Belly fat and lower testosterone often appear together in men over 40 — and they are connected through an enzyme pathway centered on aromatase. Aromatase, produced in visceral fat cells, converts circulating testosterone to estradiol. The more visceral fat a man carries, the faster this conversion runs. This connection matters because the same hormonal change that drives fat accumulation also affects sleep architecture, and the sleep loss feeds back into more fat gain. This article covers the aromatase-driven mechanism by which visceral fat lowers testosterone, how poor sleep feeds that cycle, and what the evidence says about reversibility. For the broader landscape of hormonal sleep changes, see Hormonal Sleep Disruption in Men. Belly fat is one of several factors that can lower testosterone and affect sleep — others include cortisol dysregulation, inflammatory cytokines, and GABA deficiency.
Does Belly Fat Lower Testosterone in Men?
The enzyme at the center of this relationship is aromatase (CYP19A1), which is produced in visceral fat cells and converts circulating testosterone to estradiol. More visceral fat means more aromatase, which means faster testosterone depletion. A 2025 study in the Journal of Clinical Endocrinology & Metabolism examined adipose tissue from 46 men stratified by BMI and found that aromatase mRNA and protein expression were elevated in obese men compared to lean controls (Ahmed et al., 2025). At the same time, estrogen receptor alpha (ESR1) expression was lower in obese men, and the ESR1-to-ESR2 ratio was reduced — meaning that even as estradiol production accelerated, the tissue’s ability to respond to estradiol was blunted. The combined effect: more estradiol being produced, with less receptor sensitivity to manage it.
Rising estradiol from aromatase activity suppresses the hypothalamic-pituitary-gonadal (HPG) axis. Estradiol feeds back to the hypothalamus, reducing gonadotropin-releasing hormone (GnRH) output. Lower GnRH leads to lower luteinizing hormone (LH) from the pituitary, which reduces Leydig cell stimulation in the testes — and therefore less testosterone production.
The scale of this relationship is documented in a cross-sectional analysis of 1,551 American men from NHANES 2013-2016 (Su et al., 2023). Researchers used the visceral adiposity index (VAI), a composite marker integrating waist circumference, BMI, triglycerides, and HDL, to quantify visceral fat burden. Men in the highest VAI quartile had testosterone concentrations 94.59 ng/dL lower than men in the lowest quartile. Their risk of testosterone deficiency was 5.07 times greater. Across all quartiles, each incremental rise in VAI was associated with a 1.24-fold increase in testosterone deficiency risk. The inverse association was strongest among older and obese men, pointing to compounding between age-related HPG decline and aromatase-mediated conversion.
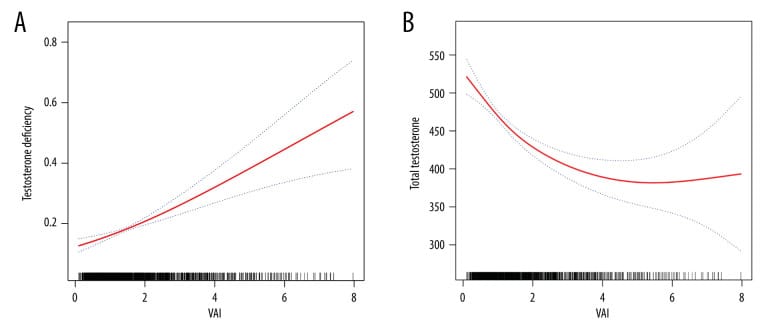
A UK Biobank study of 16,237 men showed consistent results at a larger scale, with 20-30% testosterone differences across adiposity quintiles (Hynes et al., 2024). That study also measured muscle fat infiltration (MFI) — fat deposited within skeletal muscle — and found that MFI had a steeper inverse association with free testosterone than adiposity measures like BMI or waist circumference. Fat quality and distribution, not only total fat mass, influence testosterone status.
The distinction between visceral and subcutaneous fat matters here. Subcutaneous fat — the fat beneath the skin — carries less aromatase activity. The visceral compartment, which surrounds the abdominal organs, is where aromatase upregulation concentrates, and where the negative feedback on the HPG axis originates.
How Does Poor Sleep Increase Visceral Fat?
A cross-sectional analysis of 11,252 NHANES participants (2007-2018) quantified the relationship between habitual sleep duration and visceral fat (Liu et al., 2024). Using the visceral adiposity index, researchers found an L-shaped dose-response curve: VAI increased as sleep duration fell below 7.5 hours per night. Above 7.5 hours, additional sleep did not produce further VAI reductions. The relationship was not linear — there was a threshold effect at 7.5 hours, below which visceral fat accumulation accelerated.
