Staying up late is a preference. Being unable to fall asleep before 2-4am despite wanting to, despite structure, despite effort — and having that pattern persist for months with accumulating daytime consequences — is a circadian rhythm disorder.
The two look similar from the outside. Both involve late bedtimes, difficulty waking for morning obligations, and a pull toward nighttime alertness. But the mechanisms, functional consequences, and appropriate responses differ. A 2024 review reinforced that delayed sleep phase disorder requires documented functional impairment for at least three months — not just late sleep timing (Narala et al., 2024).
This article covers how the two conditions differ, the genetic basis of delayed sleep phase disorder, what the evidence says about melatonin and light exposure, and whether the delay changes with age. For the broader picture of how circadian disruption contributes to fragmented sleep, see the Circadian Rhythm Disruption overview on circadian sleep disruption.
What Is the Difference Between a Night Owl and Delayed Sleep Phase Disorder?
Reid et al. (2012) tested this distinction in a cross-sectional study comparing 48 individuals with delayed sleep phase disorder against 25 evening-type controls. Both groups had similar chronotypes on the Morningness-Eveningness Questionnaire. The evening-type controls chose to sleep late — but they could adjust when they needed to, and their daytime function was intact. The delayed sleep phase disorder group could not adjust.
Both groups had high rates of psychiatric comorbidity — over 70% of participants met criteria for at least one past Axis-I disorder, and roughly 40% of each group met criteria for past mood, anxiety, or substance-use disorders. The prevalence and type of Axis-I disorders did not differ between the groups. This finding suggests that evening chronotype itself — regardless of whether it meets the criteria for delayed sleep phase disorder — carries psychiatric risk.
This is the core distinction. The International Classification of Sleep Disorders, Third Edition (ICSD-3) defines delayed sleep phase disorder by three criteria: a habitual sleep-wake timing delay of at least two hours relative to desired or required times, persistence for at least three months, and functional impairment or distress (Narala et al., 2024). A preference for late nights that does not cause impairment does not meet these criteria, regardless of how late the person stays up.
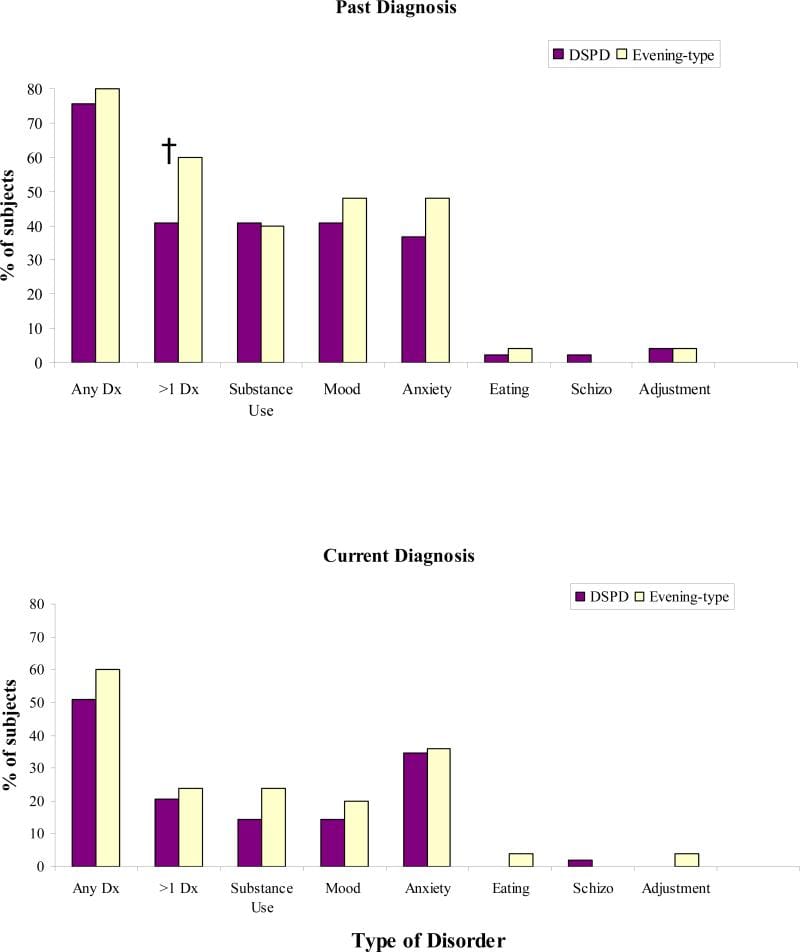
The psychiatric comorbidity finding is relevant in a different way than expected. In the Reid data, both groups had comparably high rates of psychiatric conditions — the evening chronotype itself was the shared risk factor, not the disorder classification. This suggests that the circadian delay common to both groups may independently contribute to mood instability, and that the functional impairment criterion separating the two is about the ability to entrain to a conventional schedule, not about psychiatric burden.
