Compounded semaglutide formulations can differ from brand-name Ozempic in quality, inactive ingredients, dosing format, and added ingredients — factors that may change tolerability and sleep indirectly. Analytical testing found novel impurities in injectable follow-on semaglutide products and lower-than-labeled semaglutide content in several oral follow-on products; FDA has also noted that some compounded semaglutide products include added ingredients such as vitamin B12. FDA adverse event data shows compounded GLP-1 formulations have higher reporting odds for several gastrointestinal events, which can disrupt sleep through nausea, reflux, abdominal discomfort, or delayed gastric emptying.
Many Americans now use compounded semaglutide obtained through telehealth platforms and compounding pharmacies, often at a lower advertised cost than brand-name products. Sleep-specific complaints in this population are not well quantified; primary care records documented compounded GLP-1 use in 8.2% of semaglutide or tirzepatide users, compared with survey estimates that about 23% of users received these drugs from compounders (Hendrix et al., 2025).
This article examines the differences between compounded and brand-name semaglutide formulations that may produce distinct sleep side effects — including additives, potency variation, and delivery route differences. It does not cover all GLP-1 sleep effects; for the full overview, see the parent article on Ozempic and sleep.
GLP-1 drugs can disrupt sleep through several distinct pathways. The question here is whether formulation differences amplify or alter those effects. Other articles in this series cover orexin-driven wakefulness, blood sugar instability, and appetite-related disruption.
How Does Compounded Semaglutide Differ From Brand-Name Ozempic
Compounded semaglutide is not an FDA-approved generic copy of Ozempic. It is a pharmacy-prepared, non-FDA-approved product that has not undergone FDA review for safety, effectiveness, quality, or bioequivalence. Analytical testing of 16 injectable and 8 oral follow-on semaglutide products found new impurity patterns in injectable products and lower-than-labeled semaglutide content in several oral products compared with originator products (Hach et al., 2024).
Compounding pharmacies produce semaglutide under two legal frameworks. 503A pharmacies compound patient-specific prescriptions individually. 503B outsourcing facilities are FDA-registered and follow stricter manufacturing standards, producing larger batches under more oversight. Direct-to-consumer marketing through telehealth platforms and wellness clinics can make the source and oversight pathway less transparent to consumers.
Hach et al. (2024) tested injectable and oral follow-on semaglutide products against originator products and found new impurities in injectable products, including high molecular weight proteins, trace metals, and residual solvents. Neoepitopes were identified in semaglutide follow-on products, indicating potential immunogenicity. Several oral follow-on products contained markedly less semaglutide than stated on the label.
In 2025, the American Diabetes Association issued a position statement: compounded formulations bypass manufacturing quality controls, sterility standards, and the evidence review required of FDA-approved drugs. Their safety profile cannot be assumed equivalent to brand-name semaglutide (Neumiller et al., 2025).
How widespread is this market A survey of 93 Colorado businesses advertising compounded GLP-1 products found that 41 referenced FDA approval in descriptions of their compounded products. Five described their products as generic, which is inaccurate because compounded drugs are not FDA-approved generics and have not undergone FDA bioequivalence review (DiStefano et al., 2025; FDA, 2026).
The documentation gap extends to primary care. In a study of 153,044 records, only 8.2% of compounded GLP-1 use was documented — well below the estimated 23% prevalence based on surveys. Primary care teams may lack visibility into compounded GLP-1 use, creating gaps in medication reconciliation, safety monitoring, and adverse event reporting (Hendrix et al., 2025).
Could Compounded Semaglutide Additives Affect Your Sleep
Some compounded semaglutide formulations include vitamin B12 (cyanocobalamin), L-carnitine, or other additives not present in brand-name Ozempic. Small human studies suggest high-dose vitamin B12 can affect activity, sleep time, and melatonin rhythms, but evening injection effects with semaglutide have not been studied. These additives have not been tested in combination with semaglutide for their effects on sleep.