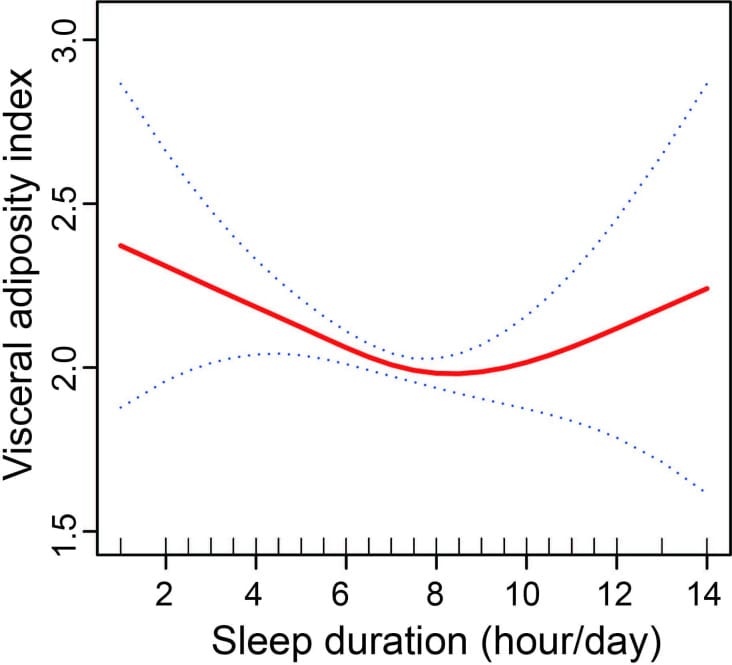
Several mechanisms explain why short sleep promotes visceral fat gain. Cortisol, which tends to rise with sleep deprivation, preferentially directs fat storage to the visceral compartment. Glucocorticoid receptors are denser in visceral adipose tissue than in subcutaneous fat, making the abdominal region the primary depot for stress-driven fat accumulation. Sleep loss also disrupts appetite-regulating hormones: ghrelin rises and leptin falls, increasing drive toward calorie-dense food. Circadian disruption from irregular or shortened sleep impairs lipolysis (fat breakdown) in abdominal depots, meaning visceral fat becomes harder to mobilize.
The testosterone connection compounds this. In a study of 10 healthy young men, one week of sleep restriction to 5 hours per night reduced daytime testosterone by 10-15% (Leproult & Van Cauter, 2011). That testosterone reduction itself promotes easier visceral fat gain, because testosterone helps maintain lean mass and counteract visceral accumulation. Short sleep therefore contributes from two directions: it promotes visceral fat storage through cortisol and appetite changes, and it lowers testosterone — the hormone that would otherwise counteract that fat gain.
For a deeper look at how cortisol from poor sleep promotes abdominal fat storage, see Can a Cortisol Spike Wake You Up at 3am?.
Why Do Men Gain Belly Fat and Sleep Worse After 40?
The sleep-testosterone relationship differs by age in ways that are not intuitive. A population study of 8,748 adults from NHANES 2011-2016 found that in young men (ages 20-40), short sleep was paradoxically associated with higher testosterone — possibly reflecting acute stress-driven hormonal responses or morning sampling timing relative to the nocturnal testosterone peak (Hernandez-Perez et al., 2024). In middle-aged men (ages 41-64), extended sleep duration (9+ hours) was associated with lower testosterone, a pattern likely reflecting underlying conditions such as obesity or sleep apnea that simultaneously drive excess sleep need and HPG axis suppression.
These age-stratified data show that the sleep-testosterone relationship changes across the lifespan. In younger men, acute sleep restriction may trigger short-term compensatory hormonal responses. In older men, the HPG axis is already declining, and the body’s capacity to compensate narrows.
The compounding effect works like this: natural age-related testosterone reduction of 1-2% per year makes visceral fat accumulation easier. As visceral fat expands, aromatase activity increases, converting more testosterone to estradiol. This aromatase-driven testosterone loss adds to the age-related change — a 50-year-old man is losing testosterone through both HPG axis changes and aromatase conversion simultaneously.
The NHANES VAI-testosterone data (Su et al., 2023) support this compounding: the inverse association between visceral fat and testosterone was strongest among older and obese men. Age-related decline and aromatase-mediated conversion reinforce each other. Each one accelerates the other.
