Intermittent hypoxia from obstructive sleep apnea suppresses circadian clock genes — BMAL1, PER, and CRY — through the HIF-1alpha pathway. CPAP corrects the breathing, but the clock gene damage persists. Studies measuring clock gene expression in CPAP-treated individuals show that one night of CPAP produces no change in BMAL1, and even two years of CPAP does not fully restore clock gene oscillation patterns (Gaspar et al., 2021; Gabryelska et al., 2025). The residual exhaustion is a circadian recovery problem, not a CPAP failure.
Approximately 30 percent of people using CPAP for obstructive sleep apnea have objectively impaired alertness even after their apnea-hypopnea index is under control (Tankere et al., 2025). The breathing is fixed. The tiredness persists. This article covers how intermittent hypoxia damages circadian clock genes through HIF-1alpha, why different organs recover at different rates, and what drives residual daytime sleepiness despite adequate CPAP use.
?-
How Does Intermittent Hypoxia Damage Your Circadian Clock Genes?
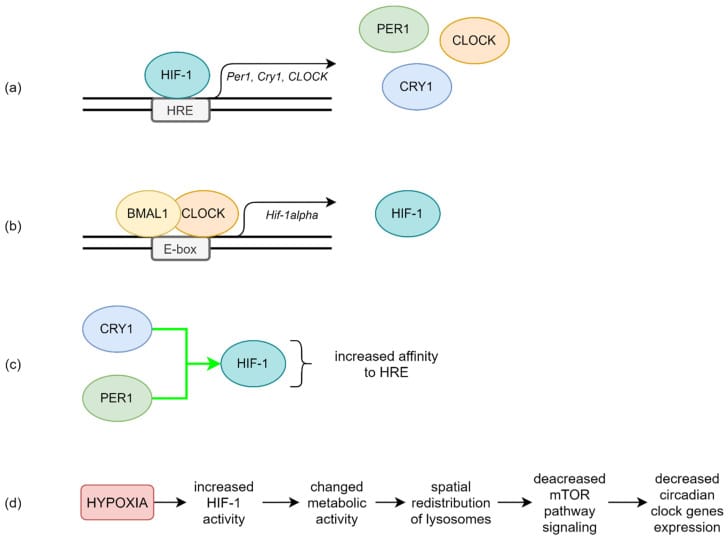
Intermittent hypoxia activates HIF-1alpha — a transcription factor that responds to low oxygen. HIF-1alpha interacts with the molecular clock at multiple points: it induces PER2 and CRY1 expression through hypoxia response elements, and it competes with BMAL1 for binding sites on E-box regulatory regions (O’Connell et al., 2020). In people with obstructive sleep apnea, HIF-1alpha is chronically elevated, and the normal circadian oscillation of clock genes including BMAL1 may be disrupted (Xie et al., 2022; Gabryelska et al., 2022).
Oxygen is a timing input for the circadian clock. In a 2017 study published in Cell Metabolism, Adamovich et al. demonstrated that oxygen cycles synchronize cellular clocks through HIF-1alpha. When mice in this animal model were exposed to moderate oxygen fluctuations, the oxygen cycles accelerated circadian phase re-entrainment — but this effect depended on HIF-1alpha. Mice genetically lacking HIF-1alpha did not respond to the oxygen timing input (Adamovich et al., 2017). This established that oxygen is a circadian zeitgeber, a time-giver that can reset or alter clock timing.
The connection between HIF-1alpha and the clock runs in both directions. O’Connell et al. (2020) described how HIF-1alpha directly induces the clock genes PER2 and CRY1 by binding to hypoxia response elements (HREs) in their promoters. At the same time, HIF-1alpha co-localizes with BMAL1 on E-box regulatory regions, meaning the two transcription factors compete for the same DNA binding sites. The interaction is also time-of-day gated — the clock controls when HIF-1alpha can be induced, and HIF-1alpha in turn alters clock gene expression. CLOCK and BMAL1 drive the expression of the HIF1A gene itself, creating a bidirectional feedback loop.
In people with obstructive sleep apnea, this feedback loop is chronically overactivated. Research has shown that serum HIF-1alpha protein is chronically elevated in OSA (Gabryelska et al., 2020). Xie et al. (2022) measured clock gene expression in 21 people with OSA and 22 controls and found that as apnea severity increased (measured by AHI), CRY2 expression decreased (R = -0.348, P = 0.012) and Timeless expression decreased (R = -0.223, P = 0.038). In people with nocturnal hypoxemia, the normal morning-to-evening variation in BMAL1 was disrupted (Xie et al., 2022).
