“Blue light is bad” has become a repeated claim, but the dose-response data behind that claim rarely gets cited. The relationship between phone screen exposure and melatonin suppression follows a sigmoidal curve with a measurable threshold, and intensity interacts with duration in non-linear ways that change the practical recommendations. This article covers the quantitative dose-response curve (irradiance and duration), age-group differences in melatonin recovery after phone use, why melanopic irradiance is the relevant metric rather than perceived brightness, and what the evidence says about Night Mode and blue-light filters. For an evaluation of blue-light-blocking glasses specifically, see Do Blue Light Glasses Block Sleep-Disrupting Light?. For the broader picture of how circadian rhythm disruption contributes to middle-of-the-night waking, see the Circadian Rhythm Disruption overview: Circadian Sleep Disruption: What Breaks Your Body Clock and Keeps You Awake.
What Is the Dose-Response Curve for Blue Light and Melatonin Suppression?
The first precise dose-response curve for blue LED-induced melatonin suppression came from a study by West and colleagues, who exposed eight healthy young adults (mean age 23.9 years) to varying irradiances of narrowband blue LED light at 469nm between 2:00 and 3:30 AM, while measuring plasma melatonin before and after each exposure (West et al., 2011).
The relationship between irradiance and suppression fit a sigmoidal function (P < 0.0001). A sigmoidal curve means the response is not proportional to the dose — at low irradiances, suppression increases gradually; around the threshold, small irradiance increases produce steep jumps in suppression; and at high irradiances, the curve flattens as suppression approaches its maximum. The calculated half-maximal effective irradiance (ED50) — the irradiance level that produces 50% of maximum suppression — was 14.19 μW/cm².
That 469nm peak wavelength matters for a specific reason: it aligns with the peak sensitivity of melanopsin, the photopigment in intrinsically photosensitive retinal ganglion cells (ipRGCs) that drive non-visual responses to light, including melatonin regulation. When the researchers compared the blue LED to a 4000K broadband white fluorescent source at matched photopic luminance (the measure of perceived brightness), the blue LED produced equal or greater melatonin suppression. That finding established that wavelength composition — rather than perceived brightness — determines circadian impact.
This is directly applicable to consumer screens because modern smartphone and tablet displays use blue-peaked LED backlights with spectral output centered near 469nm. The dose-response parameters from this study transfer to consumer screen exposure estimates.
Does Duration of Screen Use Matter More Than Brightness?
Nagare and colleagues tested 41 participants — adolescents (ages 13-18) and adults (ages 24-55) — under white light at two color temperatures (2700K and 6500K) across a range of intensities (40-1000 lux) and durations (0.5-3.0 hours) during nighttime hours (Nagare et al., 2019).
Light level was a strong predictor of melatonin suppression (F₇,₉₉₄ = 110.1, p < 0.001). Mean suppression rose from 10.9 ± 1.3% after 30 minutes to 29.5 ± 2.1% after three hours of exposure. The 6500K light source — spectrally comparable to modern smartphone and computer displays — produced a mean suppression of 24.7 ± 1.0% across conditions, higher than the warmer 2700K source at matched intensities.
The interaction between intensity and duration was non-linear. At low circadian stimulus levels, achieving meaningful suppression required longer exposure durations. At high intensity, suppression accumulated rapidly, and brief high-brightness sessions produced suppression levels comparable to longer low-brightness sessions. This non-linear interaction has practical implications: dimming a screen to low brightness does reduce per-minute circadian impact, but extending the session can offset that reduction. Conversely, brief periods of high-brightness phone use — checking messages with the screen at full brightness, for example — can produce suppression out of proportion to the session duration.
One finding of note: there was no statistically detectable age difference in suppression magnitude between the adolescent and adult groups (F₁,₃₉ = 0.19, p = 0.67). Both groups were equally vulnerable to melatonin suppression from the same light exposure. The difference between age groups, as the next section covers, is in how quickly melatonin recovers once the light exposure ends.
Do Adults and Adolescents Recover Differently After Phone Use?