One practical distinction: an evening-type person who works a schedule that accommodates late sleep — and sleeps well on that schedule — is not experiencing a disorder. A person with delayed sleep phase disorder who is allowed to sleep on their preferred schedule may sleep well in terms of architecture, but still experiences impairment because their internal clock is misaligned with social demands that cannot be restructured.
How Is Delayed Sleep Phase Disorder Diagnosed?
Clinical diagnosis involves ruling out other sleep disorders — insomnia, sleep apnea, and mood disorders — that can independently delay sleep onset. A dim-light melatonin onset measurement, typically conducted with serial saliva samples collected in dim light the evening before a sleep study, documents the delayed circadian phase objectively. Actigraphy over two weeks establishes the habitual sleep-wake pattern. The combination of documented phase delay, functional impairment history, and exclusion of other causes meets the diagnostic threshold.
Is Delayed Sleep Phase Disorder Genetic?
Patke et al. (2017) identified the first specific genetic mutation linked to delayed sleep phase disorder. In a study published in Cell, the researchers traced a CRY1 splice-site mutation (c.1657+3A>C) through multiple families with delayed sleep phase disorder. The mutation causes exon 11 to be skipped during mRNA splicing, producing a modified CRY1 protein — CRY1-delta-11 — with an in-frame deletion of 24 residues that binds CLOCK/BMAL1 with greater affinity than the normal protein.
This enhanced binding is the mechanism. CRY1 is a repressor in the core molecular clock: it shuts down CLOCK/BMAL1-mediated transcription of Period and Cryptochrome genes, which is how the clock completes its daily cycle. When CRY1-delta-11 binds CLOCK/BMAL1 more tightly, repression lasts longer, and the circadian period extends. In cell-based assays, the mutation lengthened the period from 31.6 hours to 32.1 hours — a half-hour extension that, compounded over days, pushes sleep onset progressively later (Patke et al., 2017).
The carrier frequency matters. At 0.6% in European populations, this is not rare. In a population of 10,000 adults, roughly 60 could carry this variant — many of whom may be labeled “night owls” without the genetic context.
Plavc et al. (2024) expanded the genetic picture. In a study of familial sleep phase disorders, the researchers identified novel missense variants in PER3 linked to advanced sleep phase disorder and a rare PER2 variant in a proband with delayed sleep phase disorder. These findings demonstrate that clock gene variation runs in families and that genotyping can support distinguishing between preference and disorder (Plavc et al., 2024).
There is also pharmacogenomic relevance. Magee et al. (2020) conducted a randomized controlled trial in 104 individuals with delayed sleep phase disorder and found that clinical response to melatonin varied by PER3 genotype. Individuals with the PER3 4/4 variable number tandem repeat (VNTR) showed greater improvement in sleep outcomes — including insomnia severity and sleep onset latency — compared to PER3-5 carriers. Notably, melatonin did not advance circadian phase in either genotype group; the benefit was in sleep quality rather than clock shifting. This means the same dose of melatonin can produce different clinical outcomes depending on PER3 genotype — a step toward genotype-guided treatment (Magee et al., 2020).
Does Delayed Sleep Phase Disorder Run in Families?
The Plavc et al. (2024) family study reinforced this pattern, identifying novel clock gene variants in families with sleep phase disorders across multiple generations. For individuals trying to understand why their sleep timing resists change despite behavioral effort, family history of delayed sleep is a clinically relevant data point — it signals a genetic period-lengthening mechanism rather than a behavioral habit.
Can Melatonin and Light Exposure Advance a Delayed Clock?
Van Andel et al. (2022) tested this in a three-arm double-blind randomized controlled trial with 49 adults who had both ADHD and delayed sleep phase disorder. The three arms were: melatonin alone, melatonin plus morning bright light exposure, and placebo. Dim-light melatonin onset (DLMO) — the biomarker for circadian phase — advanced by approximately 1.5 hours with melatonin and approximately 2 hours with the combination.
But actigraphic sleep onset — the time participants fell asleep, measured by wrist-worn activity monitors — did not advance in either active arm. The circadian clock moved earlier. Behavior did not follow.
This dissociation between biomarker and behavior is an often-overlooked finding in delayed sleep phase disorder research. It means the clock and the sleep are not the same thing. Advancing the molecular clock is necessary but not sufficient. Behavioral factors — screen habits, evening light exposure, work schedules, social obligations — can anchor sleep timing in place even when the underlying circadian phase has moved.
