caffeine sensitivity comes down to two independent genetic factors. Each one works through a different mechanism, and having one does not predict the other.
Factor one: metabolism speed (CYP1A2). Low et al. (2024) conducted a review of 26 studies covering 1.85 million individuals. CYP1A2 appeared as a determinant in 15 of those studies, with each allele variant accounting for 3 to 32 percent of the variation in caffeine intake. Caffeine half-life varies up to 5- to 6-fold between individuals (Grzegorzewski et al., 2022), with CYP1A2 genotype as a major source of variation. For slow metabolizers, a 3pm coffee can remain pharmacologically active at midnight.
Factor two: receptor sensitivity (ADORA2A). Retey et al. (2007) identified a polymorphism — rs5751876 (c.1083T>C) in the ADORA2A gene — that determines how closely caffeine-induced changes in sleep architecture resemble insomnia. Carriers of one genotype showed minimal sleep disruption from caffeine, while carriers of the other showed measurable reductions in sleep quality under the same conditions.
These two factors are biologically independent. A person could be a slow metabolizer (caffeine stays in the body for hours) but carry a receptor genotype that confers lower sensitivity to whatever caffeine is present. That person would experience less sleep disruption despite high circulating caffeine. The reverse — fast metabolism but high receptor sensitivity — would produce sleep disruption from even moderate doses consumed relatively early.
This is why population-level advice about caffeine cutoff times falls apart at the individual level. Two people can drink the same coffee at the same time and experience opposite effects on sleep.
How Long Does Caffeine Stay in Your Body — and Does That Change With Age?
Caffeine half-life averages 2.5 to 5 hours after acute consumption but varies up to 5- to 6-fold between individuals. Smoking shortens the half-life by approximately 50 percent. Oral contraceptives nearly double it. CYP1A2 genotype accounts for a major source of inter-individual variation. Separately, caffeine-induced insomnia increases with age in both men and women — independent of dose — suggesting that aging degrades the buffering capacity that protects younger adults from caffeine’s circadian effects.
Grzegorzewski et al. (2022) analyzed 141 pharmacokinetic publications and found that caffeine half-life ranges from 2.5 to 5 hours in the majority of adults — but this average conceals a 5- to 6-fold range of individual variation. Several factors modify this range in predictable directions.
Smoking induces CYP1A2 activity, cutting caffeine half-life by roughly 50 percent. Oral contraceptives inhibit CYP1A2, nearly doubling the half-life. Pregnancy extends caffeine half-life to as long as 15 hours in the third trimester. Drug interactions — particularly with fluvoxamine and fluoroquinolone antibiotics — can increase half-life by several-fold. CYP1A2 genotype remains a major source of variation when these modifiers are controlled for.
A separate question is whether aging changes caffeine’s effect on sleep independent of pharmacokinetics. Frozi et al. (2018) found that it does. In a population study, caffeine-induced insomnia increased with age in both men and women. This relationship was independent of dose — older adults were not consuming more caffeine. Women showed a higher raw proportion of caffeine-induced insomnia (18.3% vs 17.0%), though this difference did not remain after adjusting for covariates.
The implication is that even if your caffeine metabolism stays the same as you age, your brain’s ability to buffer caffeine’s circadian effects diminishes. A dose that caused no sleep disruption at 30 may produce measurable disruption at 50 — because the downstream circadian machinery has less tolerance for the interference.
What Does the Evidence Say About When to Stop Drinking Coffee?
Cutoff timing depends on dose, not the clock. A crossover trial found that 100 milligrams of caffeine (roughly one standard coffee) had no measurable effect on objective sleep at any timing — including 4 hours before bed. A 400-milligram dose (roughly a large or pre-workout coffee) altered sleep architecture even when consumed 12 hours before bed, with reductions in total sleep time and sleep efficiency reaching significance at shorter intervals. The “stop coffee by 2pm” rule is an oversimplification that works for high-dose consumers but penalizes moderate ones unnecessarily.