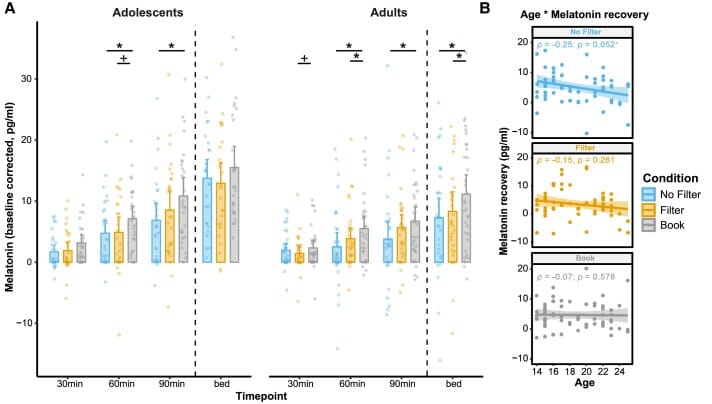
Hohn and colleagues ran a within-subject crossover study with 68 males — 33 adolescents (mean age 15.42 +/- 0.97 years) and 35 young adults (mean age 21.51 +/- 2.06 years) — across three conditions on separate nights: reading on an unfiltered smartphone, a blue-light-filtered smartphone, or a printed book, each for 90 minutes before polysomnographic sleep recording (Hohn et al., 2024).
Both age groups showed melatonin attenuation after unfiltered smartphone reading compared to book reading. The suppression during the 90-minute exposure was present in both groups — consistent with the Nagare et al. finding of equal suppression vulnerability. The divergence appeared in recovery. During the 50-minute window between the end of reading and bedtime, adolescents showed partial melatonin recovery. Adults showed no equivalent recovery by bedtime (p = 0.010).
That absent recovery had downstream consequences for sleep architecture. Only adults showed a reduction in N3 (slow-wave) sleep during the first quarter of the night following unfiltered smartphone use compared to book reading (t₃₂ = -2.38, p = 0.036). Slow-wave sleep in the first sleep quarter is the highest-density period of restorative sleep and is associated with growth hormone release, tissue repair, and memory consolidation processes.
Declarative memory consolidation — measured by word-pair recall — was not affected across light conditions (p = 0.615), and slow oscillation-spindle coupling remained unchanged. Memory encoding appeared resilient at a 90-minute exposure duration.
One finding with direct practical relevance: subjective sleepiness was not meaningfully different across light conditions in either age group, despite the measurable melatonin and sleep architecture differences. Participants did not feel less sleepy after smartphone use, even though their melatonin was suppressed and (in adults) their slow-wave sleep was reduced. Self-report is an unreliable proxy for circadian disruption from evening phone use.
Does Melanopic Irradiance Predict Melatonin Suppression Better Than Screen Brightness?
Schollhorn and colleagues tested 72 healthy males (ages 18-35) across four luminance levels representing typical smartphone, tablet, and computer display brightness, at both high and low melanopic irradiance settings, during a controlled 4-hour pre-bedtime window (Schollhorn et al., 2023).
The distinction between melanopic irradiance and photopic brightness is central. Photopic brightness — measured in candelas per square meter or lux — is weighted to the sensitivity of cone photoreceptors and reflects how bright a display appears to a viewer. Melanopic irradiance is weighted to the sensitivity of melanopsin in ipRGCs, which drive circadian entrainment and melatonin regulation. Two screens can appear equally bright to a viewer while delivering different melanopic irradiance levels, depending on their spectral composition — specifically, how much energy falls near the 480nm melanopsin sensitivity peak.
Under high melanopic irradiance conditions, increasing luminance produced dose-dependent increases in sleep onset latency, dose-dependent reductions in evening melatonin concentration, and dose-dependent delays in melatonin onset time. Under low melanopic conditions at matched photopic brightness, time to sleep onset was shortened and melatonin onset was advanced. Reducing melanopic irradiance improved circadian outcomes without changing how bright the screen appeared.
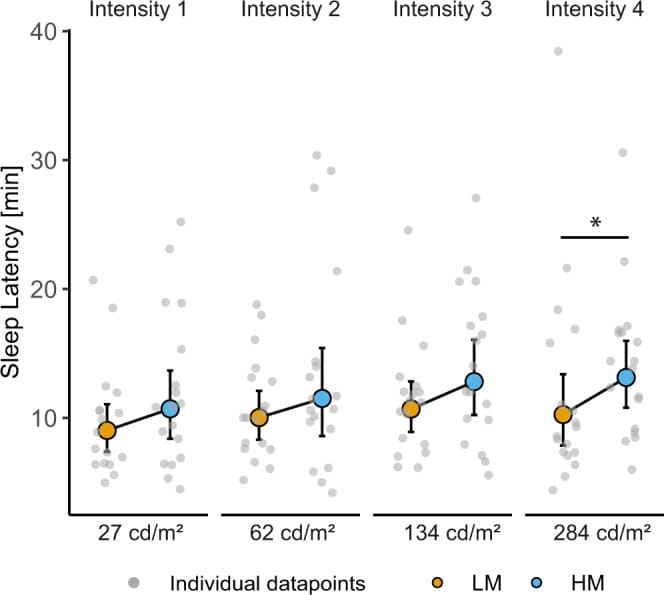
Subjective alertness was lower under low melanopic light, consistent with the dual role of ipRGC-driven melanopsin in both circadian phase regulation and acute arousal. The study applied melanopic equivalent daylight illuminance (melanopic EDI, from the CIE S 026:2018 standard) and empirically validated it as a practical metric for display design and sleep hygiene guidance. This means the relevant specification for Night Mode features is melanopic irradiance reduction — and current Night Mode implementations do not reduce melanopic output proportionally to photopic dimming.
