In semaglutide trials, lean-mass reductions have ranged from near 0% to about 40% of total weight lost, with broader GLP-1-based therapy reviews showing wide variation. Skeletal muscle is one of the body’s major heat-generating tissues. When lean mass declines, thermogenic capacity may fall, and the evening temperature changes that support sleep onset may become less robust. That does not prove semaglutide-related muscle loss causes less deep sleep, but it gives a plausible pathway through which muscle loss could contribute to poorer sleep quality or more fragmented sleep.
Muscle loss on GLP-1 drugs is widely discussed in terms of strength and metabolism. What receives less attention is its effect on sleep architecture. The sleep consequences of sarcopenia – the progressive loss of muscle mass and function – are documented in aging and metabolic research, but that evidence has not yet been studied directly in people losing lean mass on semaglutide.
This article covers the mechanisms through which muscle loss on semaglutide may affect sleep quality: thermogenesis, cortisol, and myokine communication. It does not cover all GLP-1 sleep effects; for the full overview, see the parent article on GLP-1 drugs and sleep. Other articles in this series cover orexin activation, blood sugar crashes, vivid dreams, and appetite-related sleep disruption.
How Much Muscle Do You Lose on Ozempic – and Why Does It Matter for Sleep
In published trials, lean-mass reductions have ranged from near 0% to about 40% of total weight lost. In the SEMALEAN study, participants lost approximately 3 kg of lean mass in the first 7 months. In older adults with type 2 diabetes, semaglutide accelerated sarcopenia markers including grip strength decline and reduced gait speed over 24 months. Sarcopenia is an established independent risk factor for poor sleep quality in aging research – a connection that is relevant to drug-associated lean-mass loss, but has not yet been tested as a direct semaglutide-to-sleep pathway.
A pooled review of six semaglutide trials (n = 1,541) found that lean mass loss ranged from near 0% to 40% of total weight lost, depending on age, baseline body composition, and whether participants engaged in concurrent exercise (Bikou et al., 2024). The variation is wide, and the picture is not uniformly negative – but the upper end of that range represents several kilograms of lean tissue.
The SEMALEAN study tracked 115 people with obesity (mean BMI 46.3 kg/m2) on semaglutide 2.4 mg weekly (Alissou et al., 2026). Fat mass declined by 14% at month 7 and 18% at month 12. Lean mass dropped 3 kg at month 7, then stabilized – no further lean mass loss between months 7 and 12. Sarcopenic obesity prevalence in the cohort dropped from 49% to 33%, and handgrip strength improved by 4.5 kg over the full year. This suggests a phase-dependent pattern: early on, both compartments decline, but after month 7, lean mass stabilized between months 7 and 12 while fat mass continued to decline.
The picture changes in older adults. A 24-month retrospective cohort (n = 432) of older adults with type 2 diabetes found that semaglutide use produced measurable muscle mass index decline, grip strength loss, and gait speed reduction compared to matched controls (Ren et al., 2025). Grip strength showed a gender-divergent pattern: women declined continuously, while men improved initially then declined. Semaglutide dosage, baseline muscle mass index, and baseline gait speed were independent predictors of muscle mass change.
A secondary analysis from the SLIM LIVER trial documented a 9.3% decline in psoas muscle volume over 24 weeks of semaglutide use, though gait speed improved and the prevalence of slow gait dropped from 63% to 46% (Ditzenberger et al., 2025). This dissociation matters: structural muscle loss does not always map onto functional impairment.
Where this connects to sleep: sarcopenia is linked with poor sleep quality in older adults, with plausible pathways involving sleep fragmentation, inflammation, and endocrine changes in geriatric and metabolic populations. The following sections explain each mechanism.
