Discussions of testosterone and sleep tend to focus on the bidirectional relationship: poor sleep lowers testosterone, and low testosterone worsens sleep. Both are true. But there is a molecular bridge between these two that rarely comes up — neurosteroids.
Testosterone-derived neurosteroids are positive modulators of GABA-A receptors, the receptors that maintain stable sleep through the night. When testosterone declines, the neurosteroid supply declines with it, and GABAergic tone — the brain’s ability to sustain inhibitory activity during sleep — weakens.
This article covers the testosterone-to-androstanediol-to-GABA-A receptor pathway, what happens to GABAergic tone as testosterone declines with age, and why this creates a compound problem for sleep in aging men. For GABA supplementation, see the separate article on [whether GABA supplements help you stay asleep through the night](). For the broader testosterone-sleep picture, see Why Men’s Hormones Disrupt Sleep.
What Is the Relationship Between GABA and Testosterone?
How does testosterone become a GABA modulator?
The pathway is sequential: testosterone is first reduced by 5-alpha-reductase to dihydrotestosterone (DHT), and then by 3-alpha-hydroxysteroid dehydrogenase (3-alpha-HSD) to androstanediol (5-alpha-androstan-3-alpha,17-beta-diol). Androstanediol is the neuroactive endpoint — the molecule that acts on GABA-A receptors.
In dissociated hippocampal neurons, Reddy and Jian (2010) found that androstanediol potentiated GABA-activated chloride currents with an EC50 of 5 micromolar. At 1 micromolar, androstanediol produced approximately 50% potentiation of GABA responses. In practical terms: even at low concentrations, this testosterone metabolite measurably enhances the brain’s primary inhibitory neurotransmitter.
The effect is stereospecific. Only the 3-alpha-epimer of androstanediol is active. The 3-beta-epimer produced no effect at any concentration tested (Reddy & Jian, 2010). This is precise molecular recognition, not a vague hormonal association — the GABA-A receptor distinguishes between mirror-image forms of the same molecule.
Androstanediol has minimal affinity for androgen receptors, which means its effects in the brain are GABAergic rather than androgenic (Reddy & Jian, 2010). This is an important distinction: androstanediol’s role in the brain is to modulate GABA-A receptors, not to activate testosterone-related gene expression.
Where does androstanediol bind on the GABA-A receptor?
Legesse et al. (2023) used cryo-EM — electron microscopy at near-atomic resolution — to identify where neurosteroids bind on GABA-A receptors. Positive modulators like allopregnanolone (the progesterone-derived equivalent of androstanediol) bind at the transmembrane domain, at the interface between the receptor protein and the surrounding lipid membrane. Molecular dynamics simulations showed that this binding increases pore hydration and reduces the energy barrier for chloride ions to pass through, which is the physical basis for enhanced inhibitory activity (Legesse et al., 2023).
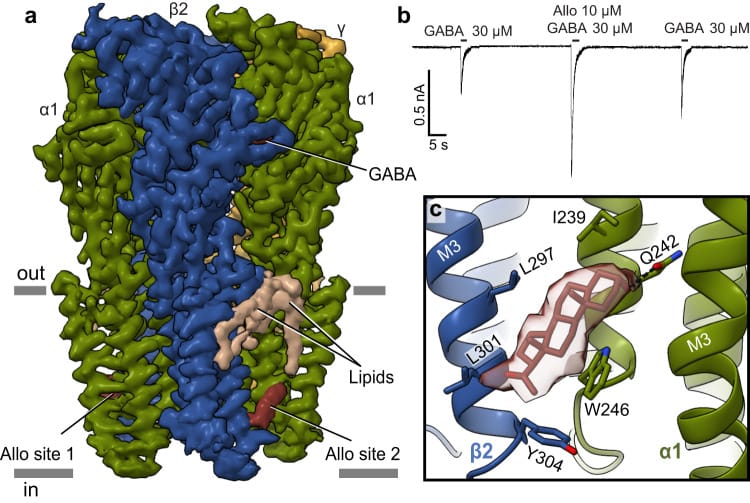
Because testosterone-derived androstanediol shares the same 3-alpha-hydroxyl steroid backbone as allopregnanolone, it is predicted to occupy the same binding site (Maguire & Mennerick, 2024). The implication: men with higher testosterone have higher androstanediol production, which translates to stronger baseline GABA-A receptor modulation — and, by extension, more stable inhibitory tone during sleep.
Does GABA Affect Hormone Levels in Men?
Does GABA regulate testosterone production?
The hypothalamus releases GnRH in pulses. Those pulses trigger luteinizing hormone (LH) from the pituitary, and LH stimulates testosterone synthesis in the testes. GABAergic neurons participate in the regulation of that GnRH pulse generator (Maguire & Mennerick, 2024). This means GABA has an indirect role in testosterone production — not by stimulating it directly, but by helping modulate the upstream hormonal cascade.
