Waking at 4 or 5 AM is not a sleep disorder in itself — it is the visible output of a circadian phase advance that begins earlier than expected and compounds through three distinct mechanisms. For adults over 60, this advance is the primary reason sleep timing moves out of alignment with social and family schedules. This article covers the cellular cause (SCN neuron loss), the optical cause (lens yellowing and pupil constriction), the evidence for light-based reversal, and the feedback loop between aging, melatonin decline, and clock gene expression. Sleep apnea and other structural sleep disorders are outside the scope here. For the broader picture of how circadian disruption drives middle-of-the-night wakeups at any age, see Circadian Rhythm Disruption: How a Misaligned Internal Clock Fragments Sleep, Drives 3AM Wakeups, and Reduces Sleep Depth.
How Many Suprachiasmatic Neurons Are Lost With Aging — and Does That Drive Phase Advance?
The suprachiasmatic nucleus contains approximately 20,000 neurons that coordinate the body’s master clock. Not all of these neurons contribute equally to circadian timing. The VIP (vasoactive intestinal peptide)-producing interneurons are the cells responsible for synchronizing neighboring SCN neurons into a coherent oscillation. When VIP neuron numbers decline, inter-neuronal coupling weakens, the amplitude of the master clock dampens, and the circadian phase set point advances earlier.
Wang et al. (2015) examined post-mortem SCN tissue from 17 older community-dwelling adults (mean age 90.4 years; 10 cognitively healthy, 7 with Alzheimer’s disease). VIP neuron count correlated R = 0.83 (p = 0.008) with the amplitude of rest-activity circadian rhythms measured by wrist actigraphy during life. AVP (arginine vasopressin) neuron counts showed no correlation with any circadian measure (p = 0.78). The VIP population — not AVP — mediates amplitude in humans.
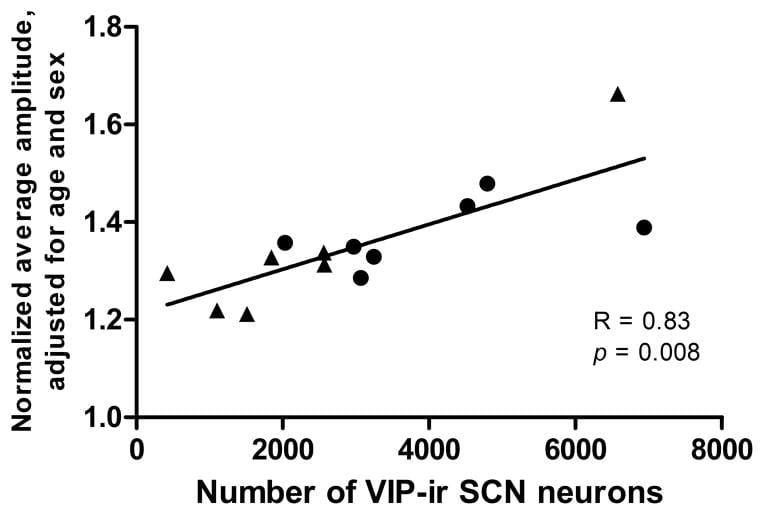
This is a network-level deterioration, not a molecular clock malfunction within individual neurons. Each SCN neuron retains its own transcription-translation feedback loop (the CLOCK/BMAL1/PER/CRY cycle), but without enough VIP interneurons to couple those individual oscillators together, the collective output weakens. The dampened amplitude allows the phase set point to advance — producing the progressively earlier wake times observed across decades of aging.
One additional finding from the Wang study: Alzheimer’s disease did not reduce circadian amplitude (p = 0.88). Instead, Alzheimer’s produced a phase delay of approximately 2.9 hours (p = 0.0018) — the opposite direction from healthy aging. Interdaily stability (the consistency of the rest-activity pattern from day to day) was also reduced in Alzheimer’s subjects (p = 0.015), reflecting greater day-to-day fragmentation. This distinction matters: the circadian direction in healthy aging is earlier, while in Alzheimer’s it is later and more fragmented.
How Does Lens Yellowing Reduce the Light That Sets Your Clock?
The SCN does not receive visual input the way the visual cortex does. Circadian entrainment depends on a dedicated set of retinal cells — ipRGCs — that contain the photopigment melanopsin, with peak sensitivity near 480 nm (blue wavelength). Two age-related changes in the eye progressively reduce the photon flux reaching these cells.
