Sleep after 40 changes. The question is whether those changes are fixed or whether some of them can be addressed once you know what is going on mechanistically. The circadian research separates age-related sleep decline into distinct mechanisms, each with different trajectories and different degrees of reversibility.
This article covers the six major circadian mechanisms that change with aging, which ones are structural and permanent, and what the evidence says about the ones that respond to behavioral changes. Circadian aging is one component of the broader circadian disruption picture. For the full overview, see Circadian Rhythm Disruption.
What Happens to the Master Clock in the Brain After 40?
Wang et al. (2015) provided the first human evidence linking SCN neuron count to circadian rhythm strength. In a postmortem study that paired brain tissue analysis with pre-death actigraphy recordings, the number of VIP-immunoreactive neurons in the SCN correlated with the amplitude of the 24-hour rest-activity rhythm — a partial correlation of R = 0.83 after adjusting for age and sex. In the same study, participants with Alzheimer’s disease did not show significant differences in VIP neuron count compared to non-AD participants, but did show a phase delay of approximately 2.9 hours in their activity rhythms (Wang et al., 2015).
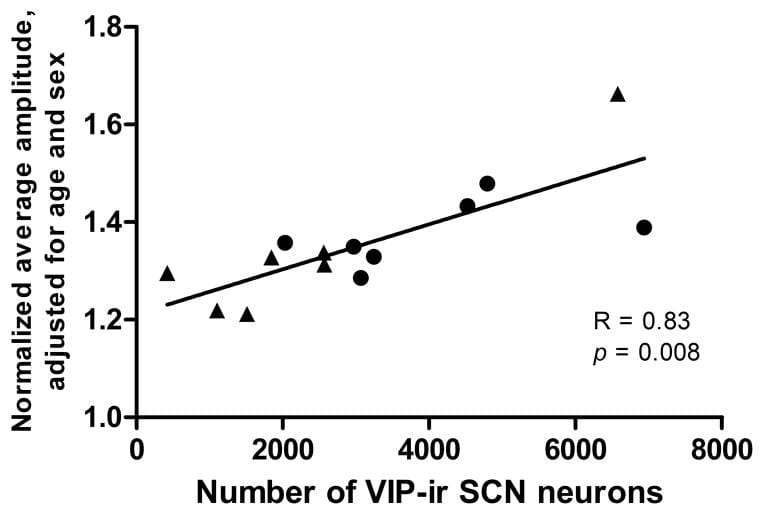
This means fewer VIP neurons produce a weaker daily rhythm — measured in actual movement patterns, not just molecular markers.
Farajnia et al. (2012) identified the mechanism at the cellular level. In aged mice, individual SCN neurons showed single-cell arrhythmia — some neurons lost rhythmic firing patterns altogether. Beyond that, neurons that retained rhythmicity fell out of sync with each other. Some subpopulations peaked at opposite phases, canceling each other’s output. The problem was not that clock genes stopped working inside each neuron. The problem was that the network lost coordination (Farajnia et al., 2012).
Nakamura et al. (2016) connected these findings in a review. SCN decline is driven by loss of neuropeptide rhythmicity — VIP, arginine vasopressin (AVP), and gastrin-releasing peptide (GRP) all lose their daily oscillation patterns — combined with impaired inter-neuronal coupling. When researchers implanted fetal SCN tissue into aged animals, behavioral rhythms were restored, confirming that the SCN is the primary locus of age-related circadian decline. The core clock genes within individual neurons remain functional. What degrades is the communication between neurons that produces a coherent output (Nakamura et al., 2016).
This is a structural change. No behavioral approach rebuilds lost VIP neurons or restores inter-neuronal coupling once it degrades. But the downstream effects of SCN weakening — peripheral clock desynchronization, sleep onset timing, temperature rhythm flattening — can be addressed through other pathways. The foundation weakens with age, but the structures built on it are not all equally rigid.
Why Does Your Body Temperature Rhythm Flatten With Age?
