Many adults over 50 produce less melatonin than they did at 30. That much is well established. What is less understood is that melatonin function declines through two separate pathways, and supplements only address one of them.
This article covers both pathways — production decline from pineal gland calcification and receptor downregulation across the body — and what each means for supplementation decisions. Understanding which pathway is responsible matters because the interventions differ. Melatonin decline is one component of circadian disruption. The full cause overview covers how the broader circadian clock changes with age in the Circadian Rhythm Disruption overview on circadian sleep disruption.
Does the Pineal Gland Calcify With Age — and How Common Is It?
Belay and Worku (2023) conducted a systematic review and meta-analysis of imaging studies across multiple countries and found a combined pineal calcification prevalence of 61.65 percent in adults. Age was the dominant predictor, with male sex and white ethnicity as independent additional risk factors (Belay & Worku, 2023).
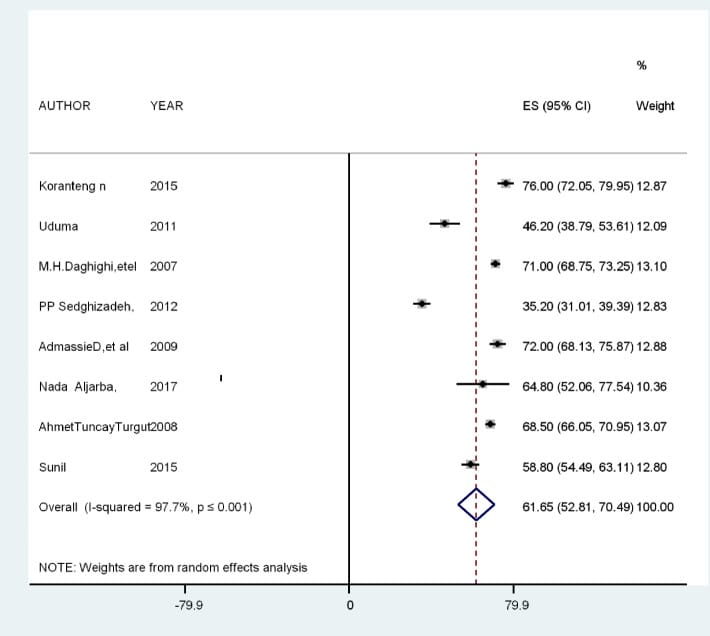
Beker-Acay et al. (2016) added volumetric detail using CT scans across age decades, measuring a median total pineal volume of 88.5 mm3 in healthy adults. Calcification volume increased progressively through the fifth and sixth decades, then declined after age 70 — likely because the gland’s uncalcified, melatonin-producing tissue has already undergone enough involution that the gland itself shrinks (Beker-Acay et al., 2016).
Tan et al. (2018) connected calcification directly to melatonin output. Melatonin levels correlated negatively with calcification size and positively with the volume of remaining uncalcified pineal tissue. This is a structural constraint — calcification leads to degeneration of pinealocytes (melatonin-producing cells) that the gland depends on for synthesis (Tan et al., 2018).
The three findings together form a coherent picture: calcification is common (62 percent of adults), it progresses with age (declining in the oldest decades when uncalcified tissue is depleted), and it directly reduces melatonin output.
How Much Does Melatonin Production Decline After 50?
Kennaway et al. (1999) quantified the decline by measuring urinary 6-sulfatoxymelatonin (aMT6s) — the primary metabolite used to measure pineal output — across age groups. The oldest group excreted 36 percent less aMT6s than the youngest. But the decline was not linear. The steepest drop occurred between young adulthood and the early 30s. After that, the curve flattened. A 55-year-old produces less melatonin than a 35-year-old, but that difference is smaller than the reduction that already occurred between ages 20 and 30 (Kennaway et al., 1999).
The midlife perception of “my melatonin is crashing” is understandable, but the steepest decline already happened a decade or two earlier. What happens after 50 is a slower attrition on top of an already-reduced baseline.