Sleep architecture also changes with age. Slow-wave sleep — the deep sleep phase during which the largest nocturnal testosterone pulses occur — decreases across the lifespan. Less slow-wave sleep means less nocturnal testosterone production, which means less hormonal resistance to visceral fat gain. For more on how age-related hormonal changes interact with sleep, see Does Andropause Cause Insomnia? and Does Growth Hormone Decline Affect Your Sleep After 40?.
Does Losing Belly Fat Increase Testosterone?
The aromatase pathway described throughout this article — aromatase in visceral fat converting testosterone to estradiol, which suppresses GnRH and LH — is proportional to fat mass. Less visceral fat means less aromatase activity, which means less testosterone-to-estradiol conversion. As estradiol levels fall, the negative feedback on the hypothalamus diminishes, GnRH and LH output can recover, and Leydig cells receive more stimulation to produce testosterone.
The NHANES data (Su et al., 2023) show that men in the lowest VAI quartile have the highest testosterone levels. While this cross-sectional data cannot prove causation, the dose-response relationship — each quartile step in VAI corresponds to a measurable testosterone change — is consistent with the mechanistic pathway: if a man moves from high to low visceral adiposity, the aromatase-mediated testosterone suppression should diminish.
Body recomposition — fat loss combined with muscle preservation — may compound the hormonal benefit. The UK Biobank data (Hynes et al., 2024) showed that lean tissue volume was positively associated with both IGF-1 and free testosterone. Reducing visceral fat while maintaining muscle mass addresses aromatase-mediated testosterone loss and supports hormonal status through preserved lean mass.
Improving sleep may reinforce visceral fat reduction. The 10-15% testosterone suppression documented in the Leproult & Van Cauter (2011) sleep restriction study was observed after one week of 5-hour sleep in young men — indicating that the suppression is linked to sleep duration rather than permanent damage. Restoring adequate sleep both preserves nocturnal testosterone production and reduces the cortisol elevation and appetite disruption that promote visceral fat accumulation. Sleep improvement and visceral fat reduction address different parts of the feedback loop: sleep normalization reduces the hormonal environment that promotes fat storage, and fat reduction removes the aromatase activity that suppresses testosterone.
Aromatase-driven testosterone suppression from belly fat often compounds with cortisol dysregulation, inflammatory cytokines, and circadian changes — each capable of independently lowering testosterone and fragmenting sleep. For a closer look at how testosterone loss affects sleep architecture, see Does Low Testosterone Cause Sleep Problems in Men?.
Belly fat rarely acts alone. The aromatase-driven testosterone suppression covered in this article often compounds with cortisol dysregulation, inflammatory cytokines, and circadian changes. Identifying which contributors might be active is a useful next step, because each one can independently lower testosterone and fragment sleep.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Does Low Testosterone Cause Belly Fat in Men?
The sections above cover the fat-to-testosterone direction — how visceral fat suppresses testosterone through aromatase. But the reverse direction is equally relevant. When testosterone declines, lean muscle mass decreases, and the metabolic rate that muscle supports drops with it. Lower testosterone also changes how and where the body stores fat, favoring the visceral compartment.
This creates a “which came first” question, but the feedback loop matters more than the entry point. The UK Biobank data (Hynes et al., 2024) showed that lean tissue volume was positively associated with free testosterone — men with more muscle tended to have higher testosterone. Whether a man enters the loop through lower testosterone (age-related or otherwise) or through visceral fat gain (from sedentary behavior, poor sleep, or dietary factors), the aromatase feedback accelerates the process in the same direction.
How Does Cortisol from Poor Sleep Affect Weight Gain?
The reason cortisol from poor sleep promotes abdominal fat gain — rather than overall fat gain — comes down to receptor density. Visceral adipose tissue has a higher concentration of glucocorticoid receptors than subcutaneous fat. When cortisol rises, these receptors are activated preferentially, directing fat storage to the abdominal cavity. The NHANES sleep-VAI data (Liu et al., 2024) showed that the visceral adiposity index increased as sleep fell below 7.5 hours, consistent with this cortisol-driven mechanism.
For the full cortisol-sleep-testosterone connection, including how cortisol patterns change across the night, see Can a Cortisol Spike Wake You Up at 3am?.
Does Visceral Fat Cause Insomnia?
The pathways from visceral fat to sleep disruption are indirect but well-documented. Visceral fat increases the risk of obstructive sleep apnea, which fragments sleep through repeated airway obstruction and arousal. Visceral adipose tissue also produces inflammatory cytokines — including TNF-alpha and IL-6 — that can independently fragment sleep architecture. And the aromatase-driven testosterone suppression described throughout this article impairs sleep maintenance, since testosterone supports consolidated sleep in men.