Weinert and Waterhouse (2007) compared core body temperature (CBT) amplitude across age groups and found a measurable reduction under normal daily conditions: 0.33 +/- 0.01 degrees Celsius in younger adults (ages 17-39) versus 0.26 +/- 0.01 degrees Celsius in elderly participants (p < 0.001). Beyond amplitude, the phase of the rhythm also advanced — the temperature nadir occurred earlier in the night (Weinert & Waterhouse, 2007).
The temperature rhythm matters for sleep because the evening CBT drop is the thermoregulatory gate for sleep onset. As core temperature decreases, vasodilation in the extremities dissipates heat, and the brain interprets this decline as a timing cue to initiate sleep. When the amplitude of the temperature rhythm is flattened, the drop is shallower and the onset cue is weaker. A ~20 percent reduction in amplitude — the difference measured between younger and elderly groups — translates into a less distinct “now is the time to sleep” cue from the thermoregulatory pathway.
Physical activity contributes to this picture. Lower daily activity levels in older adults reduce the daytime temperature peak, compressing the overall amplitude. Weinert and Waterhouse found that under constant routine conditions — where activity and other masking effects are controlled — age-dependent differences in amplitude were no longer significant, suggesting the measured difference is largely driven by lifestyle factors rather than an irreversible endogenous change. This makes the temperature rhythm amplitude one of the more addressable components of circadian aging, potentially responsive to maintained physical activity levels. The body temperature rhythm is one of the pathways the SCN uses to entrain peripheral clocks throughout the body, but the age-related amplitude difference appears to be substantially modifiable through behavior.
Does Your Eye’s Lens Block the Light Your Clock Needs?
Kessel et al. (2010) quantified how much blue light the aging lens blocks. Using spectral transmission measurements at 480 nanometers — the peak sensitivity wavelength for melanopsin, the photopigment in retinal ganglion cells responsible for circadian entrainment — they documented a progressive decrease in transmission starting in early adulthood and continuing across the lifespan. The overall decline from age 10 to 80 was 72 percent (Kessel et al., 2010).
Kessel et al. (2011) then tested whether this optical change has measurable consequences for sleep. In a Danish population study, sleep disturbance prevalence tracked with the degree of lens yellowing (p < 0.0001) — independent of age, diabetes status, smoking, and cardiovascular disease risk. The relationship held after controlling for these confounders, which means the lens yellowing itself, not just the general aging that accompanies it, is associated with sleep disruption (Kessel et al., 2011).
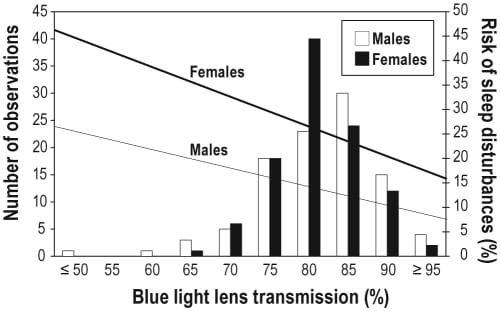
Eto and Higuchi (2023) reviewed the downstream consequences. In older adults, reduced pupil diameter and increased lens opacity attenuate the light reaching intrinsically photosensitive retinal ganglion cells (ipRGCs), weakening circadian photoreception. Melatonin suppression — the standard measure of circadian light sensitivity — is reduced in older adults compared to younger adults at the same light exposure levels. The practical meaning: older adults need higher intensity or more spectrally enriched morning light to achieve the same circadian entrainment that younger adults get from moderate daylight (Eto & Higuchi, 2023).
What makes this finding actionable is the nature of the deficit. The problem is in the lens — an optical filter — not in the retina or the neural pathway behind it. Melanopsin-containing retinal ganglion cells remain functional. The light does not reach them at sufficient intensity. Brighter morning light, blue-enriched light therapy devices, or cataract surgery (which replaces the yellowed lens with a transparent intraocular lens) all restore melanopic input. Among the age-related circadian changes, this one has the strongest behavioral workaround.