?-
Does Hypoxia Affect All Organs the Same Way?
No. Intermittent hypoxia phase-advances peripheral clocks in the kidney and lung but leaves the liver clock relatively unaffected — creating intertissue circadian misalignment. In the brain, 3 of 7 rhythmic canonical clock genes show altered expression under intermittent hypoxia, with the brain clock more sensitive than the liver clock (Koritala et al., 2021). The result is not that all clocks stop — it is that different organs run on different time, and the coordination between them breaks down.
Manella et al. (2020) demonstrated this tissue-specific effect in an animal model. When mice were exposed to acute hypoxia, the circadian clocks in the kidney and lung phase-advanced — meaning those organs’ internal timing moved earlier. The liver clock, in contrast, was relatively unaffected. When the researchers replicated intermittent hypoxia patterns that mimic obstructive sleep apnea, the same tissue-specific desynchronization appeared. The kidney and lung clocks moved. The liver clock stayed put. The organs were no longer coordinated.
This is not a case of all circadian clocks being equally suppressed. It is intertissue circadian misalignment — different organs running on different schedules despite being in the same body. The result is a form of internal jet lag that persists as long as the hypoxia exposure continues.
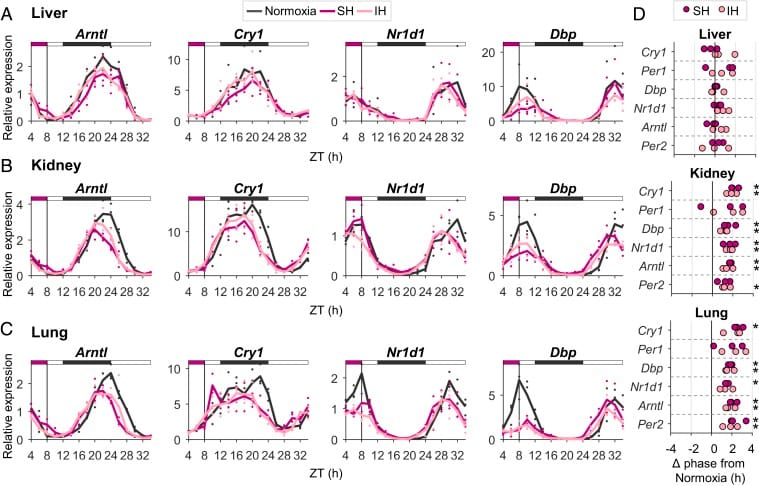
Koritala et al. (2021) examined this further in an animal model focused on the brain versus the liver. Under intermittent hypoxia, 3 of 7 rhythmic canonical clock genes showed altered expression patterns in the brain. The brain clock was more sensitive to the hypoxia than the liver clock. In the liver, Per1 and Nr1d1 (also known as Rev-Erb alpha) showed reduced amplitude, but the disruption was less widespread than in the brain.
The tissue specificity of this disruption has a direct consequence for CPAP recovery: different organs will re-entrain at different rates once the hypoxia is removed. Even after some tissues have restored their clock oscillation, others may still be running on the old, disrupted schedule. Internal desynchronization can persist well into CPAP use.
?-
How Long Does It Take for Clock Genes to Recover on CPAP?
One night of CPAP produces no measurable change in BMAL1 expression. After four months of CPAP, plasma melatonin, cortisol, and body temperature rhythms begin to recover — but clock gene oscillation patterns in blood cells remain disrupted. After two years of consistent CPAP use, hormonal and temperature rhythms are more fully restored, but gene-level circadian disruption has not fully resolved (Gaspar et al., 2021). BMAL1 showed no response to a single night of CPAP, suggesting it may be among the slower clock genes to respond (Gabryelska et al., 2025).
Gabryelska et al. (2025) measured clock gene and clock protein levels in people with OSA before and after a single night of CPAP. One night of CPAP produced no change in BMAL1 expression. CLOCK protein increased after the single night, but BMAL1 — the central activator of the circadian feedback loop — did not respond. This finding suggests BMAL1 may be slower to respond during early CPAP use.