These pathways help explain why men with high visceral adiposity often report both poor sleep quality and daytime fatigue — even when they believe they are sleeping an adequate number of hours. The sleep fragmentation from apnea, inflammation, and testosterone suppression can degrade sleep quality without reducing total sleep time (Su et al., 2023).
Can You Lose Hormonal Belly Fat Without Testosterone Replacement?
The aromatase pathway described in this article is proportional to fat mass — reducing visceral fat reduces aromatase activity, which allows endogenous testosterone production to recover without exogenous replacement. The feedback loop can be interrupted at the sleep end as well: the NHANES sleep-VAI data (Liu et al., 2024) identified 7.5 hours as the threshold below which visceral adiposity increased. Normalizing sleep above this threshold can reduce the cortisol elevation and appetite hormone disruption that drive visceral fat gain.
The testosterone suppression from short sleep also responds to sleep normalization. The 10-15% testosterone reduction documented in the Leproult & Van Cauter (2011) sleep restriction study occurred after one week of 5-hour sleep — this reflects duration-dependent suppression, not permanent change. Restoring adequate sleep removes the acute suppression and allows the nocturnal testosterone peak to return. Combining sleep improvement with visceral fat reduction addresses the feedback loop at two points: sleep normalization reduces the hormonal environment that promotes fat storage, and fat reduction removes the aromatase activity that suppresses testosterone.
Related Reading
- Hormonal Sleep Disruption in Men — How testosterone, cortisol, growth hormone, and other hormones disrupt sleep in men
- Does Low Testosterone Cause Sleep Problems in Men? — How declining testosterone fragments sleep architecture in men
- Can a Cortisol Spike Wake You Up at 3am? — The cortisol-testosterone axis and nocturnal arousal patterns
- Does Growth Hormone Decline Affect Your Sleep After 40? — How age-related growth hormone loss disrupts slow-wave sleep
- Does Andropause Cause Insomnia? What Men Over 50 Need to Know — Compound hormonal aging and sleep disruption in men over 50
- Can Low GABA Cause Waking Up at 3am? — How insufficient GABA fails to maintain sleep through the night
- What Are the Signs of Low GABA at Night? — Recognizing GABA-related indicators that disrupt sleep
- Do GABA Supplements Help You Stay Asleep Through the Night? — Evaluating GABA supplementation evidence for sleep maintenance
- Does GABA Affect Testosterone and Sleep in Men? — The GABA-testosterone connection in male sleep disruption
- How Do You Increase GABA Levels Naturally for Better Sleep? — Lifestyle and dietary approaches to support GABA function
- Can Ultra-Processed Food Lower Testosterone and Disrupt Sleep? — How endocrine-disrupting chemicals in processed food suppress testosterone and degrade sleep
- Can Inflammation Suppress Testosterone and Disrupt Your Sleep? — How inflammatory cytokines suppress testosterone production at the brain and testicular level
- Does Testosterone Replacement Therapy Affect Sleep? — How TRT affects sleep quality, its interaction with sleep apnea, and the role of body weight
References
1. Ahmed, F., et al. (2025). Altered expression of aromatase and estrogen receptors in adipose tissue from men with obesity or type 2 diabetes. Journal of Clinical Endocrinology & Metabolism, 110(10), e3410-e3424. https://pubmed.ncbi.nlm.nih.gov/39833659/
2. Hernandez-Perez, J. G., et al. (2024). Association of sleep duration and quality with serum testosterone concentrations among men and women: NHANES 2011-2016. Andrology, 12(3), 518-526. https://pubmed.ncbi.nlm.nih.gov/37452666/
3. Hynes, M. C., et al. (2024). Associations of body composition measures with circulating insulin-like growth factor-I, testosterone, and sex hormone-binding globulin concentrations in 16,000 men. International Journal of Obesity, 48(12), 1809-1817. https://pubmed.ncbi.nlm.nih.gov/39433891/
4. Leproult, R., & Van Cauter, E. (2011). Effect of 1 week of sleep restriction on testosterone levels in young healthy men. JAMA, 305(21), 2173-2174. https://pubmed.ncbi.nlm.nih.gov/21632481/
5. Liu, J., et al. (2024). Association of sleep duration with Visceral Adiposity Index: A cross-sectional study based on the NHANES 2007-2018. BMJ Open, 14(7), e082601. https://pubmed.ncbi.nlm.nih.gov/39019627/
6. Su, M., et al. (2023). The impact of visceral adiposity on testosterone levels in American adult men: A cross-sectional analysis. Medical Science Monitor, 29, e941394. https://pubmed.ncbi.nlm.nih.gov/37634076/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 6 references cited
