The phrase “3am waking” is anecdotally one of the most common sleep complaints reported by semaglutide and tirzepatide users, and the mechanism behind it connects two well-documented phenomena: GLP-1-driven appetite suppression and nocturnal blood sugar regulation. Despite the volume of consumer questions about this pattern, almost no consumer-facing content has connected these two systems in a single explanation.
This article traces the specific chain from reduced food intake to nocturnal glucose depletion to counter-regulatory hormone surge to full awakening. It does not cover all GLP-1 sleep effects (those are addressed in the sibling articles linked below). Blood sugar instability is one of the primary metabolic causes of sleep disruption, and the full metabolic framework is covered in the parent pillar page. Other articles in this cluster address the orexin-arousal mechanism, vivid dreams, and appetite-serotonin disruption.
How Does Semaglutide’s Appetite Suppression Lead to a Blood Sugar Crash at Night
The magnitude of semaglutide’s appetite suppression is well-established. The STEP clinical trial program showed 15–17% body weight loss over treatment periods, implying sustained caloric deficits of 500–1,000+ kcal/day in many participants. That degree of undereating, sustained over weeks and months, has direct consequences for overnight glucose stability.
The liver stores glucose as glycogen — a reserve that supports approximately 12 hours of fasting under normal caloric intake. When daily food intake drops by 500+ calories, glycogen stores deplete faster. A person who eats a small dinner at 6pm and sleeps until 6am is fasting for 12 hours with a glycogen reserve that may have been insufficient from the start. By 2–3am, blood glucose begins to fall.
Semaglutide’s delayed gastric emptying compounds this pattern. Dinner nutrients absorb more slowly, creating a glucose peak in the early hours of sleep followed by a steeper decline in the second half of the night — a peak-then-crash trajectory that aligns precisely with the 3am vulnerability window.
A 2025 review on integrating continuous glucose monitoring with GLP-1 receptor agonist therapy identified appetite-driven meal timing shifts and meal skipping as underrecognized contributors to nocturnal glucose instability in this population (Gupta & Pozzilli, 2025). The authors noted that GLP-1 receptor agonist-related appetite dysregulation alters nocturnal glucose dynamics in ways that conventional monitoring cannot detect.
A 2024 case report documented the opposite direction of semaglutide’s glucose effects: in a patient with severe reactive hypoglycemia after bariatric surgery, semaglutide reduced time spent below 70 mg/dL from 12% to 4% at a dose of 0.25 mg/week, and further to 1% at the 0.5 mg/week maintenance dose, as measured by continuous glucose monitoring (Fiore et al., 2024). The case illustrates that semaglutide can affect glucose stability in both directions — stabilizing it in some contexts and destabilizing it in others — depending on baseline metabolic conditions and food intake patterns.
What Happens When Semaglutide Drives Your Blood Sugar Too Low at Night
Sleep itself changes the threshold at which the counter-regulatory system activates. In a controlled study, healthy subjects required blood glucose to fall approximately 0.6 mmol/L lower during sleep than during wakefulness before their protective hormone response engaged (Gais et al., 2003). The counter-regulatory system is not absent during sleep — it is delayed. Blood glucose falls further before the alarm triggers.
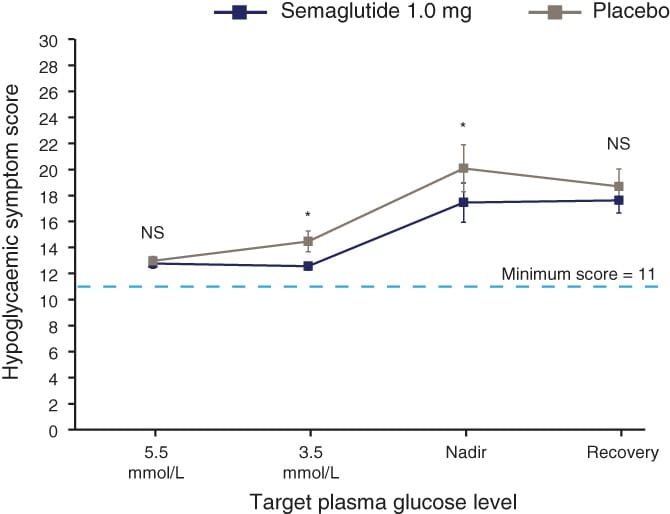
The timing of the glucose drop within the sleep period also matters. In a study comparing hypoglycemia induced during early sleep (immediately after sleep onset) versus late sleep (3.5 hours after onset), all 16 healthy subjects awoke during late-sleep hypoglycemia, compared to 10 of 16 during early-sleep hypoglycemia (Jauch-Chara et al., 2007). Despite more complete awakening during late sleep, the counter-regulatory hormone responses — epinephrine, norepinephrine, ACTH, cortisol, and growth hormone — were all significantly weaker during late-night episodes. The body wakes you up at 3am but is least equipped to correct the glucose deficit at that hour.