Phone screen exposure at night is one contributor among several. Blue light-induced melatonin suppression may compound with caffeine-driven phase delays, irregular meal timing, or hormonal changes — especially in adults over 40 whose lens transmittance is already declining. Identifying which causes might be involved is a useful next step.
Find out which causes might be driving your 3 AM wakeups →
Do Blue-Light-Blocking Glasses Improve Sleep?
Luna-Rangel and colleagues pooled three randomized controlled crossover trials — 49 total participants — that measured actigraphic sleep outcomes during periods of evening blue-light-blocking glasses use versus control conditions (Luna-Rangel et al., 2025). The effect sizes were uniformly non-detectable: sleep onset latency showed a mean difference of -4.86 minutes (95% CI: -20.23 to 10.52; p = 0.54), total sleep time changed by +8.75 minutes (p = 0.70), sleep efficiency by -0.61 percentage points (p = 0.86), and wake after sleep onset by -1.47 minutes (p = 0.83).
These null findings contrast with the biochemical studies covered above, all of which show measurable melatonin suppression from blue-enriched light. The gap between photoreceptor-level effects and actigraphic sleep architecture changes is the key unresolved question. The authors attributed the null results partly to the small cumulative sample size, short intervention durations, and the limited sensitivity of actigraphy relative to polysomnography or melatonin biomarkers. For a deeper evaluation of lens-type distinctions (amber-tinted vs. untinted filters) and the Cochrane 2023 review, see Do Blue Light Glasses Block Sleep-Disrupting Light?.
Does Blue Light Cause Sustained Suppression or Does Melatonin Recover?
Sanchez-Cano and colleagues exposed 12 healthy adults (7 women, 5 men; ages 19-55; mean 30.08 +/- 12.91 years) to either blue (464nm) or red (631nm) LED light for three hours (9 PM to midnight), with hourly salivary melatonin sampling (Sanchez-Cano et al., 2025). Baseline melatonin was equivalent (blue: 19.5 pg/mL; red: 19.7 pg/mL; p = 1.00).
After the first hour, both wavelengths produced comparable suppression — blue dropped to 6.6 pg/mL and red to 6.8 pg/mL (p = 0.754). Initial suppression at the intensities tested was not wavelength-specific. The divergence emerged at hour 2: blue light maintained suppression at 7.5 pg/mL while red light permitted melatonin to recover to 26.0 pg/mL (p = 0.019). This divergence persisted through hour 3 (blue: 8.3 pg/mL; red: 16.6 pg/mL; p = 0.013).
Men showed a stronger red-vs-blue divergence at two hours (p = 0.043). Younger participants demonstrated stronger blue-specific sustained suppression than older participants. The practical implication: the duration of blue light exposure matters for whether melatonin can begin its recovery trajectory during exposure. Extended blue-light phone use prevents the hormonal recovery that would occur under longer-wavelength light sources.
Is Night Mode or a Blue-Light Filter App Enough to Prevent Melatonin Suppression?
Heo and colleagues conducted the only double-blind RCT comparing conventional blue-light smartphones to blue-light-suppressed devices — 22 healthy adults used each device for 150 continuous minutes (7:30-10:00 PM) across two separate 3-day inpatient admissions (Heo et al., 2017).
Conventional blue-light smartphones decreased subjective sleepiness compared to the filtered devices (Cohen’s d = 0.49, p = 0.04) and increased commission errors on a sustained attention task (Cohen’s d = -0.59, p = 0.02). The alertness-promoting and attention-impairing effects of blue light were reduced by the filter. DLMO was delayed by approximately 14 minutes more under the conventional device (2.94 versus 2.70 hours post-lights-on) — directionally consistent with a circadian phase delay, though not statistically detectable at n = 22.