Which Age-Related Circadian Changes Can You Reverse?
Tahara et al. (2017) produced a key finding in this area. Peripheral clocks — in the kidney, liver, and submandibular gland — showed normal amplitude in aged animals under standard conditions. They did not break down on their own. But when the researchers tested re-entrainment (the ability to reset timing after a disruption), aged animals showed significantly impaired re-entrainment of peripheral clocks to exercise and restraint stress, while light-driven re-entrainment remained largely intact. The mechanism was reduced adrenergic receptor expression — the sympathetic nervous pathway that normally transmits timing information from the SCN to peripheral tissues had degraded. Meal timing, however, retained full entrainment power. Peripheral clocks in aged animals responded to food-based timing cues just as effectively as in young animals (Tahara et al., 2017).
This is the mechanistic basis for time-restricted eating as an aging circadian approach. When the sympathetic nervous pathway weakens with age, food timing becomes the remaining high-capacity channel for peripheral clock re-entrainment.
The circadian phase advance that defines aging in humans — where melatonin onset and core body temperature nadir shift earlier — is well-documented in the literature (Kim et al., 2022). At the molecular level, research on postmortem human prefrontal cortex found that 588 genes lose rhythmicity with age (Chen et al., 2016). The evening wake-promoting cue weakens, contributing to the pattern of earlier sleep onset and earlier morning waking that many adults over 50 experience. Kim et al. reviewed timed light therapy and low-dose melatonin as approaches that can partially counteract this phase advance — not by reversing the underlying SCN decline, but by providing external timing cues that delay the rhythm (Kim et al., 2022).
Fischer et al. (2017) added a sex-specific dimension. Chronotype — the preferred timing of sleep and activity — advances progressively from around age 20 onward. Before age 40, women tend to be earlier chronotypes than men. After 40, men become earlier chronotypes than women — a sex crossover at midlife that coincides with hormonal transitions. In men, the advance continues progressively. In women, the trajectory shows a plateau between approximately ages 35 and 55, potentially reflecting menopausal transitions, before the advance resumes (Fischer et al., 2017).
The evidence separates age-related circadian changes into two categories:
Reversible or partially addressable:
- Peripheral clock entrainment — meal timing retains full power even when exercise pathways weaken
- Melanopic light sensitivity — brighter or blue-enriched morning light compensates for lens yellowing
- Phase advance — timed light therapy and low-dose melatonin can delay timing
- Body temperature rhythm amplitude — maintaining physical activity levels may preserve the amplitude difference
Structural and irreversible:
- SCN neuron loss — VIP neurons do not regenerate
- Pineal calcification — calcium-phosphate deposits permanently replace melatonin-producing tissue
- Inter-neuronal desynchrony — lost coupling between SCN neurons is not restored by external cues
The structural changes set the floor. The reversible changes determine how much room exists above that floor — and how much of the sleep disruption experienced after 40 is addressable rather than inevitable.
Many people have more than one cause contributing to their sleep disruption. Age-related circadian changes may compound with metabolic, hormonal, inflammatory, or autonomic factors — especially after 40 when multiple causes tend to overlap. Identifying which causes might be involved is a useful next step.
Find out which causes might be driving your 3 AM wakeups →
Frequently Asked Questions
Why Do Men Over 40 Start Waking Up Earlier?
The phase advance in aging — where melatonin onset and CBT nadir shift earlier in older adults — is a well-characterized finding in circadian biology (Kim et al., 2022). Separately, postmortem research on human prefrontal cortex identified 588 genes that lose circadian rhythmicity with age (Chen et al., 2016). Fischer et al. (2017) showed that this advance is sex-specific. Before age 40, women are earlier chronotypes than men. After 40, men cross over and become the earlier chronotype, with the advance continuing progressively. The timing of this crossover coincides with hormonal changes in both sexes — testosterone decline in men and perimenopause in women — though the specific hormonal contribution to chronotype advancement remains under investigation (Kim et al., 2022; Fischer et al., 2017).