The first is lens yellowing. Kessel et al. (2011) studied 970 participants aged 30–60 and quantified crystalline lens opacity using autofluorometry. Sleep disturbance prevalence rose from 15.7% in adults aged 30–35 to 32.5% in those aged 55–60 (p = 0.0002). After adjusting for age, sex, diabetes, smoking, and ischemic heart disease, reduced blue-light transmission through the lens still predicted sleep disturbance (OR 0.97 per 1% change in transmission, p = 0.016). A 50-year-old woman with high lens transmission carried a 16.4% sleep disturbance risk compared to 37.9% for a woman with low transmission — more than a twofold difference attributable to the optical variable alone.
The mechanism is direct: the yellowed lens acts as a filter that preferentially absorbs ~460–480 nm wavelengths before they reach the retina. With less short-wavelength light stimulating ipRGCs, the melanopsin-mediated entrainment input to the SCN weakens, and the nocturnal melatonin rise is blunted. The clock receives a weaker “time of day” input and moves toward its advanced default.
The second optical mechanism is pupil constriction. Eto and Higuchi (2023) identified in a review that senile miosis — the progressive narrowing of the pupil with age — reduces total retinal photon flux before light even reaches the lens. Both lens yellowing and pupil miosis independently reduce melanopsin activation. Intraocular lens implantation (cataract surgery) can restore short-wavelength transmission by replacing the yellowed lens with a transparent artificial one, and the Kessel data support this as a corrective step for circadian entrainment. However, the Eto finding establishes that pupil miosis persists as a second barrier even after surgery — cataract removal addresses one of the two optical mechanisms, not both.
Is the Phase Advance Reversible — and What Does the Evidence Say About Evening Light?
A common assumption is that the aging clock becomes rigid — unable to respond to light the way a younger clock does. Kripke et al. (2007) tested this directly. In 106 healthy participants (50 young, mean age 23; 56 older, mean age 67), exposure to 3,000 lux bright light for 3 hours on 3 consecutive days produced maximal phase changes of approximately 3 hours in both age groups. There was no statistically detectable difference in phase-change magnitude between older and younger adults or between sexes.
What did differ was the position of the phase response curve (PRC). The inflection point — the crossover from phase delays to phase advances — occurred 1.8 hours earlier in older adults when referenced to melatonin metabolite rhythms. A “dead zone” in the mid-to-late afternoon produced negligible phase changes in both groups. The practical consequence: evening light intended to delay circadian phase in older adults must be timed approximately 1.8 hours earlier than in younger adults to fall within the delay portion of the PRC. Using the same evening timing that works in a 25-year-old may land in the advance zone for a 67-year-old — producing the opposite of the intended effect.
Constantino et al. (2025) tested this timing-dependent effect in practice with an 11-week randomized crossover trial in 36 older adults (median age 66.5; 25 female). The study compared blue-enriched white light (17,000 K, 1,100–1,200 lux) against control white light (4,000 K, 300–450 lux). Morning blue-enriched light improved interdaily stability (p = 0.004) and reduced sleep fragmentation as measured by intradaily variability (p = 0.01). Evening blue-enriched light produced the opposite: increased sleep latency (p < 0.001) and decreased sleep efficiency (p = 0.003). Overall Pittsburgh Sleep Quality Index scores improved from baseline (t = 3.20, p = 0.003), driven by the morning arm.
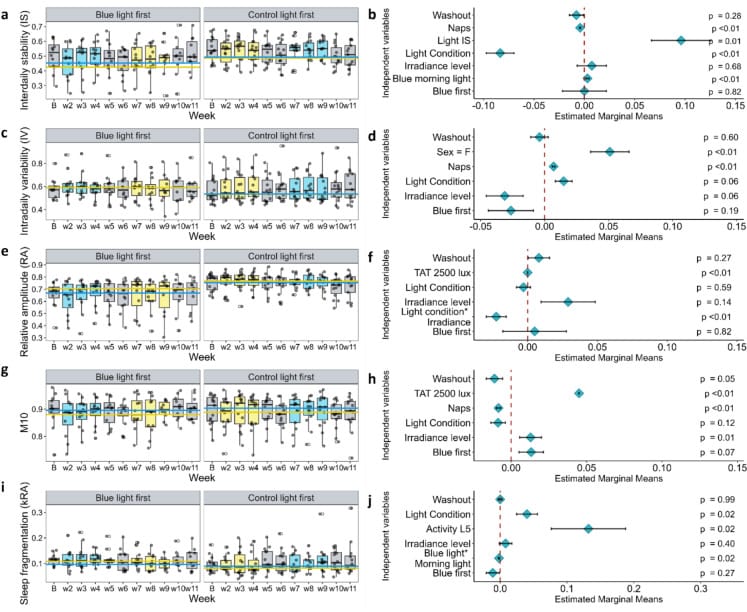
The dichotomy is not about dose — it is about timing, shaped by the 1.8-hour-earlier PRC position that Kripke identified. Morning blue light advances and stabilizes the circadian phase. Evening blue light actively worsens sleep in older adults.