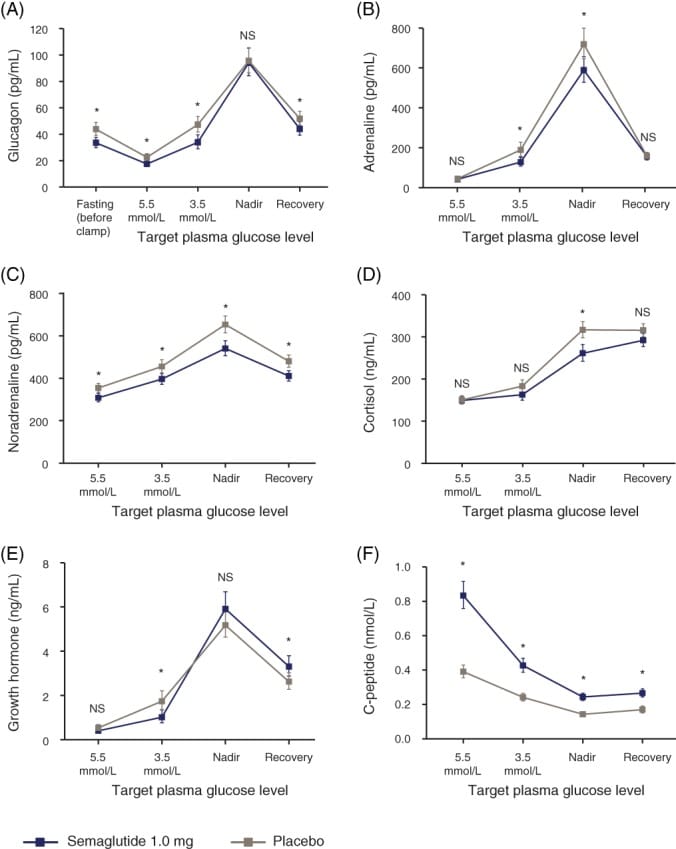
Semaglutide adds a specific layer to this vulnerability. In a randomized, placebo-controlled crossover trial, semaglutide significantly attenuated the cortisol and noradrenaline responses to experimentally induced hypoglycemia compared to placebo (Korsatko et al., 2018). The glucagon response — the first-line defense — remained intact. But the sympathoadrenal arm of counter-regulation, which drives the arousal signal, was blunted. For users already in the late-sleep window where counter-regulatory hormones are weakest, semaglutide’s attenuation of cortisol and noradrenaline represents a compounding vulnerability.
The cortisol nadir — the lowest point in cortisol’s 24-hour rhythm — occurs around 2–3am. This is the window when glycogen stores are most depleted, counter-regulatory hormone responses are weakest, and cortisol is least available to mobilize alternative fuel sources. The convergence of these three factors at the same hour is why 3am is the most reported waking time.
Why Does Semaglutide Make Nighttime Blood Sugar Drops Harder to Detect
A comprehensive review of sleep’s interaction with hypoglycemia defense found that sleep weakens the entire neuroendocrine defense cascade: glucagon, adrenaline, cortisol, and growth hormone responses are all attenuated during sleep compared to matched waking conditions (Jauch-Chara & Schultes, 2010). The review also documented a feed-forward mechanism — undetected nocturnal hypoglycemia can progressively degrade daytime hypoglycemia awareness, meaning repeated 3am episodes may reduce a person’s ability to recognize low blood sugar during waking hours as well.