Does Exercise Help Reset Circadian Timing After 40?
Tahara et al. (2017) tested exercise against other zeitgebers (time-givers) in young and aged animals. Scheduled treadmill exercise produced robust peripheral clock entrainment in young mice but weakened entrainment in aged mice. The mechanism was the same one that impaired stress-driven re-entrainment: reduced adrenergic receptor expression in peripheral tissues. The sympathetic nervous pathway — which exercise uses to transmit timing cues to the kidneys, liver, and other organs — degrades with age. Meal timing bypassed this pathway and retained full entrainment capacity in the aged group (Tahara et al., 2017).
This does not mean exercise has no circadian value after 40. Exercise still influences core body temperature, cortisol timing, and direct SCN input through activity-dependent pathways. What weakens is the specific peripheral clock re-entrainment channel that runs through adrenergic receptors. The caveat: this was demonstrated in an animal model, and human-equivalent data on peripheral clock re-entrainment by exercise across age groups is still limited.
Is Circadian Decline the Same as Needing Less Sleep?
Van Cauter et al. (2000) documented the endocrine consequences of age-related sleep changes. Growth hormone secretion — which depends on slow-wave sleep — declines with age as deep sleep decreases. Cortisol levels during the sleep period increase. These are consequences of degraded sleep architecture, not evidence that the body needs less sleep. The hormonal profile of an older adult’s sleep is worse than a younger adult’s, which is the opposite of what you would expect if the reduced sleep were meeting reduced demand (Van Cauter et al., 2000).
Alhajaji et al. (2026) confirmed that both SWS and REM decline with age across population-level data. The reductions are consistent and progressive, not a step-function at any particular age. The conclusion aligns with Van Cauter’s earlier work: the ability to produce deep sleep and REM declines, but the physiological need for what those stages provide — cellular repair, memory consolidation, hormonal regulation — does not (Alhajaji et al., 2026).
The distinction matters because misinterpreting circadian decline as reduced sleep need leads to acceptance of poor sleep as normal. A 60-year-old who sleeps 5.5 hours is not someone whose body has adapted to less. They are someone whose circadian and sleep-producing mechanisms have weakened — and some of those mechanisms are partially addressable.
Related Reading
- Circadian Rhythm Disruption: How a Misaligned Internal Clock Fragments Sleep, Drives 3AM Wakeups, and Reduces Sleep Depth — the full overview of circadian sleep disruption, including melatonin, cortisol, body temperature, peripheral clocks, and early-morning waking
- How Does a Two-Hour Weekend Sleep Delay Desynchronize Your Peripheral Clocks for Days? — how repeated weekend schedule shifts create chronic circadian misalignment
- Can Rotating Night Schedules Permanently Desynchronize Your Peripheral Clocks? — how rotating night work affects peripheral clocks and metabolic timing
- Why Does Eastbound Jet Lag Last Longer Than Westbound — and What Determines Recovery Rate? — why travel direction changes the speed of circadian recovery
- Night Owl or Delayed Sleep Phase Disorder? How to Know the Difference — how delayed circadian timing differs from preference alone
- Why Does Your Wake Time Advance by Decades — and Can You Reverse Circadian Phase Advance After 60? — why aging shifts wake time earlier and which parts remain reversible
- Does Evening Exercise Delay Your Circadian Clock — or Is Disruption Smaller Than You Think? — how exercise timing affects the circadian clock and sleep onset