Gomes et al. (2021) conducted a systematic review of light therapy for delayed sleep-wake phase disorder across five studies with 140 total participants. The studies used light intensities ranging from 225 to 10,000 lux, typically administered within 30 minutes of waking. Within-group comparisons showed improvements in sleep timing, but most studies found no statistically significant differences between intervention and control groups. The authors concluded that light therapy could be effective but that conclusive evidence remains limited. Sustained daily morning light exposure is inherently difficult for people whose circadian clock pushes them toward late sleep — they are groggy, sleep-deprived, and least motivated to sit in front of a light box at 7am (Gomes et al., 2021).
For individuals investigating how melatonin interacts with the orexin wake pathway, the timing of exogenous melatonin matters here too. In delayed sleep phase disorder, melatonin is typically administered 3-5 hours before habitual sleep onset — not at bedtime — to target the phase-advance portion of the melatonin phase-response curve. Bedtime dosing produces sedation but minimal circadian advancement.
Does Delayed Sleep Phase Disorder Change With Age?
Narala et al. (2024) reviewed the epidemiology and noted that delayed sleep phase disorder prevalence is highest in adolescents and young adults — estimated at 1-16% in this age group. This overlaps with the well-documented pubertal delay in circadian timing, where hormonal changes lengthen the circadian period and push sleep onset later. The question is which individuals revert to earlier timing as they age and which do not.
There is evidence for partial correction. As adults move into structured work schedules and caregiving responsibilities, external pressure can entrain the clock closer to a conventional schedule. But “entrained by pressure” and “resolved” are not the same. Many adults with delayed sleep phase disorder report lifelong difficulty with morning schedules — they function, but at a cost measured in chronic sleep debt, caffeine dependence, and the feeling of being perpetually out of phase.
For individuals with CRY1 or PER3 variants, the genetic contribution does not change with age. A 0.5-hour period extension from CRY1-delta-11 is present at 20 and at 60. What may change is the social and occupational context around the delay — retirement, for example, can reduce functional impairment by removing the morning schedule constraint without changing the underlying circadian timing.
The interaction between circadian delay and age-related changes in orexin-driven wake maintenance introduces an additional variable. As orexin neuron counts decline with age, the ability to sustain wakefulness through the early evening — which already extends later in delayed sleep phase disorder — may diminish, leading to a mismatch between circadian alertness and the capacity to stay awake.
Can You Correct Delayed Sleep Phase Disorder Permanently?
The CRY1 mutation identified by Patke et al. (2017) is inherited, present in every cell, and does not change. An individual who carries CRY1-delta-11 will have an extended circadian period for life. The management question is whether consistent environmental and pharmacological inputs can hold the clock at an earlier phase.
The van Andel et al. (2022) data suggest this is partially achievable. Melatonin and bright light exposure moved the circadian biomarker earlier, even if behavioral sleep timing did not follow in the short term. The implication is that long-term management requires both circadian inputs (melatonin, light) and behavioral support (consistent wake times, reduced evening light, schedule structure).
Consistency is the key requirement. Unlike a medication that addresses a static target, circadian management requires daily environmental inputs because the clock resets every 24 hours. Missing morning light exposure for several days can allow the phase to delay again. This is not a limitation of the approach — it is the nature of a clock that runs longer than 24 hours.
Is Delayed Sleep Phase Disorder a Disability?
The Reid et al. (2012) data showed that both individuals with delayed sleep phase disorder and evening-type controls had high rates of mood and anxiety disorders — over 70% met criteria for at least one past Axis-I condition. This psychiatric comorbidity, combined with chronic sleep restriction when forced onto conventional schedules, can produce measurable impairment in occupational performance, cognitive function, and quality of life.
In practice, accommodation requests for delayed sleep phase disorder may include flexible start times, remote work options, or modified schedules. The strength of documentation depends on whether the individual has objective circadian markers — such as a dim-light melatonin onset measurement showing a delayed phase — and evidence of functional impairment over at least three months.
What Is the Best Non-Drug Approach for Delayed Sleep Phase Disorder?
Gomes et al. (2021) conducted a systematic review of light therapy for delayed sleep-wake phase disorder. The parameters used across studies were:
- Intensity: 225-10,000 lux (higher intensities may allow shorter exposure)
- Duration: 15-60 minutes
- Timing: Within 30 minutes of habitual wake time
- Consistency: Daily use; effects diminish within days of stopping
Evening light restriction is the complementary approach. Blue-enriched light from screens, overhead LEDs, and ambient room lighting suppresses melatonin onset and reinforces the circadian delay. For more on how blue light affects circadian timing, the mechanism involves melanopsin receptors in intrinsically photosensitive retinal ganglion cells, which are maximally sensitive to light in the 460-480nm range.
Meal timing is an emerging area. Peripheral clocks in the liver, gut, and adipose tissue respond to food intake as a zeitgeber (time cue). Restricting food intake to a consistent daytime window may reinforce central clock entrainment, though the evidence in delayed sleep phase disorder is preliminary.
