“No exercise after 6pm” is advice that predates the evidence. Exercise is a bona fide circadian time cue — a non-photic zeitgeber with a mapped phase-response curve — and the rules governing when exercise helps or harms sleep are more specific than the blanket recommendation. A 2019 landmark study mapped when exercise advances and delays the human circadian clock. A 2025 mega-study of over four million nights quantified the dose-response relationship between exercise strain and sleep disruption. And a 2020 RCT revealed that the same evening workout produces opposite circadian outcomes depending on chronotype. This article covers what those studies found, how muscle clocks respond to training timing independently of the brain, and what the data mean for scheduling workouts. For the broader picture of how circadian disruption causes 3 AM wakeups, see Circadian Sleep Disruption.
Does the Human Circadian Clock Have a Phase Response Curve to Exercise?
Youngstedt et al. (2019) enrolled 101 participants — 53 older adults aged 59-75 and 48 younger adults aged 18-32 — in an ultrashort sleep-wake schedule with one-hour moderate treadmill bouts at each of eight circadian time points. Clock phase was assessed via the acrophase, onset, and duration of urinary 6-sulphatoxymelatonin (aMT6s), the primary urinary metabolite of melatonin.
The resulting phase-response curves revealed three distinct zones. Exercise at 7:00am and between 1:00pm and 4:00pm produced peak phase advances — the clock moved earlier. Exercise between 7:00pm and 10:00pm produced peak phase delays — the clock moved later. And exercise at 4:00am and 10:00am fell near crossover points where minimal phase change occurred.
The magnitude of these exercise-induced phase changes was comparable to bright-light exposure of equal duration. Bright light is the best-established circadian time cue in humans, and exercise producing comparable effects positions it as a potent non-photic zeitgeber — a time cue that entrains the clock through a pathway independent of retinal light exposure.
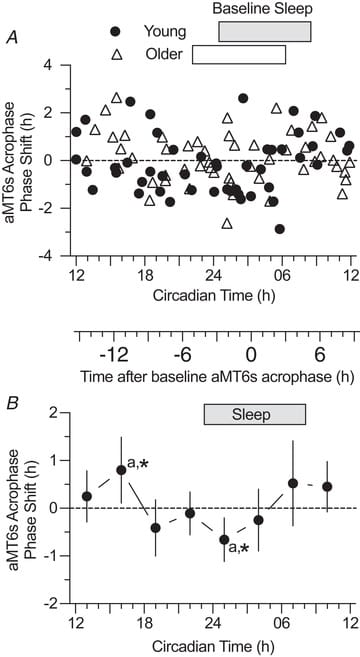
No differences in phase-response curve shape or amplitude were found between older and younger adults, or between sexes — the exercise PRC is consistent across age groups and sex. For people recovering from jet lag, adapting to rotating work schedules, or managing delayed sleep phase, exercise timing can be used strategically to advance or delay the clock: morning exercise to advance, evening exercise to delay.
Does Evening High-Intensity Interval Training Disrupt Sleep — and What Does the Largest Study Show?
Leota et al. (2025) published the largest dataset ever assembled on evening exercise and sleep: 14,689 physically active participants tracked across 4,084,354 person-nights over one year using wrist-worn biometric devices that measured both exercise strain and sleep outcomes.
The dose-response findings separated strain from timing. High-strain exercise finishing four or more hours before habitual sleep onset produced no detectable sleep disruption. But maximal-strain exercise ending within two hours of sleep onset was associated with an 80-minute delay in sleep onset, a 42.6-minute reduction in sleep duration (13.9% decrease), a 5.6 percentage-point decrease in sleep quality score, a 15.0% increase in nocturnal resting heart rate (+9.4 bpm), and a 32.6% decrease in heart rate variability. The autonomic arousal markers indicate that the disruption is not just perceptual — sympathetic activation from maximal-strain exercise persists into the sleep window. Moderate-strain exercise in the same two-hour pre-sleep window produced minimal effects, confirming that strain intensity, not clock time, is the variable driving the disruption.
A 2023 review by Kim et al. confirmed the pattern from a different angle: across nine controlled studies involving 201 participants, evening exercise consistently delayed melatonin rhythm onset and elevated nocturnal core body temperature, yet produced no negative effects on sleep efficiency or non-REM sleep architecture. No differences in sleep quality outcomes were found between high-intensity interval exercise and moderate-intensity continuous exercise. Core body temperature from any exercise returned to baseline within 30-120 minutes post-exercise — the thermoregulatory explanation for sleep disruption is time-limited and manageable with adequate buffer time.
Moderate evening exercise is compatible with sleep. Maximal-effort training within two hours of bedtime is not.
Does Your Chronotype Determine Whether Evening Exercise Helps or Hurts?
Thomas et al. (2020) enrolled 52 young sedentary adults and randomized them to five consecutive days of morning or evening treadmill exercise, measuring circadian phase via dim light melatonin onset (DLMO) before and after the five-day period.
Morning exercise produced a mean phase advance of 0.62 ± 0.18 hours across all participants. Evening exercise produced a mean phase change of -0.02 ± 0.18 hours — no net change when averaged across all chronotypes. But averaging across chronotypes hid the variable that mattered.