Vitamin B12 (cyanocobalamin) was the most common advertised additive in the Colorado market survey. In the Colorado market survey, 8 of 93 businesses advertised B12-semaglutide combinations (DiStefano et al., 2025). The first published study of compounded semaglutide used a B12 co-formulated product — meaning every participant received both compounds simultaneously, making it difficult to separate the effects of each (Chun et al., 2025).
B12 has independent neurological activity relevant to sleep. High-dose vitamin B12 has been reported in small human studies to alter melatonin-related circadian measures and nighttime activity; direct cortisol effects from evening compounded semaglutide-B12 injections have not been established (Honma et al., 1992; Mayer et al., 1996). A person injecting compounded semaglutide with B12 in the evening may be adding a compound with circadian activity close to sleep onset.
L-carnitine was advertised by at least one Colorado business as part of a compounded GLP-1 formulation. L-carnitine shuttles fatty acids into mitochondria for energy production, but its sleep effects in combination with semaglutide have not been tested.
BPC-157 appeared in one business’s offerings. FDA has identified potential safety risks for BPC-157, including possible immunogenicity and peptide impurity/API-characterization concerns; FDA’s April 22, 2026, 503A update says BPC-157 was removed from category 2 because the nominations were withdrawn, with FDA consultation still planned.
FDA has stated that the safety and effectiveness of combining semaglutide with added ingredients such as cyanocobalamin, pyridoxine, levocarnitine, or NAD has not been established; the ADA’s 2025 statement also raised safety, quality, and efficacy concerns for compounded incretin products (Neumiller et al., 2025; FDA, 2024).
Compounded products may use different containers, concentrations, excipients, or added ingredients than brand-name Ozempic. These differences may affect stability, dosing, tolerability, or adverse-event patterns, although direct sleep outcomes have not been tested.
Does Dosing Accuracy in Compounded Semaglutide Change Sleep Side Effects
Compounded semaglutide products show measurable variation in actual peptide content versus what is stated on the label. Some products are subpotent, delivering less semaglutide than intended. Others may deliver inconsistent doses between injections. Because nausea, reflux, and other adverse effects can vary with dose and exposure, unpredictable potency may mean less predictable sleep-related tolerability.
Hach et al. (2024) found that several oral follow-on semaglutide products contained markedly less semaglutide than their labels stated. This means the user could receive less semaglutide than expected at a given titration step. If product potency or exposure changes, side effects that affect sleep — especially nausea, reflux, or abdominal discomfort — may change as well.
Dissolution testing revealed that compounded oral products released semaglutide and its absorption enhancers at different rates than the branded reference — meaning altered bioavailability and plasma levels that do not match the expected pharmacokinetic curve for a given dose (Hach et al., 2024).
Delivery route adds another variable. Seven of 93 Colorado businesses advertised oral compounded semaglutide formulations (DiStefano et al., 2025). A review of 17 pharmacokinetic studies established that oral semaglutide bioavailability depends on food intake, water volume, and co-formulation with SNAC (sodium N-[8-(2-hydroxybenzoyl)amino]caprylate) — conditions that are specific to brand-name Rybelsus and are unlikely to be replicated in compounded sublingual products (Yang & Yang, 2024).
Semaglutide exposure is commonly described using pharmacokinetic measures such as Cmax, but direct evidence linking Cmax to sleep effects is limited. Variable potency or absorption may change exposure, which may change the timing or intensity of tolerability effects. Someone tracking sleep may see patterns that are harder to interpret when formulation, dose, additives, or absorption vary.
Compounded products also often use different dose-escalation schedules than the FDA-approved titration schedule. Some users may reach higher effective doses faster than intended, triggering adverse effects that can disturb sleep earlier than expected.
What Does the Safety Data Show About Compounded Semaglutide
FDA adverse event data shows compounded GLP-1 formulations have higher reporting odds for gastrointestinal adverse events, hospitalization, and medication/product-quality errors compared with non-compounded products. Hospitalization was reported with more than twice the odds among compounded-product reports compared with non-compounded-product reports. Gastrointestinal events that can disrupt sleep through nausea, reflux, abdominal pain, or delayed gastric emptying were elevated in several categories (McCall et al., 2026).