Blue-light filters reduce circadian impact but do not eliminate it. This is consistent with the melanopic irradiance findings from Schollhorn et al.: software Night Mode does not reduce melanopic output proportionally to photopic dimming. The governing variable is melanopic irradiance, and current filter implementations address brightness and color temperature without directly targeting the melanopsin-weighted spectral range.
Related Reading
- Circadian Rhythm Disruption: How a Misaligned Internal Clock Fragments Sleep, Drives 3AM Wakeups, and Reduces Sleep Depth — the full overview of circadian sleep disruption, including melatonin, cortisol, body temperature, peripheral clocks, and early-morning waking
- Why Does Melatonin Work for Jet Lag but Not Chronic Insomnia? — why melatonin works as a timing cue, not a general sleep-depth fix
- Is Your Pineal Gland Calcifying? How Age-Related Melatonin Production Loss Differs From Receptor Decline — how pineal calcification, melatonin output, and receptor decline diverge with age
- How Many Melanopic Lux Do You Need for Circadian Entrainment? — how melanopic light exposure sets the clock more directly than ordinary lux
- Do Blue Light Glasses Help You Sleep? Amber vs Standard Lenses — What Studies Show — how amber lenses, clear lenses, and evening screens differ in sleep studies
- How Long Does It Take to Reset Your Circadian Rhythm? — why the reset timeline differs for the SCN, peripheral clocks, and behavioral cues
- How Does Caffeine Delay Your Circadian Clock by 40 Minutes? — how caffeine delays circadian phase through adenosine and clock-gene timing
References
1. West, K. E., Jablonski, M. R., Warfield, B., Cecil, K. S., James, M., Ayers, M. A., Maida, J., Bowen, C., Sliney, D. H., Rollag, M. D., Hanifin, J. P., & Brainard, G. C. (2011). Blue light from light-emitting diodes elicits a dose-dependent suppression of melatonin in humans. Journal of Applied Physiology, 110(3), 619-626. https://pubmed.ncbi.nlm.nih.gov/21164152/
2. Nagare, R., Rea, M. S., Plitnick, B., & Figueiro, M. G. (2019). Nocturnal melatonin suppression by adolescents and adults for different levels, spectra, and durations of light exposure. Journal of Biological Rhythms, 34(2), 178-194. https://pubmed.ncbi.nlm.nih.gov/30803301/
3. Hohn, C., Hahn, M. A., Gruber, G., Pletzer, B., Cajochen, C., & Hoedlmoser, K. (2024). Effects of evening smartphone use on sleep and declarative memory consolidation in male adolescents and young adults. Brain Communications, 6(3), fcae173. https://pubmed.ncbi.nlm.nih.gov/38846535/
4. Schollhorn, I., Stefani, O., Lucas, R. J., Spitschan, M., Slawik, H. C., & Cajochen, C. (2023). Melanopic irradiance defines the impact of evening display light on sleep latency, melatonin and alertness. Communications Biology, 6(1), 228. https://pubmed.ncbi.nlm.nih.gov/36854795/
5. Sanchez-Cano, A., Luesma-Bartolome, M. J., Solanas, E., & Orduna-Hospital, E. (2025). Comparative effects of red and blue LED light on melatonin levels during three-hour exposure in healthy adults. Life, 15(5), 715. https://pubmed.ncbi.nlm.nih.gov/40430143/
6. Luna-Rangel, F. A., Gonzalez-Bedolla, B., Salazar-Ortega, M. J., Torres-Mancilla, X. M., & Martinez-Cadena, S. (2025). Efficacy of blue-light blocking glasses on actigraphic sleep outcomes: A systematic review and meta-analysis of randomized controlled crossover trials. Frontiers in Neurology, 16, 1699303. https://pubmed.ncbi.nlm.nih.gov/41341515/
7. Heo, J. Y., Kim, K., Fava, M., Mischoulon, D., Papakostas, G. I., Kim, M. J., Kim, D. J., Chang, K. J., Oh, Y., Yu, B. H., & Jeon, H. J. (2017). Effects of smartphone use with and without blue light at night in healthy adults: A randomized, double-blind, cross-over, placebo-controlled comparison. Journal of Psychiatric Research, 87, 61-70. https://pubmed.ncbi.nlm.nih.gov/28017916/
Written by Kat Fu, M.S., M.S. ? Last reviewed: May 2026 ? 7 references cited