Gardiner et al. (2025) designed the best-controlled test of this question to date: a placebo-controlled, double-blind, crossover trial with 7 conditions. Participants received either placebo, 100 mg caffeine, or 400 mg caffeine at 12, 8, or 4 hours before bedtime, with a 48-hour washout between each condition.
The results were unambiguous on dose. At 100 mg — the equivalent of one standard cup of brewed coffee — there was no measurable effect on total sleep time, sleep efficiency, sleep onset latency, or slow-wave sleep at any timing point. Even 4 hours before bed, 100 mg produced no detectable disruption.
At 400 mg, effects appeared at all timing points. At 12 hours before bed, caffeine delayed sleep initiation and shifted sleep architecture toward lighter stages — increasing the proportion of N1 and N2 sleep and reducing N3 (deep sleep) duration by approximately 20 minutes. Total sleep time and sleep efficiency were not significantly affected at the 12-hour interval. At 8 hours, sleep efficiency dropped and sleep fragmentation increased. At 4 hours, total sleep time, sleep efficiency, sleep onset latency, and deep sleep were all significantly impaired — with total sleep time reduced by approximately 50 minutes.
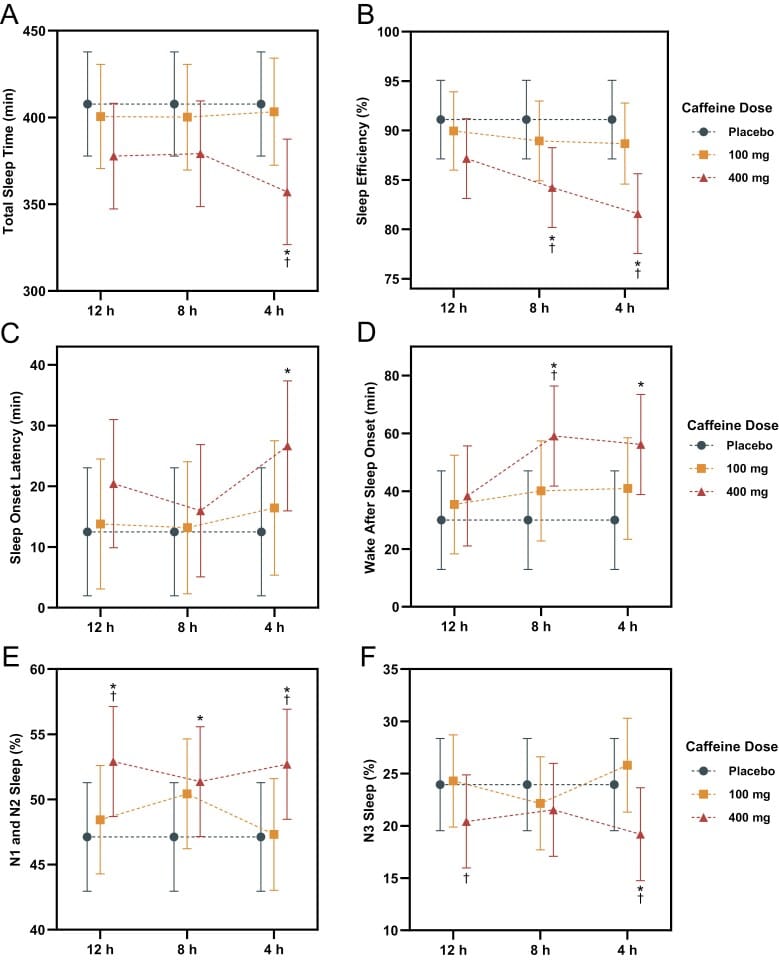
Chang, Cheng, and Cheng (2025) added an age dimension through a meta-analysis of 22 controlled crossover trials. Younger adults appeared more sensitive to caffeine’s effects on multiple sleep parameters, including slow-wave sleep. The relationship between age and specific sleep outcomes may vary, though the authors noted that linear relationships between continuous age and dose variables were not statistically significant — suggesting the pattern is more complex than a simple gradient.