Gobetti et al. (2026) studied 17 people who had their pineal glands surgically removed — producing no melatonin at all. Despite this, they maintained a normal 24-hour circadian cycle. Activity patterns continued cycling daily, as measured by actigraphy. The suprachiasmatic nucleus (SCN), the brain’s master circadian clock, does not depend on melatonin to keep time. However, some of these individuals reported poorer subjective sleep quality, though most had only minor sleep disturbances (Gobetti et al., 2026).
Melatonin production loss can degrade sleep quality but does not collapse circadian rhythm. The SCN runs on light input and internal oscillators. Production decline costs the body a sleep-quality cue and a neuroprotective molecule, not the circadian scaffold itself.
What Happens to Your Melatonin Receptors as You Age?
Wu et al. (2007) examined postmortem human brain tissue and found that MT1 receptor-expressing neurons in the SCN decreased with age — and decreased further in Alzheimer’s disease. Other neuropeptides in the same SCN region — arginine vasopressin (AVP) and vasoactive intestinal peptide (VIP) — remained unchanged. The decline was receptor-specific, not generalized neuronal loss (Wu et al., 2007).
Sánchez-Hidalgo et al. (2009) extended this finding beyond the brain. In an aging rat model, MT1 and MT2 receptor mRNA expression declined in the spleen, liver, kidney, and heart — though notably, the thymus showed increased receptor expression with age (Sánchez-Hidalgo et al., 2009).
Klosen (2024) published a 37-year review of MT1 and MT2 receptor localization in the brain. One finding explains why receptor decline receives little attention: melatonin receptors are expressed at low levels. Standard immunohistochemistry faces sensitivity and specificity challenges in detecting them. This low expression baseline means that even modest age-related reductions can drop receptor density below the threshold needed for a functional response (Klosen, 2024).
The receptor pathway creates a problem that supplementation cannot address. A person over 60 may have both reduced production (from pineal calcification) and reduced receptor density (from age-related downregulation). Supplemental melatonin addresses the first problem. It does not restore receptor expression in the SCN, liver, heart, or immune tissues.
Can Melatonin Supplements Compensate for Both Production Loss and Receptor Decline?
Tuft et al. (2023) reviewed the risks of melatonin supplementation in older adults. The recommended dose for older adults is 0.3 to 1.0 milligrams, taken 30 to 60 minutes before desired sleep onset. Doses of 5 to 10 milligrams, available over the counter, exceed the physiological range by an order of magnitude (Tuft et al., 2023).
Preclinical research on melatonin receptor pharmacology suggests that sustained supraphysiological melatonin concentrations can desensitize MT2 receptors through internalization, potentially reducing the effectiveness of high doses over time (Liu et al., 2016).
Hardeland (2012) reviewed the multiple causes of age-related melatonin decline, including SCN deterioration, impaired neuronal transmission to the pineal, and pineal calcification. Production loss can be partially compensated for by exogenous melatonin. However, the high interindividual variability in age-related melatonin decline — pineal output can vary by a factor of 20 among apparently healthy individuals — helps explain why supplementation produces variable results in older adults (Hardeland, 2012).
Melatonin supplementation has a limited but useful role in compensating for age-related production decline — and the effective dose is lower than what many products suggest. Supplementation cannot restore receptor sensitivity. For older adults, melatonin is one piece of a larger circadian maintenance approach, not a complete answer on its own.
Many people have more than one cause contributing to their sleep disruption. Melatonin production and receptor changes — the mechanisms covered here — may compound with metabolic, hormonal, inflammatory, or autonomic factors. Identifying which causes might be involved is a useful next step.
Find out which causes might be driving your 3 AM wakeups →
Frequently Asked Questions
Can You Reverse Pineal Gland Calcification?