The challenge with non-drug approaches is that they require the delayed individual to do the hardest thing at the hardest time: expose themselves to bright light and maintain a consistent wake time when their clock is telling them to sleep. Compliance is an inherent difficulty in this population — the treatment timing conflicts directly with the disorder’s core symptom.
What Lux Level Is Needed for Morning Light Therapy to Work?
Light therapy devices marketed for circadian use typically deliver 10,000 lux at a specified distance (usually 20-30 cm from the face). Compliance at this proximity for 30-60 minutes is the limiting factor. Outdoor exposure — a 20-minute walk in morning sunlight — delivers comparable or higher intensity without the proximity constraint, and adds exercise timing as a secondary circadian signal.
A delayed circadian clock may compound with other causes of sleep disruption — hormonal changes, metabolic imbalance, or autonomic activation. Identifying which causes might be contributing is a useful next step.
Find out which causes might be driving your 3 AM wakeups →
Related Reading
- Circadian Rhythm Disruption: How a Misaligned Internal Clock Fragments Sleep, Drives 3AM Wakeups, and Reduces Sleep Depth — the full overview of circadian sleep disruption, including melatonin, cortisol, body temperature, peripheral clocks, and early-morning waking
- How Does a Two-Hour Weekend Sleep Delay Desynchronize Your Peripheral Clocks for Days? — how repeated weekend schedule shifts create chronic circadian misalignment
- Can Rotating Night Schedules Permanently Desynchronize Your Peripheral Clocks? — how rotating night work affects peripheral clocks and metabolic timing
- Why Does Eastbound Jet Lag Last Longer Than Westbound — and What Determines Recovery Rate? — why travel direction changes the speed of circadian recovery
- Why Does Your Wake Time Advance by Decades — and Can You Reverse Circadian Phase Advance After 60? — why aging shifts wake time earlier and which parts remain reversible
- Which Circadian Mechanisms Change After 40 — and Which Ones Are Reversible? — the broader aging mechanisms that shift circadian timing and sleep depth
- Does Evening Exercise Delay Your Circadian Clock — or Is Disruption Smaller Than You Think? — how exercise timing affects the circadian clock and sleep onset
- Does Time-Restricted Eating Entrain Your Peripheral Clocks? — how meal timing signals peripheral clocks outside the brain
References
- Reid, K. J., Jaksa, A. A., Eisengart, J. B., Baron, K. G., Lu, B., Kane, P., Kang, J., & Zee, P. C. (2012). Systematic evaluation of Axis-I DSM diagnoses in delayed sleep phase disorder and evening-type circadian preference. Sleep Medicine, 13(9), 1171–1177. https://pubmed.ncbi.nlm.nih.gov/22910327/
- Gomes, J. N., Dias, C., Brito, R. S., Lopes, J. R., Oliveira, I. A., Silva, A. N., & Salles, C. (2021). Light therapy for the treatment of delayed sleep-wake phase disorder in adults: a systematic review. Sleep Science, 14(2), 155–163. https://pubmed.ncbi.nlm.nih.gov/34381579/
- Magee, M., Sletten, T. L., Murray, J. M., Gordon, C. J., Lovato, N., Bartlett, D. J., Kennaway, D. J., Lockley, S. W., Lack, L. C., Grunstein, R. R., Archer, S. N., & Rajaratnam, S. M. W. (2020). The PER3 polymorphism rs57875989 moderates melatonin treatment effects on sleep outcomes in delayed sleep-wake phase disorder. Journal of Pineal Research, 69(4), e12684. https://pubmed.ncbi.nlm.nih.gov/32682347/
- Narala, B., Ahsan, M., Ednick, M., & Kier, C. (2024). Delayed sleep wake phase disorder in adolescents: an updated review. Current Opinion in Pediatrics, 36(1), 124–132. https://pubmed.ncbi.nlm.nih.gov/38054481/
- Patke, A., Murphy, P. J., Onat, O. E., Krieger, A. C., Özçelik, T., Campbell, S. S., & Young, M. W. (2017). Mutation of the human circadian clock gene CRY1 in familial delayed sleep phase disorder. Cell, 169(2), 203–215.e13. https://pubmed.ncbi.nlm.nih.gov/28388406/
- Plavc, L., Skubic, C., Dolenc Grošelj, L., & Rozman, D. (2024). Variants in the circadian clock genes PER2 and PER3 associate with familial sleep phase disorders. Chronobiology International, 41(5), 757–766. https://pubmed.ncbi.nlm.nih.gov/38695651/
- van Andel, E., Bijlenga, D., Vogel, S. W. N., Beekman, A. T. F., & Kooij, J. J. S. (2022). Attention-deficit/hyperactivity disorder and delayed sleep phase syndrome in adults: a randomized clinical trial on the effects of chronotherapy on sleep. Journal of Biological Rhythms, 37(6), 673–689. https://pubmed.ncbi.nlm.nih.gov/36181304/