McCall et al. (2026) analyzed 81,078 GLP-1 adverse event reports in the FAERS database, of which 707 involved compounded products. The results show a different pharmacovigilance profile.
Compounded formulations had elevated reporting odds ratios for gastrointestinal events: abdominal pain ROR 2.84, diarrhea ROR 1.59, and nausea ROR 1.27. All three can disrupt sleep through nocturnal discomfort and arousal.
Hospitalization had higher reporting odds with compounded formulations (ROR 2.35, 95% CI: 1.94-2.83), suggesting greater severity among reported cases while not proving population incidence.
Medication and quality errors were more common with compounded products: preparation errors ROR 48.92, contamination ROR 19.00, and compounding issues ROR 8.51. These are medication-use, manufacturing, or handling problems that can produce unpredictable dosing and unexpected reactions.
A suicidality finding (ROR 6.34, 95% CI: 4.32-8.99) was also identified. This may reflect confounding factors — compounded users may differ demographically from brand-name users — but it is a psychiatric safety signal that qualified prescribers should be aware of (McCall et al., 2026).
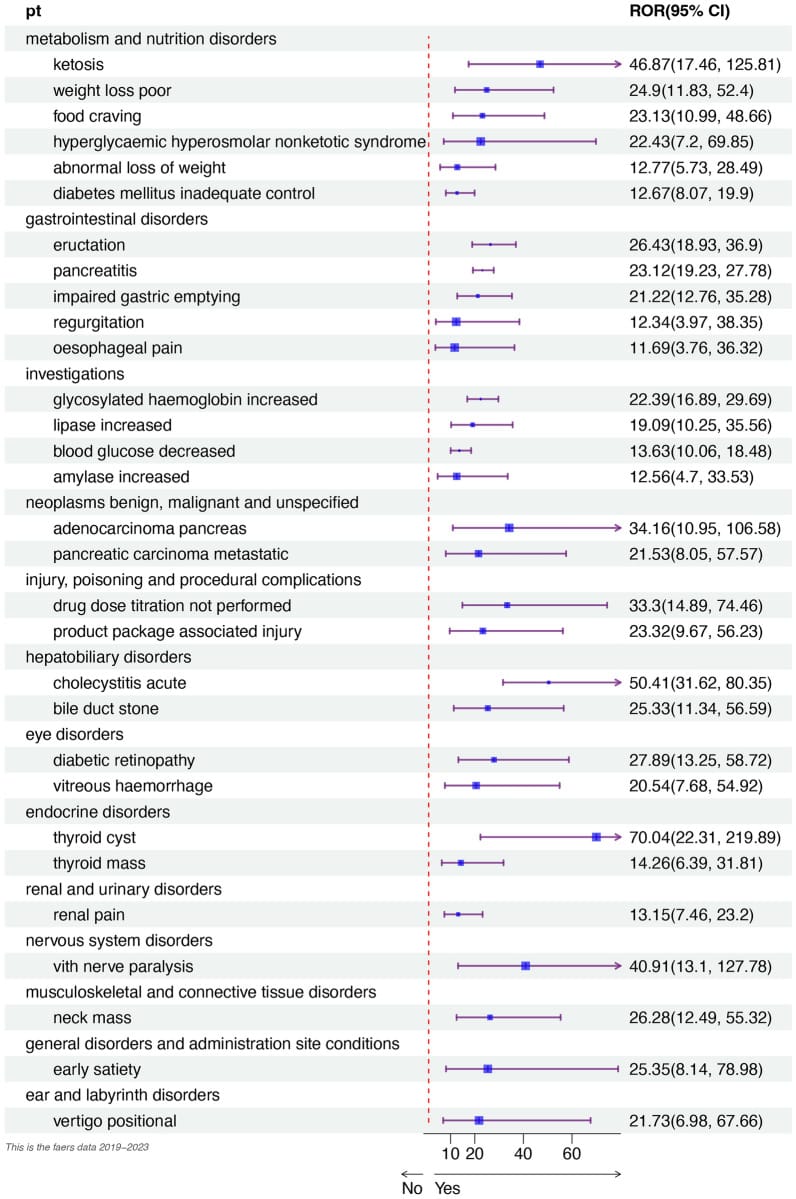
A separate FAERS analysis of brand-name oral semaglutide identified impaired gastric emptying and regurgitation as adverse-event signals (Xiong et al., 2024) — both of which can plausibly disturb sleep when symptoms occur at night. Compounded formulations with higher GI adverse-event reporting odds may increase the likelihood that these symptoms affect sleep.
