If you have spent money on blue light glasses expecting better sleep, the brand and marketing copy on the box are not the variables that determine whether they work. The variable is what wavelengths the lenses absorb — and whether those wavelengths overlap with the narrow band that drives melatonin suppression through melanopsin, a photopigment in the retina.
This article covers the wavelength science behind why lens type matters, the two RCTs where amber lenses improved sleep, why reviews that pool all lens types together produce null results, and how to separate the light-exposure problem from the screen-content problem. For the broader picture of how circadian disruption contributes to 3 am wakeups, see the parent overview on circadian sleep disruption.
What Do Blue Light Glasses Block — and Which Wavelength Matters for Sleep?
Melatonin suppression from light exposure is wavelength-dependent. The photopigment responsible is melanopsin, expressed in a subset of retinal ganglion cells (ipRGCs) that project directly to the suprachiasmatic nucleus — the master circadian pacemaker in the hypothalamus. When melanopsin absorbs light in its activation range, it sends a “daytime” input to the circadian clock, suppressing pineal melatonin production.
Prayag, Najjar, and Gronfier (2019) established that the melanopsin-driven response has a threshold around 1.5 melanopic lux. Below this intensity, melatonin suppression is minimal regardless of wavelength. Above it, the spectral composition of the light determines how much suppression occurs. Melanopsin’s peak sensitivity sits at approximately 480-485 nm, as established by action spectrum studies (St Hilaire et al., 2022).
St Hilaire et al. (2022) mapped the full action spectrum for melatonin suppression in humans using 6.5 hours of monochromatic light at six wavelengths. The peak sensitivity for sustained melatonin suppression fell at 481 nm. But the study revealed something more nuanced: the photoreceptor contribution changes over the duration of exposure. During the first quarter (approximately 97 minutes), S-cones (short-wavelength cones, peak ~441 nm) and L+M cones contributed 51% and 47% of the melatonin suppression response, respectively, with ipRGCs contributing only 2%. By the fourth quarter of the 6.5-hour exposure, ipRGCs dominated at 92% of the response, with peak sensitivity at 485 nm (St Hilaire et al., 2022).
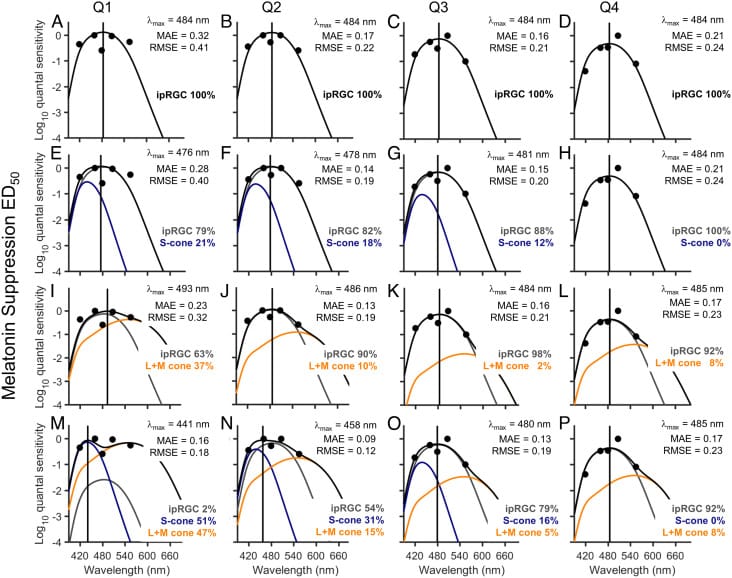
This has a practical implication for lens selection. Brief screen exposure (under 30 minutes) may involve cone-mediated suppression at shorter wavelengths (~440 nm). Sustained evening exposure — the kind that occurs during two or three hours of screen use before bed — is dominated by melanopsin at 480-485 nm. Lenses that block only the violet range (<420 nm) miss the wavelengths that matter during sustained exposure.
Najjar, Prayag, and Gronfier (2024) added an age dimension. In younger adults, the melatonin suppression action spectrum was well-fit by a single melanopsin template peaking at 485 nm. In older adults, age-related changes in ocular optics may alter spectral transmission to the retina, and the best-fit model combined melanopsin, S-cone, and M-cone contributions with peak sensitivity closer to 500 nm. For adults over 50, the effective suppression band may extend further into blue-green wavelengths than melanopsin alone would predict (Najjar et al., 2024).