Does the Aging-Circadian-Melatonin Feedback Loop Accelerate Itself?
Verma et al. (2023) reviewed the bidirectional relationship between aging, circadian rhythm deterioration, and melatonin decline. The key finding: this is not a one-directional decline but a self-reinforcing loop. Endogenous melatonin synthesis and nocturnal secretion decrease progressively from the third decade, with both the amplitude and duration of the melatonin peak reduced in older adults. The diminished melatonin output weakens the hormonal input that reinforces the SCN’s phase-setting function. At the SCN level, reduced neuronal synchronization, impaired photic responsiveness, and downregulated expression of BMAL1, PER, and CRY clock genes collectively weaken the entrainment output transmitted to peripheral oscillators throughout the body. The downstream consequences — phase advance, fragmented sleep, blunted temperature and cortisol rhythms, and impaired immune regulation — feed forward to accelerate cellular aging.
The loop can be described in a single chain: SCN VIP neuron loss reduces circadian amplitude, which weakens the melatonin production drive, which removes the hormonal reinforcement that supports SCN coupling, which further reduces amplitude. The optical degradation described in the lens yellowing section above reduces the photic input that could partially compensate for this internal decline. Both the hormonal and photic inputs converge to accelerate phase advance across decades.
Can the loop be interrupted? Baser et al. (2025) reviewed evidence including a 15-month Dutch randomized trial in approximately 200 nursing home residents (mean age 86 years) that tested combined bright light plus 2.5 mg nightly melatonin. The combined approach reduced agitation and improved sleep quality — demonstrating that multimodal circadian targeting retains effectiveness even at advanced age. The proposed molecular mechanism: melatonin acts through MT1 and MT2 receptors, activating PI3K/AKT neuroprotective pathway and SIRT1 upregulation, stabilizing mitochondrial function and suppressing neuroinflammation. These pathways may slow the amplitude decline itself rather than only compensating for its effects — suggesting that intervening at two points in the loop (photic input via light, hormonal input via melatonin) is more effective than targeting either alone.
Circadian phase advance with aging is rarely the only factor disrupting sleep after 60. Hormonal changes, metabolic alterations, and autonomic imbalance may all compound the clock problem. Identifying which causes might be involved is a useful next step.
Find out which causes might be driving your 3 AM wakeups →
Is Waking Up at 4–5 AM Normal Aging or an Addressable Problem?
The distinction is between an advanced chronotype and a functionally impairing condition. Phase advance alone — waking earlier and sleeping earlier — is a normal trajectory of the aging clock. It becomes a problem when the earlier wake time is not accompanied by an earlier bedtime, producing a net reduction in sleep duration, or when the advanced timing conflicts with social obligations, family schedules, or desired activities.
Advanced Sleep Phase Disorder (ASPD) is the formal designation when the advance is accompanied by daytime impairment. The Kripke (2007) data are relevant here: phase-change capacity is preserved in older adults, meaning the circadian clock can still respond to appropriately timed light. The barrier to correction is not rigidity of the clock — it is the 1.8-hour earlier position of the phase response curve, which requires adjusted timing of any light-based approach. Early waking without daytime impairment does not require correction. Early waking with impairment is addressable.
At What Age Does Circadian Phase Advance Typically Begin?
The three mechanisms described in this article do not all begin at the same age. Melatonin amplitude starts declining from the third decade of life (Verma et al., 2023). Crystalline lens yellowing is measurable in the 40s–50s, as reflected in the Kessel (2011) cohort where sleep disturbance prevalence doubled between ages 30 and 60. SCN VIP neuron loss accumulates across decades and becomes functionally consequential by the 60s–70s, as reflected in the Wang (2015) data from adults with a mean age of 90. The convergence of all three mechanisms explains why the phase advance tends to accelerate rather than plateau — each mechanism removes a compensatory input that could buffer the others.