GLP-1 receptor agonists can affect digestive, cardiovascular, neurological, immune, and respiratory systems; direct evidence for compounded-product-specific sleep-wake effects remains limited (He et al., 2025). Compounded products that deliver variable doses or contain untested additives add uncertainty to these effects.
And the pharmacovigilance picture is incomplete. Because compounded GLP-1 use may be underdocumented in medical records — 8.2% documented versus surveys reporting about 23% of users receiving medications from compounders — adverse events from compounded products may be less visible to routine care teams. FAERS data cannot estimate true population-level incidence, and underdocumentation may make compounded-product safety signals harder to interpret (Hendrix et al., 2025).
Formulation differences are one factor in a multi-factor problem. Whether you use compounded or brand-name semaglutide, GLP-1 drugs also affect blood sugar stability, orexin-driven arousal, appetite-related neurotransmitter production, and circadian timing — each of which may compound the effects of additives or dosing variability. Sleep disruption from metabolic causes often overlaps with hormonal, circadian, or autonomic factors.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Is Compounded Semaglutide as Effective as Brand-Name Ozempic for Weight Loss
One small study of compounded semaglutide co-formulated with B12 showed 4.57% weight loss at 3 months — lower than the about 15% mean weight loss reported with pharmaceutical-grade semaglutide 2.4 mg in STEP 1 over 68 weeks. Whether this difference reflects subpotency, shorter duration, or the uncontrolled setting is unclear. No head-to-head comparison exists (Chun et al., 2025).
Chun et al. (2025) followed 94 participants receiving weekly subcutaneous injections escalating from 0.25 mg to 2.4 mg. After 3 months, average weight loss was 4.11 kg (4.57%), with fat mass decreasing by 2.67 kg. The 3-month versus 68-week timeframe makes direct comparison with STEP trial results unreliable.
The weight-loss difference may matter for sleep apnea: less weight loss may mean less improvement in obstructive sleep apnea severity. GLP-1 receptor agonists may reduce sleep apnea severity largely through weight loss, while possible direct respiratory effects remain unresolved (Mifsud et al., 2025).
Does Compounded Semaglutide Cause More Nausea That Disrupts Sleep
FDA pharmacovigilance data shows compounded GLP-1 formulations have a higher reporting odds ratio for nausea (ROR 1.27), abdominal pain (ROR 2.84), and diarrhea (ROR 1.59) compared to brand-name products. Nausea is a plausible sleep disruptor among GLP-1 users because it can prevent comfortable sleep positioning and increase arousal (McCall et al., 2026).
Nocturnal nausea can increase discomfort and arousal, which may reduce sleep quality independently of the GLP-1 drug itself. A person experiencing nausea at 2 or 3 AM may have more difficulty returning to sleep because discomfort and arousal are present at the same time. With compounded formulations showing higher GI adverse event rates, this pattern may occur more frequently than with brand-name Ozempic. For the full GI-sleep pathway discussion, see the parent article on Ozempic and sleep.
Can You Switch From Compounded to Brand-Name Ozempic if Sleep Worsens
Switching from compounded to brand-name semaglutide requires a qualified prescriber who can write for FDA-approved products and insurance that covers them — or willingness to pay cash. The dosing may not translate directly because compounded products may have been delivering more or less semaglutide than labeled. A qualified prescriber can help establish the equivalent dose and manage the transition.