This means the 460-490 nm blocking range often cited for amber lenses is a reasonable starting point, but broader-spectrum amber lenses that extend blocking into the 500-530 nm range may be more relevant for older adults.
Do Amber Lenses Improve Sleep? What the Randomized Trials Show
Burkhart and Phelps (2009) conducted the first controlled trial of amber lenses and sleep. Twenty adults were randomized to wear either amber-tinted lenses (blocking wavelengths below ~530 nm) or UV-only control lenses for three hours before bedtime over two weeks. The amber group showed improved self-reported sleep quality and mood on sleep diary measures (Burkhart & Phelps, 2009).
The study had limitations: small sample (n=20), reliance on self-report rather than actigraphy or polysomnography, and a UV-only control that was not a true sham (participants could tell which lens was amber). But it was the first to demonstrate that blocking the melanopsin-relevant wavelength band produced a measurable sleep quality change.
Shechter et al. (2018) addressed several of those limitations. In a randomized, crossover controlled trial of 14 adults with insomnia, participants wore amber lenses or clear (sham) lenses for two hours before bedtime over seven consecutive nights per condition, with a four-week washout period between conditions.
The results, measured by wrist actigraphy:
- Total sleep time was higher in the amber lens condition (p = 0.035)
- Participants reported subjective improvements in sleep quality, total sleep time, and soundness of sleep on the Pittsburgh Insomnia Rating Scale (PIRS)
Increased total sleep time from a lens-only approach is a noteworthy effect in sleep research. The amber lenses achieved this by reducing melanopic light input during the two hours preceding sleep onset — allowing endogenous melatonin to rise on its natural schedule (Shechter et al., 2018).
The distinction between these two studies and the negative findings in reviews comes down to lens type. Amber lenses absorb the 460-530 nm range where melanopsin is active. Standard “blue light” lenses — the kind sold at optical shops and online retailers — typically block wavelengths below 400-420 nm. That range is in the near-UV/violet portion of the spectrum, which is not where melanopsin-driven melatonin suppression occurs. A lens that blocks 410 nm but transmits 480 nm is cosmetically “blue light blocking” but functionally irrelevant to the melanopsin pathway.
Why Do Some Studies Say Blue Light Glasses Do Not Work?
Singh et al. (2023) published the Cochrane review that generated headlines declaring blue light glasses ineffective for sleep. The review included 17 RCTs comparing blue-light-filtering lenses to non-filtering lenses, of which six assessed sleep outcomes. Three trials reported positive effects on sleep; three reported no effect. The overall result was indeterminate (very low-certainty evidence).
The review did not stratify by lens filtering range. This is one gap that matters. A trial using amber lenses that block 460-530 nm tests a different optical approach than a trial using lightly coated transparent lenses that block below 420 nm. Combining them into a single analysis is equivalent to pooling trials of two different drugs and concluding “medication” does not work because the average effect washes out (Singh et al., 2023).
Luna-Rangel et al. (2025) conducted a meta-analysis of three crossover RCTs (n = 49) that used actigraphy as the outcome measure. The pooled results:
- Sleep onset latency: -4.86 minutes favoring blue-blocking lenses (95% CI: -20.23 to 10.52; p = 0.54)
- Total sleep time: +8.75 minutes favoring blue-blocking lenses (95% CI: -35.31 to 52.82; p = 0.70)
- Sleep efficiency: -0.61% (95% CI: -7.58 to 6.35; p = 0.86)
None reached statistical significance. The authors noted that the combined sample was small and heterogeneity was low, which suggests the studies were measuring the same (null) effect. All three included studies used amber-tinted lenses versus clear controls, indicating that the null actigraphy result cannot be attributed to lens-type pooling. The authors noted that small sample sizes, short intervention periods (one week), and heterogeneous participant characteristics (insomnia patients, athletes, and healthy adults) limit the strength of these conclusions (Luna-Rangel et al., 2025).

The evidence for amber lenses is mixed. Two individual RCTs (Burkhart 2009, Shechter 2018) found positive results on subjective sleep quality and actigraphy-measured sleep time. However, when three amber-lens crossover RCTs were pooled in the Luna-Rangel (2025) meta-analysis, the actigraphy outcomes did not reach statistical significance. Transparent or lightly tinted “blue light” lenses that block only violet/near-UV wavelengths show null results consistently. The Cochrane review (Singh 2023) combined studies using different lens types without stratification, which adds a lens-classification confound to its findings — but lens type alone does not explain all null results in the literature.