Gaspar et al. (2021) followed a longer timeline. In a case-control study, they compared clock gene expression in peripheral blood mononuclear cells (PBMCs) from people with OSA who had used CPAP for four months versus two years, alongside healthy controls. Neither duration of CPAP fully reverted clock gene expression to control levels. The four-month group showed partial improvements. The two-year group showed more recovery in hormonal and physiological rhythms — plasma melatonin, cortisol, and body temperature oscillations were largely restored. But at the gene level, clock oscillation patterns in blood cells remained partially disrupted even at two years.
This creates a layered recovery picture. The body’s hormonal and temperature rhythms respond to CPAP within months to years. The underlying gene expression program — the molecular clock itself — recovers more slowly and may not fully normalize within the studied timeframes.
Gabryelska et al. (2025) added another dimension. In a study of CLOCK, CRY1, PER1, and HIF-1alpha protein levels in people with OSA, they found that gene expression levels and protein levels often diverged. Posttranscriptional regulation — the processes that control how much protein is made from a given amount of mRNA — means that measuring mRNA alone may underestimate the extent of clock disruption. A gene can show recovering mRNA levels while the protein product remains elevated or abnormal.
?-
Why Are 30 Percent of CPAP Users Still Exhausted?
In a study of 122 CPAP-treated individuals, 30 percent had objectively impaired alertness despite controlled AHI. The strongest predictor of residual impairment was not AHI but hypoxic burden — the cumulative oxygen desaturation load per hour (Tankere et al., 2025). People with higher pre-CPAP hypoxic burden retained more daytime impairment even after the apnea events were eliminated. Clock gene expression in blood correlates with chronotype amplitude (Gabryelska et al., 2024), suggesting a possible molecular link between disrupted clock oscillation and daily experience.
Tankere et al. (2025) studied 122 people who had been CPAP-treated for obstructive sleep apnea, with a median CPAP usage of 5.6 hours per night. Despite adequate CPAP adherence, 30 percent of the group had objectively impaired alertness. The researchers tested which pre-CPAP measurements best predicted this residual impairment. AHI — the standard measure of apnea severity — was not the strongest predictor. Hypoxic burden was. Hypoxic burden measures the cumulative oxygen desaturation load per hour, and people who scored higher on this metric (16.8 versus 8.2 in the impaired versus unimpaired groups, p=0.02) were more likely to have persistent alertness problems. An arousal index above 25 per hour predicted subjective sleepiness.
The distinction between AHI and hypoxic burden is important. AHI counts the number of apnea and hypopnea events per hour. It does not measure how deep the oxygen drops are during each event. A person with fewer apneas but deeper oxygen desaturations accumulates a higher hypoxic burden than someone with frequent but shallow events. Since HIF-1alpha activation depends on the depth and duration of oxygen drops, hypoxic burden is a more direct measure of the circadian clock gene damage that accumulates during untreated sleep apnea.
Gabryelska et al. (2024) connected clock gene expression to chronotype characteristics. In a study of 184 participants (130 with OSA and 54 controls), clock gene expression correlated with chronotype amplitude. Morning expression of BMAL1, CLOCK, CRY1, PER1, NPAS2, and NR1D1 all correlated with chronotype amplitude in the OSA group. This suggests that clock gene disruption may correspond to differences in daily energy fluctuation patterns.
Wang et al. (2025) established the dose-response relationship. In a study of 30 people with OSA of varying severity and 10 controls, PER1, PER3, CRY1, BMAL1, CLOCK, and HIF-1alpha showed disrupted diurnal expression patterns compared to controls, and the magnitude of disruption increased with apnea severity. Greater severity means more clock gene damage, which implies a longer and less complete recovery trajectory on CPAP.
?-
Many people with sleep apnea have more than one cause contributing to their sleep disruption. The circadian clock gene damage from intermittent hypoxia may compound with metabolic, hormonal, inflammatory, or autonomic factors. Identifying which causes might be involved is a useful next step.
Find out which causes might be driving your 3 AM wakeups →
?-
Frequently Asked Questions
Does the Severity of Sleep Apnea Predict How Long Recovery Takes?
Apnea severity — measured by both AHI and hypoxic burden — correlates with the magnitude of clock gene disruption. In a study of 30 people with OSA and 10 controls, PER1, PER3, CRY1, BMAL1, and CLOCK expression all showed greater disruption with increasing severity (Wang et al., 2025). Greater disruption implies a longer recovery trajectory, though no study has directly measured recovery timelines stratified by pre-CPAP severity.