The ADA recommends reinstating brand-name therapy when shortage conditions resolve (Neumiller et al., 2025). The practical barrier is cost: brand-name and compounded cash prices can vary by insurance coverage, pharmacy, dose, savings eligibility, and product source.
If sleep disruption is worse on a compounded formulation, the first question is whether the dose is equivalent. Someone on a subpotent compounded product may be functionally at a lower dose than they think — switching to brand-name at the “same” labeled dose could mean a larger increase in actual semaglutide exposure than expected, temporarily worsening sleep side effects.
Are Sublingual Semaglutide Formulations Different for Sleep Than Injectable
Sublingual and oral compounded semaglutide products have different absorption characteristics than injectable formulations. Brand-name oral semaglutide (Rybelsus) uses the absorption enhancer SNAC and requires specific dosing conditions to achieve adequate exposure. Compounded oral products may not replicate these conditions, which could produce variable exposure and less predictable tolerability effects (Yang & Yang, 2024).
SNAC (sodium N-[8-(2-hydroxybenzoyl)amino]caprylate) is the absorption enhancer in brand-name Rybelsus that supports semaglutide absorption after oral dosing. Without an effective absorption strategy, oral semaglutide would be expected to have very low bioavailability.
Compounded sublingual products are intended to absorb through the oral mucosa rather than through the stomach. No published study has characterized the plasma concentration curve for sublingual semaglutide. Variable absorption may mean variable peak concentration, which may mean variable timing and intensity of tolerability effects (Yang & Yang, 2024).
Does the Pharmacy Source of Compounded Semaglutide Affect Side Effects
Yes. 503B outsourcing facilities registered with the FDA follow stricter manufacturing standards than 503A pharmacies compounding patient-specific prescriptions. Direct-to-consumer marketing through telehealth platforms and wellness clinics can make the actual compounding source harder to evaluate. The FAERS data showing elevated adverse events does not distinguish between these sources, but the quality control differences are structural (DiStefano et al., 2025).
503A pharmacies compound prescriptions for individuals based on a specific prescriber’s order. 503B facilities function more like small pharmaceutical manufacturers — larger batches, FDA registration, current good manufacturing practice (cGMP) requirements.
The direct-to-consumer market can obscure which compounding framework, if any, is being used. The Colorado market survey found businesses advertising unapproved peptides like BPC-157, referencing FDA approval in ways that could mislead consumers, and describing compounded GLP-1 products as generic — a characterization that has no regulatory basis (DiStefano et al., 2025).
If you are using compounded semaglutide and experiencing sleep disruption, knowing whether your pharmacy is a 503A, 503B, or unregulated source is relevant for evaluating whether formulation quality may be a contributing factor.
Related Reading
- Metabolic Sleep Disruption — The full metabolic sleep framework covering glucose regulation, insulin resistance, mitochondrial decline, NAD+ loss, and fat metabolism
- Does Ozempic Cause Insomnia What GLP-1 Drugs Do to Your Sleep — The broad overview of GLP-1 sleep side effects and mechanisms
- Why Does Ozempic Make You Tired But Keep You Awake — How GLP-1 drugs activate orexin/wakefulness circuitry while appetite suppression can create fatigue