Can Cataract Surgery Restore Circadian Light Sensitivity?
The Kessel (2011) data identified lens transmittance as a modifiable, age-independent variable: at the same chronological age, women with higher lens transmission had half the sleep disturbance risk of women with lower transmission. Replacing the opacified lens with a transparent intraocular lens restores the short-wavelength transmission pathway, and the epidemiological data support this as a meaningful optical correction for circadian entrainment.
However, Eto and Higuchi (2023) identified that senile pupil miosis constitutes a second, independent optical barrier. Even after cataract surgery restores lens clarity, the constricted pupil reduces the total number of photons entering the eye before light reaches the new lens. Both mechanisms — lens opacity and pupil miosis — independently reduce melanopsin activation and contribute to weakened circadian entrainment. Cataract surgery addresses one of the two optical pathways, not both.
References
1. Wang, J. L., Lim, A. S., Chiang, W. Y., Hsieh, W. H., Lo, M. T., Schneider, J. A., Buchman, A. S., Bennett, D. A., Hu, K., & Saper, C. B. (2015). Suprachiasmatic neuron numbers and rest-activity circadian rhythms in older humans. Annals of Neurology, 78(2), 317–322. https://pubmed.ncbi.nlm.nih.gov/25921596/
2. Kessel, L., Siganos, G., Jorgensen, T., & Larsen, M. (2011). Sleep disturbances are related to decreased transmission of blue light to the retina caused by lens yellowing. Sleep, 34(9), 1215–1219. https://pubmed.ncbi.nlm.nih.gov/21886359/
3. Kripke, D. F., Elliott, J. A., Youngstedt, S. D., & Rex, K. M. (2007). Circadian phase response curves to light in older and young women and men. Journal of Circadian Rhythms, 5, 4. https://pubmed.ncbi.nlm.nih.gov/17623102/
4. Barroggi Constantino, D., Lederle, K. A., Middleton, B., Revell, V. L., Sletten, T. L., Williams, P., Skene, D. J., & van der Veen, D. R. (2025). The bright and dark side of blue-enriched light on sleep and activity in older adults. GeroScience, 47(3), 3927–3939. https://pubmed.ncbi.nlm.nih.gov/39821044/
5. Verma, A. K., Khan, M. I., Ashfaq, F., & Rizvi, S. I. (2023). Crosstalk between aging, circadian rhythm, and melatonin. Rejuvenation Research, 26(6), 229–241. https://pubmed.ncbi.nlm.nih.gov/37847148/
6. Eto, T., & Higuchi, S. (2023). Review on age-related differences in non-visual effects of light: melatonin suppression, circadian phase shift and pupillary light reflex in children to older adults. Journal of Physiological Anthropology, 42(1), 11. https://pubmed.ncbi.nlm.nih.gov/37355647/
7. Baser, K. H. C., Haskologlu, I. C., & Erdag, E. (2025). Molecular links between circadian rhythm disruption, melatonin, and neurodegenerative diseases: An updated review. Molecules, 30(9), 1888. https://pubmed.ncbi.nlm.nih.gov/40363695/
Related Reading
- Circadian Rhythm Disruption: How a Misaligned Internal Clock Fragments Sleep, Drives 3AM Wakeups, and Reduces Sleep Depth — the full overview of circadian sleep disruption, including melatonin, cortisol, body temperature, peripheral clocks, and early-morning waking
- How Does a Two-Hour Weekend Sleep Delay Desynchronize Your Peripheral Clocks for Days? — how repeated weekend schedule shifts create chronic circadian misalignment
- Can Rotating Night Schedules Permanently Desynchronize Your Peripheral Clocks? — how rotating night work affects peripheral clocks and metabolic timing
- Why Does Eastbound Jet Lag Last Longer Than Westbound — and What Determines Recovery Rate? — why travel direction changes the speed of circadian recovery
- Night Owl or Delayed Sleep Phase Disorder? How to Know the Difference — how delayed circadian timing differs from preference alone
- Which Circadian Mechanisms Change After 40 — and Which Ones Are Reversible? — the broader aging mechanisms that shift circadian timing and sleep depth
- Does Evening Exercise Delay Your Circadian Clock — or Is Disruption Smaller Than You Think? — how exercise timing affects the circadian clock and sleep onset
- Does Time-Restricted Eating Entrain Your Peripheral Clocks? — how meal timing signals peripheral clocks outside the brain