When chronotype was accounted for, the evening exercise data split in opposite directions. Late chronotypes experienced an approximately 0.5-hour phase advance from both morning and evening exercise — their clocks moved earlier regardless of when they trained. Early chronotypes experienced a 0.49-hour advance from morning exercise but a 0.41-hour delay from evening exercise — their clocks moved later when they trained in the evening.
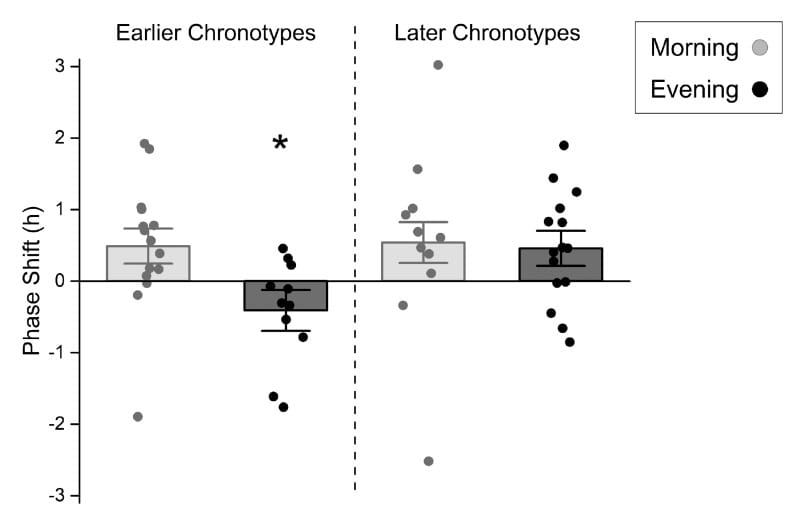
The same exercise at the same clock time produced opposite circadian outcomes depending on chronotype. For an early chronotype, evening exercise compounds circadian misalignment by pushing the clock later. For a late chronotype, evening exercise may provide a corrective advance that would not come from morning exercise alone. Chronotype self-assessment using validated tools like the Munich Chronotype Questionnaire (MCTQ) or corrected mid-sleep on free days (MSFsc) is a useful starting point for personalized exercise timing decisions.
Can Exercise Timing Entrain Your Peripheral Muscle Clocks?
Kemler, Wolff, and Esser (2020) used Period2::Luciferase mouse models and C2C12 myotubes to map how contraction timing changes the phase of the peripheral muscle clock — independent of the suprachiasmatic nucleus (SCN) master clock in the hypothalamus.
The phase-response data from skeletal muscle explants showed a time-of-day dependent pattern. Morning contraction at ZT5 (five hours after light-dark cycle onset) produced a 100-minute phase advance. Evening contraction at ZT11 produced a 62-minute phase delay. Nighttime contraction at ZT17 produced no measurable phase change.
To test whether the muscle clock was responding to contraction itself or to hormonal and neural input from the brain, the researchers stimulated C2C12 myotubes electrically. In cells with no connection to circulating neurohumoral factors, electrical stimulation during peak Bmal1 expression produced 27-minute delays, and stimulation during the clock’s transition phase produced 50-minute advances. Contraction alone — without hormonal, neural, or temperature input — is sufficient to change muscle clock phase.
This provides a cellular mechanism for why morning and evening training produce different metabolic gene expression patterns in muscle tissue even in people who show no sleep disruption from evening exercise. The sleep disruption question and the peripheral clock question are separate. Even when evening exercise does not disrupt sleep, it is still moving the muscle clock in a phase-delaying direction — which affects downstream glucose uptake timing and lipid metabolism rhythms in skeletal muscle.
Exercise timing interacts with other circadian time cues. If your clock is already moved by light exposure, meal timing, or medication, the effect of exercise timing may compound or counteract those other time cues. Identifying which causes might be contributing to your sleep disruption is a useful next step.
Find out which causes might be driving your 3 AM wakeups →
Frequently Asked Questions
Should You Avoid Working Out Within Two Hours of Bedtime?
The two-hour threshold matters specifically for maximal-effort exercise — sprints, high-intensity intervals at maximum capacity, or competition-level output. Moderate-intensity exercise — a tempo run, a strength session at submaximal weight, a brisk walk — does not carry the same risk within that window.
Kim et al. (2023) found that elevated core body temperature from any exercise returns to baseline within 30-120 minutes post-exercise — the primary thermoregulatory mechanism cited for sleep disruption. The practical rule: moderate evening exercise is fine. All-out effort sessions within two hours of bedtime are not. If you train hard late, finishing at least four hours before sleep onset eliminates the disruption based on the Leota et al. (2025) dose-response data.
Does Morning Exercise Improve Sleep More Than Evening Exercise?
Shen et al. (2025) randomized 58 sedentary males into morning exercise (6-8am), evening exercise (6-8pm), or sedentary control groups for 12 weeks. Both exercise groups shortened sleep latency compared to their own baseline, with the sedentary control showing no change.