- Why Does Ozempic Wake You Up at 3am — How lower overnight fuel availability and counter-regulatory hormones can contribute to 3am waking
- Does Ozempic Give You Vivid Dreams — How GLP-1 drugs may affect dream intensity through arousal and neuropsychiatric pathways
- Does Eating Too Little on Ozempic Cause Insomnia — How appetite suppression can affect tryptophan, serotonin, melatonin, and overnight glucose stability
- Does Your Ozempic Injection Day Disrupt Your Sleep The 24-48 Hour Pattern — Why sleep disruption can follow a weekly pattern tied to injection timing and peak drug exposure
- Does Your Semaglutide Dose Increase Restart Your Insomnia Why Each Titration Can Disrupt Sleep Again — Why each titration step can restart sleep disruption before adaptation catches up
- Can You Take Melatonin, Magnesium, or Sleep Aids While on Ozempic — How melatonin, magnesium, DORAs, and other sleep aids intersect with GLP-1 physiology
- Does Ozempic Give You Anxiety at Night Why GLP-1 Drugs May Contribute to Nighttime Panic — How orexin activation, HPA-axis signaling, and glucose instability may contribute to nighttime anxiety
- Do GLP-1 Drugs Disrupt Your Body Clock Ozempic, Circadian Rhythms, and Melatonin — How GLP-1 secretion rhythms, food timing, peripheral clocks, and melatonin intersect
- Ozempic Night Sweats: How GLP-1 Drugs May Affect Temperature at Night — How GLP-1 drugs may affect nighttime sweating through autonomic tone and thermoregulation
- Ozempic Nausea and Sleep: How Stomach Side Effects Keep You Awake at Night — How delayed gastric emptying, reflux, and nausea can fragment sleep
- Will Your Sleep Go Back to Normal When You Stop Ozempic — What is known and not known about sleep after semaglutide discontinuation
- Does Ozempic Muscle Loss Affect Your Sleep Sarcopenia, Temperature, and Deep Sleep — How lean-mass loss, thermoregulation, and deep sleep can interact during GLP-1 weight loss
- Could Ozempic Contribute to Restless Legs at Night Iron, B12, and the Dopamine Connection — How iron, B12, dopamine, and rapid weight loss may intersect with restless legs at night
- Ozempic and Sleep After 40: Why GLP-1 Side Effects Can Feel Different as You Age — Why GLP-1 sleep effects can land differently after 40 because of muscle, hormones, and glucose regulation
References
- Chun, E., Siojo, A., Rivera, D., Reyna, K., Legere, H., Joseph, R., & Pojednic, R. (2025). Weight loss and body composition after compounded semaglutide treatment in a real world setting. Diabetes, Obesity & Metabolism, 27(3), 1536-1543. https://pubmed.ncbi.nlm.nih.gov/39776038/
- DiStefano, M. J., Dardouri, M., Moore, G. D., Saseen, J. J., & Nair, K. V. (2025). Compounded glucagon-like peptide-1 receptor agonists for weight loss: The direct-to-consumer market in Colorado. Journal of Pharmaceutical Policy and Practice, 18(1), 2441220. https://pubmed.ncbi.nlm.nih.gov/39776466/
- Hach, M., Engelund, D. K., Mysling, S., Mogensen, J. E., Schelde, O., Haselmann, K. F., Lamberth, K., Vilhelmsen, T. K., Malmstrøm, J., Højlys-Larsen, K. B., Rasmussen, T. S., Borch-Jensen, J., Mortensen, R. W., Jensen, T. M. T., Kesting, J. R., Catarig, A. M., Asgreen, D. J., Christensen, L., & Staby, A. (2024). Impact of manufacturing process and compounding on properties and quality of follow-on GLP-1 polypeptide drugs. Pharmaceutical Research, 41(10), 1991-2014. https://pubmed.ncbi.nlm.nih.gov/39379664/
- He, X., Zhao, Z., Sun, Y., & Jiang, X. (2025). Unraveling the safety profile of GLP-1 receptor agonists: Mechanistic insights with a focus on semaglutide. European Journal of Medicinal Chemistry, 300, 118163. https://pubmed.ncbi.nlm.nih.gov/40975962/