The mixed evidence reflects multiple factors: lens-type classification (particularly in the Cochrane review), small samples, short intervention durations, and population heterogeneity. It does not demonstrate that evening melanopic light exposure is irrelevant to sleep — but it does indicate that amber lenses may not produce large or consistent actigraphy-measurable effects across all populations.
Is It the Blue Light or the Content Keeping You Awake?
The wavelength-dependent melatonin suppression from screen light is a photobiological effect. Melanopsin in ipRGCs responds to the spectral composition of incoming light, and if the light contains sufficient energy in the 460-490 nm range above the ~1.5 melanopic lux threshold, pineal melatonin production is suppressed. This is a physiological response that does not depend on what the screen is displaying — a white spreadsheet and a social media feed produce similar melanopic irradiance if the display settings and viewing distance are the same.
The arousal from content is a separate mechanism. Scrolling through emotionally engaging material — news, social media arguments, work emails, video content — activates sympathetic arousal, elevates cortisol, and delays the psychological wind-down that precedes sleep onset. This effect is content-dependent and behavioral, not wavelength-dependent. A person reading an engaging article on a Kindle Paperwhite (which has minimal melanopic light output) can still delay sleep onset through cognitive activation.
This distinction matters because amber lenses address the melanopic light problem but not the content problem. Someone who puts on amber glasses and spends two hours scrolling before bed has blocked one of two pathways. The melatonin suppression from the light is reduced, but the cortisol elevation and arousal from the content remain.
Practical implications:
- Amber lenses reduce melanopic light input, allowing melatonin to rise on schedule — they address the photobiological pathway
- Content choices in the 1-2 hours before bed affect cognitive and emotional arousal — this requires behavioral changes independent of any lens
- Screen brightness and color temperature interact with both pathways: dimmer, warmer-toned screens reduce melanopic irradiance even without lenses, and less visually stimulating content reduces arousal
Neither approach alone eliminates all screen-related sleep disruption. Both address a different contributing factor.
Evening light exposure from screens is one circadian input among several. Melatonin suppression from melanopic light may compound with hormonal changes, metabolic changes, or autonomic imbalance — especially after 40. Identifying which causes might be contributing is a useful next step.
Find out which causes might be driving your 3 AM wakeups →
Frequently Asked Questions
How Long Before Bed Should You Stop Using Screens?
The 2-hour window is not an arbitrary recommendation. It aligns with the exposure duration at which melanopsin takes over from cone-mediated responses as the primary driver of melatonin suppression (St Hilaire et al., 2022). The Burkhart and Phelps (2009) trial used a 3-hour pre-bedtime lens-wearing period; the Shechter et al. (2018) trial used 2 hours. Both found positive effects.
If eliminating screens before bed is not practical, the evidence suggests that either wearing amber lenses during that window or reducing screen brightness below the ~1.5 melanopic lux threshold identified by Prayag et al. (2019) can limit the suppressive effect on melatonin production.
Does Night Mode on Your Phone Reduce Melatonin Suppression?
Night mode works on the right principle — it reduces the 460-490 nm content of the emitted light. The limitation is magnitude. Smartphone and tablet displays at standard brightness produce 30-80 melanopic lux at typical viewing distance. Night mode reduces this, but the reduction varies by device, setting intensity, and display brightness. The warmest night-mode settings on some devices can bring melanopic output below 10 lux, which approaches the suppression threshold — but this also makes the screen visibly orange and reduces color accuracy.
Amber lenses are more consistent because they filter all incoming light from the environment, not just the display. Overhead LED lighting, which often peaks around 450-460 nm, contributes melanopic irradiance that night mode on a phone does not address. If the goal is reducing total melanopic input in the evening, lenses handle the visual field; night mode handles only the screen.
Do Blue Light Glasses Help Older Adults Differently?
This change occurs because the crystalline lens yellows with age, which preferentially absorbs shorter wavelengths before they reach the retina. Pupil size also decreases with age (senile miosis), reducing total light reaching the retina. Both are established age-related optical changes that reduce the melanopic light input at 480 nm relative to longer wavelengths, which may alter the relative contribution of different photoreceptor pathways to the melatonin suppression response (Najjar et al., 2024).