Some supplement marketing positions oral GABA as a way to raise testosterone. The direct evidence for this is limited. The more established pathway is indirect: if GABA supports sleep quality — and slow-wave sleep in particular — then it may support testosterone production through sleep architecture rather than through a direct hormonal mechanism.
Does slow-wave sleep affect testosterone production?
Ukraintseva et al. (2018) tested this directly. In a randomized crossover study of 12 healthy men, they used acoustic stimulation to selectively suppress slow-wave sleep (SWS) by 54.2% on one night, without changing total sleep time, sleep efficiency, or time in other sleep stages. The result: morning salivary testosterone declined on SWS-suppressed nights compared to control nights (p = 0.017).
The upstream androgen precursor 17-alpha-hydroxyprogesterone also declined (p = 0.011), while cortisol, androstenedione, and DHEA did not change. This indicates the testosterone reduction was tied to the slow-wave sleep disruption — not a general stress response from poor sleep.
The logic follows: if GABA supports slow-wave sleep (it does — GABA-A receptor activation is the primary mechanism generating slow oscillations during NREM sleep), and slow-wave sleep supports nocturnal testosterone synthesis (the Ukraintseva data confirms this), then adequate GABAergic tone indirectly supports testosterone production through sleep architecture.
This is the more honest framing of the GABA-testosterone relationship: GABA does not directly stimulate testosterone. It supports the sleep stage during which the largest nocturnal testosterone pulse occurs.
Does Declining Testosterone Reduce GABAergic Sleep Protection in Men?
What does the population data show?
Barrett-Connor et al. (2008) measured serum testosterone in 1,312 community-dwelling men and then assessed sleep quality approximately 3.4 years later using both wrist actigraphy and in-laboratory polysomnography. Men with lower total testosterone had lower sleep efficiency, a greater number of nocturnal awakenings, and less time in slow-wave sleep. The relationship was dose-dependent: the lower the testosterone, the worse the sleep architecture.
Testosterone levels showed no independent relationship to total sleep duration. The hormone’s association was with sleep quality and architecture — the depth and continuity of sleep, not how long men stayed in bed.
One important finding: the associations between low testosterone and reduced sleep quality were attenuated after adjusting for body mass index and waist circumference (Barrett-Connor et al., 2008). Visceral adipose tissue contains aromatase, the enzyme that converts testosterone to estradiol. More visceral fat means more testosterone is diverted toward estradiol and away from the androstanediol pathway — reducing the neurosteroid supply that modulates GABA-A receptors. Adiposity and testosterone decline may compound each other’s effects on GABAergic sleep protection.
The NHANES data (Hernandez-Perez et al., 2024; n = 8,748) added age-stratified nuance. Among middle-aged men (41-64 years), extended sleep of nine or more hours was associated with low testosterone (OR = 2.03; 95% CI: 1.10-3.73), consistent with excessive sleep as a marker of underlying metabolic or endocrine disruption. The relationship between sleep and testosterone is not linear — it varies by age and context.
Does testosterone directly cause sleep architecture changes?
Cross-sectional data shows correlation. The Morssinkhof et al. (2023) study provides causal evidence.
This prospective study enrolled 73 transgender individuals — 38 transmasculine participants beginning testosterone therapy and 35 transfeminine participants beginning estrogen/antiandrogen therapy. Sleep was recorded across seven nights using ambulatory EEG before and after three months of hormone therapy.
In transmasculine participants receiving testosterone, slow-wave sleep duration decreased by a mean of 7 minutes (95% CI: -12 to -3) and by 1.7 percentage points of total sleep time. REM sleep latency decreased by 39% (95% CI: -52% to -22%), and REM sleep duration increased by a mean of 17 minutes (95% CI: 7-26). These changes recapitulated the lower-SWS, earlier-REM pattern characteristic of cisgender male sleep.
Transfeminine participants receiving estrogen and antiandrogen therapy showed no statistically detectable changes in any sleep architecture parameter after three months.
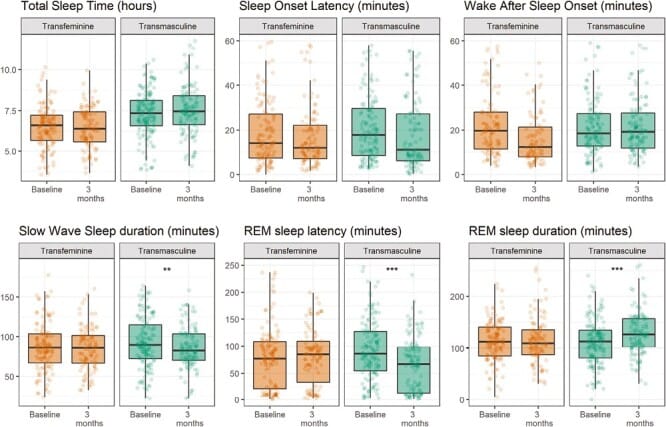
The asymmetry is notable. Testosterone produced measurable, directionally consistent changes in sleep architecture within three months. Estrogen did not. This argues that androgens exert a direct influence on GABAergic sleep-regulatory circuits — consistent with the androstanediol-GABA-A pathway described above (Morssinkhof et al., 2023).