- Does Time-Restricted Eating Entrain Your Peripheral Clocks? — how meal timing signals peripheral clocks outside the brain
References
Alhajaji, R., Jahrami, H., Pandi-Perumal, S. R., & BaHammam, A. S. (2026). Sleep health in the older adults: Architecture, circadian changes, and common sleep disorders. Ageing Research Reviews, 118, 103101. https://pubmed.ncbi.nlm.nih.gov/41825783/
Chen, C.-Y., Logan, R. W., Ma, T., Lewis, D. A., Tseng, G. C., Sibille, E., & McClung, C. A. (2016). Effects of aging on circadian patterns of gene expression in the human prefrontal cortex. Proceedings of the National Academy of Sciences, 113(1), 206-211. https://pubmed.ncbi.nlm.nih.gov/26699485/
Eto, T., & Higuchi, S. (2023). Review on age-related differences in non-visual effects of light: Melatonin suppression, circadian phase shift and pupillary light reflex in children to older adults. Journal of Physiological Anthropology, 42(1), 11. https://pubmed.ncbi.nlm.nih.gov/37355647/
Farajnia, S., Michel, S., Deboer, T., vanderLeest, H. T., Houben, T., Rohling, J. H. T., Ramkisoensing, A., Yasenkov, R., & Meijer, J. H. (2012). Evidence for neuronal desynchrony in the aged suprachiasmatic nucleus clock. The Journal of Neuroscience, 32(17), 5891-5899. https://pubmed.ncbi.nlm.nih.gov/22539850/
Fischer, D., Lombardi, D. A., Marucci-Wellman, H., & Roenneberg, T. (2017). Chronotypes in the US — Influence of age and sex. PLOS ONE, 12(6), e0178782. https://pubmed.ncbi.nlm.nih.gov/28636610/
Kessel, L., Lundeman, J. H., Herbst, K., Andersen, T. V., & Larsen, M. (2010). Age-related changes in the transmission properties of the human lens and their relevance to circadian entrainment. Journal of Cataract & Refractive Surgery, 36(2), 308-312. https://pubmed.ncbi.nlm.nih.gov/20152615/
Kessel, L., Siganos, G., Jorgensen, T., & Larsen, M. (2011). Sleep disturbances are related to decreased transmission of blue light to the retina caused by lens yellowing. Sleep, 34(9), 1215-1219. https://pubmed.ncbi.nlm.nih.gov/21886359/
Kim, J. H., Elkhadem, A. R., & Duffy, J. F. (2022). Circadian rhythm sleep-wake disorders in older adults. Sleep Medicine Clinics, 17(2), 241-252. https://pubmed.ncbi.nlm.nih.gov/35659077/
Nakamura, T. J., Takasu, N. N., & Nakamura, W. (2016). The suprachiasmatic nucleus: Age-related decline in biological rhythms. The Journal of Physiological Sciences, 66(5), 367-374. https://pubmed.ncbi.nlm.nih.gov/26915078/
Tahara, Y., Takatsu, Y., Shiraishi, T., Kikuchi, Y., Yamazaki, M., Motohashi, H., Muto, A., Sasaki, H., Haraguchi, A., Kuriki, D., Nakamura, T. J., & Shibata, S. (2017). Age-related circadian disorganization caused by sympathetic dysfunction in peripheral clock regulation. npj Aging and Mechanisms of Disease, 3, 16030. https://pubmed.ncbi.nlm.nih.gov/28721279/
Van Cauter, E., Leproult, R., & Plat, L. (2000). Age-related changes in slow wave sleep and REM sleep and relationship with growth hormone and cortisol levels in healthy men. JAMA, 284(7), 861-868. https://pubmed.ncbi.nlm.nih.gov/10938176/
Wang, J. L., Lim, A. S., Chiang, W.-Y., Hsieh, W.-H., Lo, M.-T., Schneider, J. A., Buchman, A. S., Bennett, D. A., Hu, K., & Saper, C. B. (2015). Suprachiasmatic neuron numbers and rest-activity circadian rhythms in older humans. Annals of Neurology, 78(2), 317-322. https://pubmed.ncbi.nlm.nih.gov/25921596/
Weinert, D., & Waterhouse, J. (2007). The circadian rhythm of core temperature: Effects of physical activity and aging. Physiology & Behavior, 90(2-3), 246-256. https://pubmed.ncbi.nlm.nih.gov/17069866/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 13 references cited