The practical takeaway is that cutoff timing without dose context is incomplete advice. One cup of coffee in the afternoon is a different pharmacological event than a large cold brew or pre-workout supplement containing 300 to 400 mg.
Many people have more than one cause contributing to their sleep disruption. Circadian timing delays from caffeine may compound with metabolic, hormonal, inflammatory, or autonomic factors. Identifying which causes might be involved is a useful next step.
Find out which causes might be driving your 3 AM wakeups →
Frequently Asked Questions
Does Caffeine Suppress Melatonin Production?
Caffeine does not suppress melatonin production directly. It delays the timing of dim-light melatonin onset — meaning melatonin still rises to normal levels but starts later. The downstream effect is that your biological “ready for sleep” cue arrives approximately 40 minutes later than it would otherwise.
The distinction between suppression and delay matters. Bright light at night suppresses melatonin — it reduces the total amount produced. Caffeine does something different. In the Burke et al. (2015) inpatient study, participants who received caffeine showed a delayed melatonin curve, not a flattened one. The amplitude of melatonin release was preserved. The onset was pushed later.
This means caffeine is not reducing your melatonin levels. It is changing when they rise. The result is a circadian phase delay that may persist beyond a single night — your clock has been moved, and it does not reset to its prior position the next day without appropriate morning light exposure.
Does Decaf Coffee Affect Sleep?
Decaf coffee contains 2 to 15 milligrams of residual caffeine per cup. For most people this is negligible — the dose-response evidence from Gardiner et al. (2025) shows no measurable sleep effect from 100 milligrams, making 2 to 15 milligrams unlikely to produce detectable disruption.
Decaffeination removes approximately 97 percent of caffeine from coffee beans, leaving 2 to 15 mg per 8-ounce cup depending on the bean and brewing method. Given that the Gardiner et al. (2025) crossover trial found zero measurable sleep disruption from 100 mg at any timing point, residual caffeine in decaf is well below the threshold for concern — even for slow metabolizers or those with high-sensitivity ADORA2A genotypes.
The more relevant question for decaf drinkers is whether other compounds in coffee (chlorogenic acids, diterpenes) have independent effects on sleep. Current evidence does not support a meaningful sleep-disrupting effect from these compounds at typical intake levels.
How Much Caffeine Does It Take to Disrupt Sleep?
A meta-analysis of 22 controlled crossover trials found that caffeine reduced total sleep time by approximately 35 minutes on average, but the effect was dose-dependent. At moderate doses (100 to 200 mg), effects were minimal. At higher doses (300 to 400 mg), total sleep time, sleep efficiency, and slow-wave sleep were all impaired.
Chang, Cheng, and Cheng (2025) pooled data from 22 crossover trials and found an overall reduction in total sleep time of approximately 35 minutes, a roughly 5 percent decrease in sleep efficiency, and a reduction in slow-wave sleep percentage. But these averages obscure the dose-response gradient.
At moderate doses (100 to 200 mg), reductions in total sleep time and sleep efficiency were small and often did not reach statistical thresholds. At higher doses (300 to 400 mg), the effects on total sleep time and sleep efficiency were consistent and measurable across studies.
Younger adults appeared more sensitive to caffeine’s disruptive effects on multiple sleep parameters. The threshold for disruption depends on both dose and age, though the precise age-stratified patterns require further investigation.