Wang et al. (2025) showed a dose-response relationship: the worse the apnea, the more disrupted the clock genes. This is a logical inference — more damage takes longer to repair — but it remains an inference. No study has yet followed two groups with different baseline severity through matched CPAP durations and compared their clock gene recovery rates. The dose-response data tells us that severity predicts the depth of the hole; it does not yet tell us the exact timeline for climbing out.
Can Circadian-Targeted Approaches Speed Up Recovery on CPAP?
No controlled trial has tested circadian-targeted approaches — timed light, meal timing, low-dose melatonin — as adjuncts to CPAP for accelerating clock gene recovery. The mechanisms support the hypothesis: timed morning light re-entrains the SCN, consistent meal timing re-entrains peripheral clocks (especially liver and pancreas), and low-dose melatonin reinforces phase timing. These are approaches with independent circadian evidence that could theoretically complement CPAP (Sartor et al., 2024).
Sartor et al. (2024) reviewed the bidirectional relationship between the circadian clock and hypoxia, noting that HIF and clock components interact at both the structural protein level (bHLH-PAS domains) and the transcriptional level. This bidirectionality means that approaches targeting the clock side of the equation — light timing, meal timing, melatonin — could in theory help re-entrain the disrupted oscillation that HIF-1alpha has altered. None of these have been tested in a controlled trial alongside CPAP. The mechanistic rationale is sound, but direct evidence is not yet available.
Does Untreated Sleep Apnea Cause Permanent Clock Damage?
The evidence suggests clock gene disruption from sleep apnea is not permanent but recovers slowly. After two years of consistent CPAP, hormonal and temperature rhythms are largely restored, but gene-level oscillation patterns remain partially disrupted (Gaspar et al., 2021). No study has followed clock gene recovery beyond two years to determine whether full restoration eventually occurs.
Gaspar et al. (2021) showed that two years of CPAP restored melatonin, cortisol, and body temperature rhythms, but the clock gene expression program in PBMCs had not fully normalized. This is partial recovery, not permanent damage — but the timeline is long enough that it has not yet been measured to completion. Manella et al. (2020) showed in an animal model that the desynchronization between organs is tissue-specific, which suggests that different tissues will recover on different schedules. Some organs may normalize within months; others may take years. No human study has tracked multi-tissue clock gene recovery over the timeframes that would be needed to answer whether full restoration eventually occurs.
?-
Related Reading
- Circadian Rhythm Disruption: How a Misaligned Internal Clock Fragments Sleep, Drives 3AM Wakeups, and Reduces Sleep Depth — the full overview of circadian sleep disruption, including melatonin, cortisol, body temperature, peripheral clocks, and early-morning waking
- Does Your Circadian Clock Control When Insulin Works? Prediabetes, the Dawn Phenomenon, and Sleep — how circadian insulin timing, glucose rhythm, and dawn physiology affect sleep continuity
- Can Fixing Your Gut Fix Your Sleep? How Gut Bacteria Entrain Your Body Clock — how gut bacteria, the vagus nerve, and microbial rhythms interact with sleep timing
- Can Chronic Inflammation Disrupt Your Circadian Clock Genes? — how cytokines and NF-kB signaling suppress central and peripheral clock genes
- Can Your Body Temperature Keep You From Falling Asleep? — how thermoregulation, vasodilation, and temperature rhythm affect sleep onset and depth
- Do Beta Blockers Suppress Melatonin? What Blood Pressure Medication Does to Your Body Clock — how beta blockers, melatonin suppression, and medication timing affect circadian sleep
References