Morning exercise uniquely advanced DLMO and moved the sleep-wake phase earlier, consistent with the advance zone of the exercise phase-response curve. Morning exercise also reduced body fat as early as week four and decreased total cholesterol and triglycerides. Evening exercise did not advance DLMO but produced superior vascular outcomes: greater blood flow rate, more effective systolic blood pressure reduction, and enhanced hemodynamic function. Both groups decreased arterial stiffness. Exercise timing does not determine whether adaptation occurs, but it determines which physiological pathway is preferentially activated — metabolic and circadian for morning, vascular for evening.
Can Exercise Timing Address Delayed Sleep Phase?
Lee et al. (2024) synthesized 17 studies examining timed exercise for circadian clock adjustment in people with sleep phase disorders. Morning exercise produced the largest benefit for delayed sleep phase (where the clock needs advancing), while evening exercise could benefit those with advanced sleep phase who need to delay their clock — consistent with the delay zone of the exercise phase-response curve mapped by Youngstedt et al. (2019).
Resistance training of a single limb upregulated core clock genes Cry1, Per2, and Bmal1 in the trained leg but not the untrained control leg. This is direct human evidence that local exercise stimulus entrains peripheral clocks — the same mechanism Kemler et al. (2020) demonstrated in isolated mouse muscle cells, now confirmed in human tissue.
In rotating-schedule worker studies, nocturnal cycling exercise produced measurable adjustments in melatonin oscillation aligned with a 9-hour phase delay in the sleep-wake cycle. Age, sex, and fitness level all affected the physiological response, which means personalized timing plans outperform one-size-fits-all recommendations. The review positioned exercise-timed clock resetting as a behavioral complement to melatonin and light therapy — an additive behavioral tool.
References
- Youngstedt, S. D., Elliott, J. A., & Kripke, D. F. (2019). Human circadian phase-response curves for exercise. The Journal of Physiology, 597(8), 2253–2268. https://pubmed.ncbi.nlm.nih.gov/30784068/
- Thomas, J. M., Kern, P. A., Bush, H. M., McQuerry, K. J., Black, W. S., Clasey, J. L., & Pendergast, J. S. (2020). Circadian rhythm phase shifts caused by timed exercise vary with chronotype. JCI Insight, 5(3), e134270. https://pubmed.ncbi.nlm.nih.gov/31895695/
- Kemler, D., Wolff, C. A., & Esser, K. A. (2020). Time-of-day dependent effects of contractile activity on the phase of the skeletal muscle clock. The Journal of Physiology, 598(17), 3631–3644. https://pubmed.ncbi.nlm.nih.gov/32537739/
- Kim, N., Ka, S., & Park, J. (2023). Effects of exercise timing and intensity on physiological circadian rhythm and sleep quality: A systematic review. Physical Activity and Nutrition, 27(3), 52–63. https://pubmed.ncbi.nlm.nih.gov/37946447/
- Lee, K., Hong, K.-S., Park, J., & Park, W. (2024). Readjustment of circadian clocks by exercise intervention is a potential therapeutic target for sleep disorders: A narrative review. Physical Activity and Nutrition, 28(2), 35–42. https://pubmed.ncbi.nlm.nih.gov/39097996/
- Leota, J., Presby, D. M., Le, F., Czeisler, M. E., Mascaro, L., Capodilupo, E. R., Wiley, J. F., Drummond, S. P. A., Rajaratnam, S. M. W., & Facer-Childs, E. R. (2025). Dose-response relationship between evening exercise and sleep. Nature Communications, 16, 3297. https://pubmed.ncbi.nlm.nih.gov/40234380/
- Shen, B., Zheng, H., Liu, H., Chen, L., & Yang, G. (2025). Differential benefits of 12-week morning vs. evening aerobic exercise on sleep and cardiometabolic health: A randomized controlled trial. Scientific Reports, 15, 18298. https://pubmed.ncbi.nlm.nih.gov/40419564/
Related Reading
- Circadian Rhythm Disruption: How a Misaligned Internal Clock Fragments Sleep, Drives 3AM Wakeups, and Reduces Sleep Depth — the full overview of circadian sleep disruption, including melatonin, cortisol, body temperature, peripheral clocks, and early-morning waking
- How Does a Two-Hour Weekend Sleep Delay Desynchronize Your Peripheral Clocks for Days? — how repeated weekend schedule shifts create chronic circadian misalignment
- Can Rotating Night Schedules Permanently Desynchronize Your Peripheral Clocks? — how rotating night work affects peripheral clocks and metabolic timing
- Why Does Eastbound Jet Lag Last Longer Than Westbound — and What Determines Recovery Rate? — why travel direction changes the speed of circadian recovery
- Night Owl or Delayed Sleep Phase Disorder? How to Know the Difference — how delayed circadian timing differs from preference alone
- Why Does Your Wake Time Advance by Decades — and Can You Reverse Circadian Phase Advance After 60? — why aging shifts wake time earlier and which parts remain reversible
- Which Circadian Mechanisms Change After 40 — and Which Ones Are Reversible? — the broader aging mechanisms that shift circadian timing and sleep depth
- Does Time-Restricted Eating Entrain Your Peripheral Clocks? — how meal timing signals peripheral clocks outside the brain