- Hendrix, N., Velásquez, E. E., Pham, H., & Bazemore, A. (2025). Documentation of compounded GLP-1 receptor agonists in a large primary care dataset. Pharmacoepidemiology and Drug Safety, 34(10), e70227. https://pubmed.ncbi.nlm.nih.gov/41024632/
- Honma, K., Kohsaka, M., Fukuda, N., Morita, N., & Honma, S. (1992). Effects of vitamin B12 on plasma melatonin rhythm in humans: Increased light sensitivity phase-advances the circadian clock Experientia, 48(8), 716-720. https://pubmed.ncbi.nlm.nih.gov/1516676/
- Mayer, G., Kröger, M., & Meier-Ewert, K. (1996). Effects of vitamin B12 on performance and circadian rhythm in normal subjects. Neuropsychopharmacology, 15(5), 456-464. https://pubmed.ncbi.nlm.nih.gov/8914118/
- McCall, K. L., Mastro Dwyer, K. A., Casey, R. T., Samana, T. N., Sulicz, E. K., Tso, S. Y., Yalanzhi, E. R., & Piper, B. J. (2026). Safety analysis of compounded GLP-1 receptor agonists: A pharmacovigilance study using the FDA adverse event reporting system. Expert Opinion on Drug Safety, 25(3), 581-588. https://pubmed.ncbi.nlm.nih.gov/40285721/
- Mifsud, C. S., Kolla, B. P., Rushlow, D. R., & Mansukhani, M. P. (2025). The impact of GLP-1 agonists on sleep disorders: Spotlight on sleep apnea. Expert Opinion on Pharmacotherapy, 26(14-15), 1529-1538. https://pubmed.ncbi.nlm.nih.gov/41114602/
- Neumiller, J. J., Bajaj, M., Bannuru, R. R., McCoy, R. G., Pekas, E. J., Segal, A. R., & ElSayed, N. A. (2025). Compounded GLP-1 and dual GIP/GLP-1 receptor agonists: A statement from the American Diabetes Association. Diabetes Care, 48(2), 177-181. https://pubmed.ncbi.nlm.nih.gov/39620926/
- U.S. Food and Drug Administration. (2024, July 26). FDA alerts health care providers, compounders and patients of dosing errors associated with compounded injectable semaglutide products. https://www.fda.gov/drugs/human-drug-compounding/fda-alerts-health-care-providers-compounders-and-patients-dosing-errors-associated-compounded
- U.S. Food and Drug Administration. (2026, March 3). FDA warns 30 telehealth companies against illegal marketing of compounded GLP-1s. https://www.fda.gov/news-events/press-announcements/fda-warns-30-telehealth-companies-against-illegal-marketing-compounded-glp-1s
- U.S. Food and Drug Administration. (2026, April 22). Bulk drug substances nominated for use in compounding under section 503A. https://www.fda.gov/media/94155/download
- U.S. Food and Drug Administration. (n.d.). FDA’s concerns with unapproved GLP-1 drugs used for weight loss. https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/medications-containing-semaglutide-marketed-type-2-diabetes-or-weight-loss
- U.S. Food and Drug Administration. (n.d.). Understanding the risks of compounded drugs. https://www.fda.gov/drugs/human-drug-compounding/understanding-risks-compounded-drugs
- Wilding, J. P. H., Batterham, R. L., Calanna, S., Davies, M., Van Gaal, L. F., Lingvay, I., McGowan, B. M., Rosenstock, J., Tran, M. T. D., Wadden, T. A., Wharton, S., Yokote, K., Zeuthen, N., Kushner, R. F., & STEP 1 Study Group. (2021). Once-weekly semaglutide in adults with overweight or obesity. The New England Journal of Medicine, 384(11), 989-1002. https://pubmed.ncbi.nlm.nih.gov/33567185/
- Xiong, S., Gou, R., Liang, X., Wu, H., Qin, S., Li, B., Luo, C., & Chen, J. (2024). Adverse events of oral GLP-1 receptor agonist (semaglutide tablets): A real-world study based on FAERS from 2019 to 2023. Diabetes Therapy, 15(8), 1717-1733. https://pubmed.ncbi.nlm.nih.gov/38776037/
- Yang, X.-D., & Yang, Y.-Y. (2024). Clinical pharmacokinetics of semaglutide: A systematic review. Drug Design, Development and Therapy, 18, 2555-2570. https://pubmed.ncbi.nlm.nih.gov/38952487/
Written by Kat Fu, M.S., M.S. – Last reviewed: May 2026 – 18 references cited