Adamovich, Y., Ladeuix, B., Golik, M., Koeners, M. P., & Asher, G. (2017). Rhythmic oxygen levels reset circadian clocks through HIF1alpha. Cell Metabolism, 25(1), 93-101. https://pubmed.ncbi.nlm.nih.gov/27773695/
Gabryelska, A., Turkiewicz, S., Karuga, F. F., Sochal, M., Strzelecki, D., & Bialasiewicz, P. (2022). Disruption of circadian rhythm genes in obstructive sleep apnea patients — possible mechanisms involved and clinical implication. International Journal of Molecular Sciences, 23(2), 709. https://pubmed.ncbi.nlm.nih.gov/35054894/
Gabryelska, A., Turkiewicz, S., Gajewski, A., Bialasiewicz, P., Strzelecki, D., Chalubinski, M., & Sochal, M. (2024). Investigating the link between circadian clock gene expressions, chronotype, insomnia, and daytime sleepiness in patients with obstructive sleep apnea. International Journal of Molecular Sciences, 25(16), 9062. https://pubmed.ncbi.nlm.nih.gov/39201748/
Gabryelska, A., Turkiewicz, S., Gajewski, A., Bialasiewicz, P., Strzelecki, D., Ditmer, M., Chalubinski, M., & Sochal, M. (2025). Elucidating the interplay of hypoxia-inducible factor and circadian clock signaling in obstructive sleep apnea patients. International Journal of Molecular Sciences, 26(3), 971. https://pubmed.ncbi.nlm.nih.gov/39940739/
Gabryelska, A., Turkiewicz, S., Gajewski, A., Jaromirska, J., Strzelecki, D., Bialasiewicz, P., Chalubinski, M., & Sochal, M. (2025). Assessment of continuous positive airway pressure effect on the circadian clock signaling pathway in obstructive sleep apnea patients. Scientific Reports, 15(1), 11273. https://pubmed.ncbi.nlm.nih.gov/40175421/
Gaspar, L. S., Hesse, J., Yalcin, M., Santos, B., Carvalhas-Almeida, C., Ferreira, M., Moita, J., Relogio, A., Cavadas, C., & Alvaro, A. R. (2021). Long-term continuous positive airway pressure treatment ameliorates biological clock disruptions in obstructive sleep apnea. EBioMedicine, 65, 103248. https://pubmed.ncbi.nlm.nih.gov/33647771/
Koritala, B. S. C., Lee, Y. Y., Bhadri, S. S., Gaspar, L. S., Stanforth, C., Wu, G., Ruben, M. D., Francey, L. J., & Smith, D. F. (2021). Intermittent hypoxia alters the circadian expression of clock genes in mouse brain and liver. Genes, 12(10), 1627. https://pubmed.ncbi.nlm.nih.gov/34681021/
Manella, G., Aviram, R., Bolshette, N., Muvkadi, S., Golik, M., Smith, D. F., & Asher, G. (2020). Hypoxia induces a time- and tissue-specific response that elicits intertissue circadian clock misalignment. Proceedings of the National Academy of Sciences, 117(1), 779-786. https://pubmed.ncbi.nlm.nih.gov/31848250/
O’Connell, E. J., Martinez, C. A., Liang, Y. G., Cistulli, P. A., & Cook, K. M. (2020). Out of breath, out of time: interactions between HIF and the circadian rhythms. American Journal of Physiology-Cell Physiology, 319(3), C533-C540. https://pubmed.ncbi.nlm.nih.gov/32726159/
Sartor, F., Ferrero-Bordera, B., Haspel, J., Sperandio, M., Holloway, P. M., & Merrow, M. (2024). Circadian clock and hypoxia. Circulation Research, 134(6), 618-634. https://pubmed.ncbi.nlm.nih.gov/38484033/
Tankere, P., Taillard, J., PetitJean, T., Le-Cam, P., Ricordeau, F., Blanchard, M., Vanbuis, J., Nofal, A., Tamisier, R., Peter-Derex, L., & Stauffer, E. (2025). Residual sleepiness and impaired alertness in treated obstructive sleep apnoea: role of hypoxic burden and sleep fragmentation. Thorax, 80(4), 245-247. https://pubmed.ncbi.nlm.nih.gov/39832943/
Wang, H. M., Shieh, K. R., & Chang, E. T. (2025). Correlation of the expression of circadian-clock genes with the severity of obstructive sleep apnea in patients. Chronobiology International, 42(3), 428-439. https://pubmed.ncbi.nlm.nih.gov/40113269/
Xie, T., Guo, D., Luo, J., Guo, Z., Zhang, S., Wang, A., Wang, X., Wang, X., Cao, W., Su, L., Guo, J., Huang, R., & Xiao, Y. (2022). The relationship between HIF1alpha and clock gene expression in patients with obstructive sleep apnea. Nature and Science of Sleep, 14, 397-404. https://pubmed.ncbi.nlm.nih.gov/35299629/
Written by Kat Fu, M.S., M.S. ? Last reviewed: May 2026 ? 13 references cited