In a crossover polysomnography study, awakening rates during controlled nocturnal hypoglycemia were 27% lower in the 4–8 hour sleep window and 20% lower across the full sleep period compared to normoglycemic control nights (Jennum et al., 2015). Nocturnal hypoglycemia paradoxically suppressed the ability to wake up — the brain mounted a hormonal stress response (elevated adrenaline, cortisol, and growth hormone) but the behavioral awakening response was blunted. The suppression was most pronounced in the second half of the night, consistent with circadian weakening of the arousal system.
Continuous glucose monitoring data have confirmed that traditional fingerstick monitoring vastly underestimated the frequency of nocturnal hypoglycemic episodes (Kulzer et al., 2024). Many overnight blood sugar drops never produce conscious symptoms or remembered awakenings. CGM reveals a higher burden of unrecognized nighttime glucose events than subjective reporting captures.
For semaglutide users, sleep suppression of counter-regulatory defenses, semaglutide’s attenuation of cortisol and noradrenaline responses (Korsatko et al., 2018), and reduced food intake depleting glycogen stores faster all compound simultaneously. The result is a triple vulnerability: glucose falls further, the body detects the drop later, and the hormonal response is weaker when it finally activates.
Can a Bedtime Snack Prevent Semaglutide-Related 3am Waking
The mechanism is straightforward: complex carbohydrates replenish liver glycogen, and protein slows the rate of glucose release from that glycogen, extending the supply through the second half of the night. The objective is not a higher blood glucose level — it is a more stable blood glucose trajectory that avoids the steep drop between 2am and 4am.
Clinical data support the principle that glucose stability — not glucose level — is what matters for sleep continuity. In a two-year real-world study, semaglutide as add-on therapy in type 1 diabetes reduced glycemic variability (coefficient of variation from 46.3% to 33.6%) and increased time in range from 46% to 71% (Al Hayek et al., 2025). A randomized controlled trial in type 1 diabetes patients with obesity found semaglutide improved time in range by 8.8 percentage points while maintaining less than 4% time below range (Shah et al., 2025). Both studies demonstrate that reduced variability — fewer peaks and crashes — is the metric most associated with better glycemic outcomes.
For GLP-1 users experiencing unexplained 3am waking, a two-week continuous glucose monitoring trial can confirm or rule out blood sugar instability as the cause. CGM reveals nocturnal patterns that are invisible to subjective experience, and research shows that traditional monitoring methods underestimate the frequency of nocturnal hypoglycemia (Kulzer et al., 2024).
Any changes to food timing or composition while taking semaglutide should be discussed with a prescriber. The bedtime snack approach addresses the glycogen depletion mechanism specifically — it does not replace medical supervision of GLP-1 therapy. The non-drug version of this mechanism and its management is covered in the blood sugar drop 3am article.
Blood sugar instability during sleep is one of the most common metabolic causes of 3am waking — with or without GLP-1 medication. This mechanism often overlaps with cortisol rhythm disruption, hormonal changes, and circadian timing issues. Identifying which causes might be contributing to your specific pattern is a useful next step.
Find out which causes might be driving your 3am wakeups →
What Else Do People Ask About Semaglutide and 3am Waking
Does This Happen in People Without Diabetes
The majority of GLP-1 prescriptions for weight management go to people without diabetes, yet the nocturnal glucose research base is almost entirely drawn from type 1 and type 2 diabetes populations. This is a significant evidence gap. A 2025 review specifically noted the absence of randomized controlled trials assessing combined CGM and GLP-1 receptor agonist use in obesity without diabetes, despite widespread off-label use in this group (Gupta & Pozzilli, 2025). The physiological chain — appetite suppression leading to reduced glycogen stores leading to nocturnal glucose decline — operates through the same metabolic pathway regardless of diabetes status.
Why Is 3am the Most Common Time to Wake Up on Semaglutide
Three factors converge between 2am and 4am. First, liver glycogen has been declining for 8–10 hours since dinner. Second, cortisol — which mobilizes glucose from protein and fat — sits at its 24-hour nadir. Third, the counter-regulatory hormone response is weakest during late sleep: a study of healthy subjects found that epinephrine, norepinephrine, cortisol, and growth hormone responses to hypoglycemia were all significantly attenuated during late-night episodes compared to early-night episodes, despite more complete awakening (Jauch-Chara et al., 2007). The combination of depleted fuel, absent cortisol support, and weakened hormonal defense makes 3am the hour of maximum metabolic vulnerability.