Related Reading
- Circadian Rhythm Disruption: How a Misaligned Internal Clock Fragments Sleep, Drives 3AM Wakeups, and Reduces Sleep Depth — the full overview of circadian sleep disruption, including melatonin, cortisol, body temperature, peripheral clocks, and early-morning waking
- Why Does Melatonin Work for Jet Lag but Not Chronic Insomnia? — why melatonin works as a timing cue, not a general sleep-depth fix
- Is Your Pineal Gland Calcifying? How Age-Related Melatonin Production Loss Differs From Receptor Decline — how pineal calcification, melatonin output, and receptor decline diverge with age
- How Many Melanopic Lux Do You Need for Circadian Entrainment? — how melanopic light exposure sets the clock more directly than ordinary lux
- How Much Blue Light From Your Phone Suppresses Melatonin? The Dose-Duration Relationship — how screen brightness, duration, and melanopic irradiance affect melatonin timing
- Do Blue Light Glasses Help You Sleep? Amber vs Standard Lenses — What Studies Show — how amber lenses, clear lenses, and evening screens differ in sleep studies
- How Long Does It Take to Reset Your Circadian Rhythm? — why the reset timeline differs for the SCN, peripheral clocks, and behavioral cues
References
1. Burke, T. M., Markwald, R. R., McHill, A. W., Chinoy, E. D., Snider, J. A., Bessman, S. C., Jung, C. M., O’Neill, J. S., & Wright, K. P., Jr. (2015). Effects of caffeine on the human circadian clock in vivo and in vitro. Science Translational Medicine, 7(305), 305ra146. https://pubmed.ncbi.nlm.nih.gov/26378246/
2. Jagannath, A., Varga, N., Dallmann, R., Rando, G., Gosselin, P., Ebrahimjee, F., Taylor, L., Mosneagu, D., Stefaniak, J., Walsh, S., Palumaa, T., Di Pretoro, S., Sanghani, H., Wakaf, Z., Churchill, G. C., Galione, A., Peirson, S. N., Boison, D., Brown, S. A., Foster, R. G., & Vasudevan, S. R. (2021). Adenosine integrates light and sleep signalling for the regulation of circadian timing in mice. Nature Communications, 12(1), 2113. https://pubmed.ncbi.nlm.nih.gov/33837202/
3. Reichert, C. F., Deboer, T., & Landolt, H. P. (2022). Adenosine, caffeine, and sleep-wake regulation: State of the science and perspectives. Journal of Sleep Research, 31(4), e13597. https://pubmed.ncbi.nlm.nih.gov/35575450/
4. Low, J. J., Tan, B. J., Yi, L. X., Zhou, Z. D., & Tan, E. K. (2024). Genetic susceptibility to caffeine intake and metabolism: A systematic review. Journal of Translational Medicine, 22(1), 961. https://pubmed.ncbi.nlm.nih.gov/39438936/
5. Rétey, J. V., Adam, M., Khatami, R., Luhmann, U. F. O., Jung, H. H., Berger, W., & Landolt, H. P. (2007). A genetic variation in the adenosine A2A receptor gene (ADORA2A) contributes to individual sensitivity to caffeine effects on sleep. Clinical Pharmacology & Therapeutics, 81(5), 692-698. https://pubmed.ncbi.nlm.nih.gov/17329997/
6. Grzegorzewski, J., Bartsch, F., Köller, A., & König, M. (2021). Pharmacokinetics of caffeine: A systematic analysis of reported data for application in metabolic phenotyping and liver function testing. Frontiers in Pharmacology, 12, 752826. https://pubmed.ncbi.nlm.nih.gov/35280254/
7. Frozi, J., de Carvalho, H. W., Ottoni, G. L., Cunha, R. A., & Lara, D. R. (2018). Distinct sensitivity to caffeine-induced insomnia related to age. Journal of Psychopharmacology, 32(1), 89-95. https://pubmed.ncbi.nlm.nih.gov/28879806/
8. Gardiner, C. L., Weakley, J., Burke, L. M., Fernandez, F., Johnston, R. D., Leota, J., Russell, S., Munteanu, G., Townshend, A., & Halson, S. L. (2025). Dose and timing effects of caffeine on subsequent sleep: A randomized clinical crossover trial. Sleep, 48(4), zsae230. https://pubmed.ncbi.nlm.nih.gov/39377163/
9. Chang, Y. H., Cheng, Y. C., & Cheng, W. J. (2025). Age- and dose-specific effects of caffeine on sleep: A meta-analysis of controlled crossover trials. Sleep Medicine, 136, 106874. https://pubmed.ncbi.nlm.nih.gov/41124973/
Written by Kat Fu, M.S., M.S. ? Last reviewed: May 2026 ? 9 references cited