What is the compound problem for aging men?
Here is where the pieces converge into a compound problem:
1. Testosterone declines approximately 1-2% per year from around age 30, compounding over decades.
2. Less testosterone means less androstanediol — the neurosteroid that modulates GABA-A receptors.
3. Weaker GABA-A modulation reduces the brain’s ability to maintain stable inhibitory tone during sleep, impairing slow-wave sleep and sleep continuity.
4. Reduced slow-wave sleep lowers nocturnal testosterone synthesis (Ukraintseva et al., 2018).
5. Lower testosterone production feeds back into step 2.
Each side of this loop can accelerate the other. And adiposity amplifies the problem: visceral fat diverts testosterone toward estradiol via aromatase, reducing the androstanediol supply further.
For men experiencing sleep maintenance insomnia after 40, this testosterone-GABA compound mechanism may be contributing — alongside, or instead of, the explanations that are more commonly discussed.
The testosterone-GABA connection is one piece of a larger picture. Cortisol disruption, metabolic instability, inflammation, and circadian misalignment might all be compounding the hormonal sleep disruption. Men experiencing sleep maintenance insomnia after 40 often have more than one of these mechanisms at work — and the combination looks different from person to person.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Does GABA Increase Testosterone?
Can GABA Supplements Help Testosterone Production?
What Supplements Support Both GABA and Testosterone?
Does GABA Deficiency Affect Men Differently Than Women?
Related Reading
- Hormonal Sleep Disruption in Men — How testosterone, cortisol, growth hormone, and GABA interact to fragment sleep in men
- Can Low GABA Cause Waking Up at 3am? — The GABAergic mechanism behind 3am waking
- What Are the Signs of Low GABA at Night? — How to recognize impaired GABAergic function
- Do GABA Supplements Help You Stay Asleep Through the Night? — Trial evidence for oral GABA and the blood-brain barrier question
- How Do You Increase GABA Levels Naturally for Better Sleep? — Exercise, yoga, fermented foods, and gut health for GABA support
- Does Low Testosterone Cause Sleep Problems in Men? — The bidirectional testosterone-sleep feedback loop
- Can a Cortisol Spike Wake You Up at 3am? — Cortisol-testosterone axis and nocturnal waking
- Does Growth Hormone Decline Affect Your Sleep After 40? — GH-SWS coupling and age-related decline
- Does Andropause Cause Insomnia? What Men Over 50 Need to Know — Compound hormonal decline in aging men
- Can Belly Fat Lower Your Testosterone and Disrupt Your Sleep? — How visceral fat suppresses testosterone through aromatase and disrupts sleep
- Can Ultra-Processed Food Lower Testosterone and Disrupt Sleep? — How endocrine-disrupting chemicals in processed food suppress testosterone and degrade sleep
- Can Inflammation Suppress Testosterone and Disrupt Your Sleep? — How inflammatory cytokines suppress testosterone production at the brain and testicular level
- Does Testosterone Replacement Therapy Affect Sleep? — How TRT affects sleep quality, its interaction with sleep apnea, and the role of body weight
References
1. Reddy DS, Jian K. (2010). The testosterone-derived neurosteroid androstanediol is a positive allosteric modulator of GABAA receptors. Journal of Pharmacology and Experimental Therapeutics, 334(3), 1031-1041. PubMed
2. Ukraintseva YV, et al. (2018). Slow-wave sleep and androgens: selective slow-wave sleep suppression affects testosterone and 17-alpha-hydroxyprogesterone secretion. Sleep Medicine, 48, 117-122. PubMed
3. Barrett-Connor E, et al. (2008). The association of testosterone levels with overall sleep quality, sleep architecture, and sleep-disordered breathing. Journal of Clinical Endocrinology & Metabolism, 93(7), 2602-2609. PubMed
4. Morssinkhof MWL, et al. (2023). Influence of sex hormone use on sleep architecture in a transgender cohort. Sleep, 46(12), zsad271. PubMed
5. Legesse DH, et al. (2023). Structural insights into opposing actions of neurosteroids on GABAA receptors. Nature Communications, 14, 5091. PubMed
6. Maguire JL, Mennerick S. (2024). Neurosteroids: mechanistic considerations and clinical prospects. Neuropsychopharmacology, 49(1), 73-82. PubMed
7. Hernandez-Perez JG, et al. (2024). Association of sleep duration and quality with serum testosterone concentrations among men and women: NHANES 2011-2016. Andrology, 12(3), 456-467. PubMed
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