Does Semaglutide Make It Harder for Your Body to Detect Low Blood Sugar
The Korsatko et al. (2018) trial found that semaglutide preserved the glucagon response to hypoglycemia — the first-line hormonal defense — but significantly reduced cortisol and noradrenaline responses compared to placebo. Hypoglycemic symptom recognition scores were also lower with semaglutide. The authors assessed these changes as unlikely to be clinically dangerous in isolation. However, during late-night sleep, the sympathoadrenal system is already at its weakest, and the counter-regulatory threshold is already shifted lower by approximately 0.6 mmol/L. Adding semaglutide’s attenuation on top of sleep’s existing suppression creates a compounding effect that the isolated trial conditions did not capture.
Does Semaglutide Affect Blood Sugar Differently Than Tirzepatide at Night
Semaglutide acts on the GLP-1 receptor alone. Tirzepatide activates both GLP-1 and GIP receptors, and GIP receptor activation has distinct effects on insulin secretion and glucose metabolism. Whether tirzepatide’s dual mechanism produces a different nocturnal glucose trajectory remains unstudied. Both drugs cause comparable appetite suppression at therapeutic doses, meaning both can produce the glycogen depletion pattern that drives 3am waking. Real-world CGM data comparing overnight glucose variability between semaglutide and tirzepatide in non-diabetic users does not yet exist (Gupta & Pozzilli, 2025).
Should You Use a Continuous Glucose Monitor to Track Semaglutide-Related Blood Sugar Drops
A 2024 review found that continuous glucose monitoring revealed a substantially higher burden of unrecognized nighttime hypoglycemic events than previous monitoring methods had detected (Kulzer et al., 2024). For semaglutide users who wake consistently between 2am and 4am, continuous glucose monitoring data can show whether blood glucose drops precede the waking event — confirming the mechanism described in this article — or whether the waking pattern has a different cause. This is a diagnostic question to discuss with a prescriber, not a self-treatment decision.
Related Reading
- Metabolic Sleep Disruption — The full metabolic sleep framework covering glucose regulation, insulin resistance, mitochondrial decline, NAD+ loss, and fat metabolism
- Does Ozempic Cause Insomnia What GLP-1 Drugs Do to Your Sleep — The broad overview of GLP-1 sleep side effects and mechanisms
- Why Does Ozempic Make You Tired But Keep You Awake — How GLP-1 drugs activate orexin/wakefulness circuitry while appetite suppression can create fatigue
- Does Ozempic Give You Vivid Dreams — How GLP-1 drugs may affect dream intensity through arousal and neuropsychiatric pathways
- Does Eating Too Little on Ozempic Cause Insomnia — How appetite suppression can affect tryptophan, serotonin, melatonin, and overnight glucose stability
- Does Your Ozempic Injection Day Disrupt Your Sleep The 24-48 Hour Pattern — Why sleep disruption can follow a weekly pattern tied to injection timing and peak drug exposure
- Does Your Semaglutide Dose Increase Restart Your Insomnia Why Each Titration Can Disrupt Sleep Again — Why each titration step can restart sleep disruption before adaptation catches up
- Can You Take Melatonin, Magnesium, or Sleep Aids While on Ozempic — How melatonin, magnesium, DORAs, and other sleep aids intersect with GLP-1 physiology
- Does Ozempic Give You Anxiety at Night Why GLP-1 Drugs May Contribute to Nighttime Panic — How orexin activation, HPA-axis signaling, and glucose instability may contribute to nighttime anxiety
- Do GLP-1 Drugs Disrupt Your Body Clock Ozempic, Circadian Rhythms, and Melatonin — How GLP-1 secretion rhythms, food timing, peripheral clocks, and melatonin intersect
- Ozempic Night Sweats: How GLP-1 Drugs May Affect Temperature at Night — How GLP-1 drugs may affect nighttime sweating through autonomic tone and thermoregulation