Chlubek and Sikora (2020) analyzed fluoride concentrations in human pineal glands and found that fluoride accumulation can be substantial — exceeding other soft tissues by a wide margin. The pineal gland sits outside the blood-brain barrier and receives disproportionately high blood flow relative to its size. Fluoride accumulation appears to be a secondary phenomenon to calcification, and above a certain threshold the two become strongly correlated (Chlubek & Sikora, 2020).
Fluoride is a plausible amplifier, not a proven initiator — calcification occurs in fluoride-deficient environments too. No known approach reverses established pineal calcification in humans.
At What Age Does Melatonin Decline Occur Fastest?
Kennaway et al. (1999) measured urinary aMT6s across age ranges and confirmed the curve was not linear. The sharpest decline occurred from the late teens and twenties into the early thirties. Beyond that, the reduction continued at a slower rate (Kennaway et al., 1999).
If sleep onset difficulty is new in a person’s 50s, the more likely contributor is receptor decline or other circadian changes (SCN weakening, light exposure patterns, cortisol timing) rather than a sudden drop in melatonin production — because the production-side decline started decades earlier.
Does Pineal Calcification Affect Alzheimer’s Risk?
Savaskan et al. (2005) identified MT2 receptor expression decreases in the hippocampus of people with Alzheimer’s disease. MT2 receptors in this region may mediate some of melatonin’s protective effects in the hippocampus. Their loss narrows the window where melatonin-based approaches can help — the region that needs melatonin’s protective effects becomes progressively less able to receive them (Savaskan et al., 2005).
Wu et al. (2007) showed the same pattern in the SCN: MT1 receptor density was lower in people with Alzheimer’s than in age-matched controls without dementia. Normal aging reduces MT1 receptors in the SCN, and Alzheimer’s accelerates that reduction further. The specificity of this loss — with neighboring neuropeptide pathways remaining intact — suggests it is tied to the melatonin response pathway rather than general neurodegeneration (Wu et al., 2007).
The direction of causality remains open. Reduced receptor expression could remove a protective mechanism and accelerate disease progression, or the neurodegenerative process itself may destroy receptor-expressing neurons. What is established is that people with Alzheimer’s have less capacity to use melatonin — whether endogenous or supplemental — in the brain regions where it would provide protection.
Related Reading
- Circadian Rhythm Disruption: How a Misaligned Internal Clock Fragments Sleep, Drives 3AM Wakeups, and Reduces Sleep Depth — the full overview of circadian sleep disruption, including melatonin, cortisol, body temperature, peripheral clocks, and early-morning waking
- Why Does Melatonin Work for Jet Lag but Not Chronic Insomnia? — why melatonin works as a timing cue, not a general sleep-depth fix
- How Many Melanopic Lux Do You Need for Circadian Entrainment? — how melanopic light exposure sets the clock more directly than ordinary lux
- How Much Blue Light From Your Phone Suppresses Melatonin? The Dose-Duration Relationship — how screen brightness, duration, and melanopic irradiance affect melatonin timing
- Do Blue Light Glasses Help You Sleep? Amber vs Standard Lenses — What Studies Show — how amber lenses, clear lenses, and evening screens differ in sleep studies
- How Long Does It Take to Reset Your Circadian Rhythm? — why the reset timeline differs for the SCN, peripheral clocks, and behavioral cues