For older adults, this has two implications for lens selection. First, amber lenses that block only up to 490 nm may not cover the full effective suppression band, which extends toward 500-530 nm in aging eyes. Second, the overall reduction in retinal light input from lens yellowing and miosis means that older adults may be more sensitive to the melanopic light that does get through — because a smaller absolute amount of light can still exceed the suppression threshold relative to their reduced baseline.
Broader-spectrum amber or orange lenses that extend blocking into the 500-530 nm range may be more appropriate for adults over 50 than the narrower-band amber lenses tested in the original RCTs.
Related Reading
- Circadian Rhythm Disruption: How a Misaligned Internal Clock Fragments Sleep, Drives 3AM Wakeups, and Reduces Sleep Depth — the full overview of circadian sleep disruption, including melatonin, cortisol, body temperature, peripheral clocks, and early-morning waking
- Why Does Melatonin Work for Jet Lag but Not Chronic Insomnia? — why melatonin works as a timing cue, not a general sleep-depth fix
- Is Your Pineal Gland Calcifying? How Age-Related Melatonin Production Loss Differs From Receptor Decline — how pineal calcification, melatonin output, and receptor decline diverge with age
- How Many Melanopic Lux Do You Need for Circadian Entrainment? — how melanopic light exposure sets the clock more directly than ordinary lux
- How Much Blue Light From Your Phone Suppresses Melatonin? The Dose-Duration Relationship — how screen brightness, duration, and melanopic irradiance affect melatonin timing
- How Long Does It Take to Reset Your Circadian Rhythm? — why the reset timeline differs for the SCN, peripheral clocks, and behavioral cues
- How Does Caffeine Delay Your Circadian Clock by 40 Minutes? — how caffeine delays circadian phase through adenosine and clock-gene timing
References
1. Prayag, A. S., Najjar, R. P., & Gronfier, C. (2019). Melatonin suppression is exquisitely sensitive to light and primarily driven by melanopsin in humans. Journal of Pineal Research, 66(4), e12562. https://pubmed.ncbi.nlm.nih.gov/30697806/
2. St Hilaire, M. A., Ámundadóttir, M. L., Rahman, S. A., Rajaratnam, S. M. W., Rüger, M., Brainard, G. C., Czeisler, C. A., Andersen, M., Gooley, J. J., & Lockley, S. W. (2022). The spectral sensitivity of human circadian phase resetting and melatonin suppression to light changes dynamically with light duration. Proceedings of the National Academy of Sciences of the United States of America, 119(51), e2205301119. https://pubmed.ncbi.nlm.nih.gov/36508661/
3. Najjar, R. P., Prayag, A. S., & Gronfier, C. (2024). Melatonin suppression by light involves different retinal photoreceptors in young and older adults. Journal of Pineal Research, 76(1), e12930. https://pubmed.ncbi.nlm.nih.gov/38241677/
4. Burkhart, K., & Phelps, J. R. (2009). Amber lenses to block blue light and improve sleep: A randomized trial. Chronobiology International, 26(8), 1602-1612. https://pubmed.ncbi.nlm.nih.gov/20030543/
5. Shechter, A., Kim, E. W., St-Onge, M.-P., & Westwood, A. J. (2018). Blocking nocturnal blue light for insomnia: A randomized controlled trial. Journal of Psychiatric Research, 96, 196-202. https://pubmed.ncbi.nlm.nih.gov/29101797/
6. Singh, S., Keller, P. R., Busija, L., McMillan, P., Makrai, E., Lawrenson, J. G., Hull, C. C., & Downie, L. E. (2023). Blue-light filtering spectacle lenses for visual performance, sleep, and macular health in adults. Cochrane Database of Systematic Reviews, 8(8), CD013244. https://pubmed.ncbi.nlm.nih.gov/37593770/
7. Luna-Rangel, F. A., González-Bedolla, B., Salazar-Ortega, M. J., Torres-Mancilla, X. M., & Martínez-Cadena, S. (2025). Efficacy of blue-light blocking glasses on actigraphic sleep outcomes: a systematic review and meta-analysis of randomized controlled crossover trials. Frontiers in Neurology, 16, 1699303. https://pubmed.ncbi.nlm.nih.gov/41341515/
Written by Kat Fu, M.S., M.S. ? Last reviewed: May 2026 ? 7 references cited