- Ozempic Nausea and Sleep: How Stomach Side Effects Keep You Awake at Night — How delayed gastric emptying, reflux, and nausea can fragment sleep
- Will Your Sleep Go Back to Normal When You Stop Ozempic — What is known and not known about sleep after semaglutide discontinuation
- Does Ozempic Muscle Loss Affect Your Sleep Sarcopenia, Temperature, and Deep Sleep — How lean-mass loss, thermoregulation, and deep sleep can interact during GLP-1 weight loss
- Could Ozempic Contribute to Restless Legs at Night Iron, B12, and the Dopamine Connection — How iron, B12, dopamine, and rapid weight loss may intersect with restless legs at night
- Does Compounded Semaglutide Affect Sleep Differently Than Brand-Name Ozempic — How dose variability, formulation differences, and adverse-event tracking can affect sleep side effects
- Ozempic and Sleep After 40: Why GLP-1 Side Effects Can Feel Different as You Age — Why GLP-1 sleep effects can land differently after 40 because of muscle, hormones, and glucose regulation
References
- Al Hayek, A., Klonoff, D. C., Al Zahrani, W. M., Ibrahim, S. E., & Al Dawish, M. A. (2025). Evaluating the effect of Semaglutide as add-on therapy on glycemic control and continuous glucose monitoring outcomes in adults with type 1 diabetes: A two-year real-world data study. Journal of Diabetes and Its Complications, 39(7), 109064. https://pubmed.ncbi.nlm.nih.gov/40318459/
- Fiore, A., Santoro, G., Lombardo, A., Spitali, F., Sceusa, G., & Gullo, D. (2024). Efficacy of Semaglutide in Reactive Hypoglycemia Related to Dumping Syndrome after Bariatric Surgery. Endocrine, Metabolic & Immune Disorders Drug Targets. https://pubmed.ncbi.nlm.nih.gov/39041260/
- Gais, S., Born, J., Peters, A., Schultes, B., Heindl, B., Fehm, H. L., & Kern, W. (2003). Hypoglycemia counterregulation during sleep. Sleep, 26(1), 55-59. https://pubmed.ncbi.nlm.nih.gov/12627733/
- Gupta, P., & Pozzilli, P. (2025). Optimising obesity management: integrating continuous glucose monitoring with GLP-1 receptor agonists. Diabetes Research and Clinical Practice, 228, 112434. https://pubmed.ncbi.nlm.nih.gov/40849048/
- Jauch-Chara, K., Hallschmid, M., Gais, S., Oltmanns, K. M., Peters, A., Born, J., & Schultes, B. (2007). Awakening and counterregulatory response to hypoglycemia during early and late sleep. Diabetes, 56(7), 1938-1942. https://pubmed.ncbi.nlm.nih.gov/17400929/
- Jauch-Chara, K., & Schultes, B. (2010). Sleep and the response to hypoglycaemia. Best Practice & Research Clinical Endocrinology & Metabolism, 24(5), 801-815. https://pubmed.ncbi.nlm.nih.gov/21112027/
- Jennum, P., Stender-Petersen, K., Rabol, R., Jorgensen, N. R., Chu, P. L., & Madsbad, S. (2015). The Impact of Nocturnal Hypoglycemia on Sleep in Subjects With Type 2 Diabetes. Diabetes Care, 38(11), 2151-2157. https://pubmed.ncbi.nlm.nih.gov/26407587/
- Korsatko, S., Jensen, L., Brunner, M., Sach-Friedl, S., Tarp, M. D., Holst, A. G., Heller, S. R., & Pieber, T. R. (2018). Effect of once-weekly semaglutide on the counterregulatory response to hypoglycaemia in people with type 2 diabetes: A randomized, placebo-controlled, double-blind, crossover trial. Diabetes, Obesity & Metabolism, 20(11), 2565-2573. https://pubmed.ncbi.nlm.nih.gov/29893488/
- Kulzer, B., Freckmann, G., Ziegler, R., Schnell, O., Glatzer, T., & Heinemann, L. (2024). Nocturnal Hypoglycemia in the Era of Continuous Glucose Monitoring. Journal of Diabetes Science and Technology, 18(5), 1052-1060. https://pubmed.ncbi.nlm.nih.gov/39158988/
- Shah, V. N., Akturk, H. K., Kruger, D., Ahmann, A., Bhargava, A., Bakoyannis, G., Pyle, L., & Snell-Bergeon, J. K. (2025). Semaglutide in Adults with Type 1 Diabetes and Obesity. NEJM Evidence, 4(8), EVIDoa2500173. https://pubmed.ncbi.nlm.nih.gov/40550013/
Written by Kat Fu, M.S., M.S. – Last reviewed: May 2026 – 10 references cited