- How Does Caffeine Delay Your Circadian Clock by 40 Minutes? — how caffeine delays circadian phase through adenosine and clock-gene timing
References
- Belay, D. G., & Worku, M. G. (2023). Prevalence of pineal gland calcification: A systematic review and meta-analysis. Systematic Reviews, 12(1), 32. https://pubmed.ncbi.nlm.nih.gov/36879256/
- Beker-Acay, M., Turamanlar, O., Horata, E., Unlu, E., Fidan, N., & Oruc, S. (2016). Assessment of pineal gland volume and calcification in healthy subjects: Is it related to aging? Journal of the Belgian Society of Radiology, 100(1), 13. https://pubmed.ncbi.nlm.nih.gov/30038974/
- Chlubek, D., & Sikora, M. (2020). Fluoride and pineal gland. Applied Sciences, 10(8), 2885. https://doi.org/10.3390/app10082885
- Gobetti, R. A. P., Bueno, C., Soster, L. M. S. F. A., Monazzi, A. C. C. B. L., do Amaral, F. G., Capellano, A. M., da Silva, N. S., & Cipolla-Neto, J. (2026). Sleep and rhythmic profile after pineal gland removal in humans. Journal of Sleep Research, 35(2), e70171. https://pubmed.ncbi.nlm.nih.gov/40808320/
- Hardeland, R. (2012). Melatonin in aging and disease — Multiple consequences of reduced secretion, options and limits of treatment. Aging and Disease, 3(2), 194-225. https://pubmed.ncbi.nlm.nih.gov/22724080/
- Kennaway, D. J., Lushington, K., Dawson, D., Lack, L., van den Heuvel, C., & Rogers, N. (1999). Urinary 6-sulfatoxymelatonin excretion and aging: New results and a critical review of the literature. Journal of Pineal Research, 27(4), 210-220. https://pubmed.ncbi.nlm.nih.gov/10551768/
- Klosen, P. (2024). Thirty-seven years of MT1 and MT2 melatonin receptor localization in the brain: Past and future challenges. Journal of Pineal Research, 76(3), e12955. https://pubmed.ncbi.nlm.nih.gov/38606787/
- Liu, J., Clough, S. J., Hutchinson, A. J., Adamah-Biassi, E. B., Popovska-Gorevski, M., & Dubocovich, M. L. (2016). MT1 and MT2 melatonin receptors: A therapeutic perspective. Annual Review of Pharmacology and Toxicology, 56, 361-383. https://pubmed.ncbi.nlm.nih.gov/26514204/
- Sánchez-Hidalgo, M., Guerrero Montáez, J. M., Carrascosa-Salmoral, M. D. P., Naranjo Gutierrez, M. D. C., Lardone, P. J., & de la Lastra Romero, C. A. (2009). Decreased MT1 and MT2 melatonin receptor expression in extrapineal tissues of the rat during physiological aging. Journal of Pineal Research, 46(1), 29-35. https://pubmed.ncbi.nlm.nih.gov/18513209/
- Savaskan, E., Ayoub, M. A., Ravid, R., Angeloni, D., Fraschini, F., Meier, F., Eckert, A., Müller-Spahn, F., & Jockers, R. (2005). Reduced hippocampal MT2 melatonin receptor expression in Alzheimer’s disease. Journal of Pineal Research, 38(1), 10-16. https://pubmed.ncbi.nlm.nih.gov/15617532/
- Tan, D. X., Xu, B., Zhou, X., & Reiter, R. J. (2018). Pineal calcification, melatonin production, aging, associated health consequences and rejuvenation of the pineal gland. Molecules, 23(2), 301. https://pubmed.ncbi.nlm.nih.gov/29385085/
- Tuft, C., Matar, E., Menczel Schrire, Z., Grunstein, R. R., Yee, B. J., & Hoyos, C. M. (2023). Current insights into the risks of using melatonin as a treatment for sleep disorders in older adults. Clinical Interventions in Aging, 18, 49-59. https://pubmed.ncbi.nlm.nih.gov/36660543/
- Wu, Y.-H., Zhou, J.-N., Van Heerikhuize, J., Jockers, R., & Swaab, D. F. (2007). Decreased MT1 melatonin receptor expression in the suprachiasmatic nucleus in aging and Alzheimer’s disease. Neurobiology of Aging, 28(8), 1239-1247. https://pubmed.ncbi.nlm.nih.gov/16837102/
Written by Kat Fu, M.S., M.S. ? Last reviewed: May 2026 ? 13 references cited