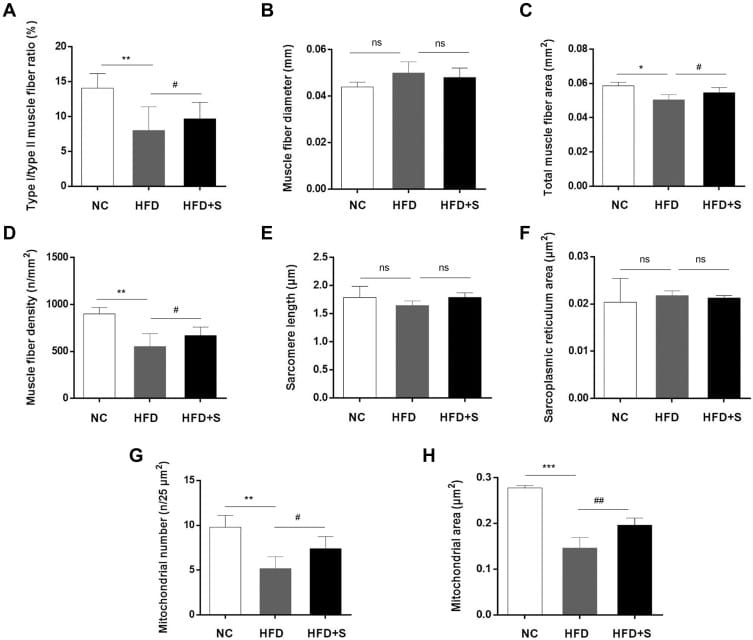
Figure 4. Semaglutide improved the lesions and microstructure of gastrocnemius in obese mice. Quantitative detection of (A) type I/type II muscle fiber ratio, (B) muscle fiber diameter, (C) total muscle fiber area, (D) muscle fiber density, (E) sarcomere length, (F) sarcoplasmic reticulum area, (G) mitochondrial area, and (H) mitochondrial number. Source: Ren et al. (2022), Drug Design, Development and Therapy, 16, 3723-3735. PMC9594960. CC BY-NC 3.0.
Does Losing Muscle on Semaglutide Reduce Your Deep Sleep
Skeletal muscle is a major heat-generating tissue, producing metabolic heat through both resting metabolism and non-shivering thermogenesis. Sleep onset is supported by heat dissipation and a decline in core body temperature. When muscle mass declines, baseline heat production may fall, which could make the normal evening temperature transition less robust.
Muscle as a Heat Source
Skeletal muscle contributes to resting energy expenditure and is one of the body’s major sites of glucose and fatty-acid metabolism. When muscle mass drops – as occurs in some semaglutide trials where lean-mass reductions reach the upper end of the near-0-to-40% range (Bikou et al., 2024) – the body’s baseline heat production falls. This matters because temperature regulates sleep onset.
The Temperature Gate for Deep Sleep
Sleep onset is closely tied to thermoregulation. Heat loss through the skin and the evening decline in core body temperature help the body move toward sleep, and stronger heat dissipation before bed is associated with shorter sleep-onset latency.
With less muscle generating metabolic heat during the day, thermogenic capacity may be lower. In someone already vulnerable to fragmented sleep, that could make the temperature transition into sleep less stable.
Brown Adipose Tissue Compensation
GLP-1 receptor agonist research suggests a possible brown adipose tissue (BAT) thermogenesis pathway, including hypothalamic VMH-AMPK pathway activity in animal and mechanistic models (Beiroa et al., 2014). In animal models, GLP-1 receptor agonists stimulate BAT activity and promote white adipocyte browning – generating additional heat through uncoupling protein 1 (UCP1) activation. This may partially compensate for heat production lost from declining skeletal muscle.
However, BAT volume in adult humans is limited and declines with age. The translational evidence for direct BAT activation by GLP-1 receptor agonists in humans remains sparse, and it is not yet known how much BAT thermogenesis offsets lean-mass loss during GLP-1 therapy in humans (Hropot et al., 2023). In adults over 40, age-related declines in skeletal muscle and BAT activity make this question more relevant.
The net effect: reduced total thermogenic capacity could make sleep-onset temperature regulation more vulnerable, especially when lean-mass loss occurs alongside low caloric intake, low protein intake, or low resistance training.
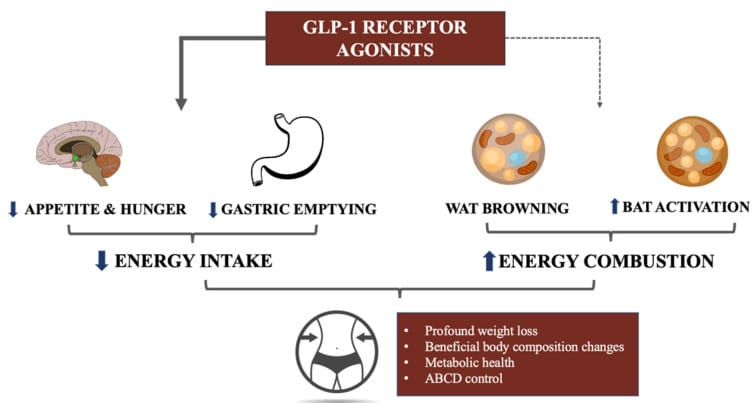
Figure 2. The potential mechanisms of GLP-1RAs action: from well-established effects in reducing energy intake combined to their potential role in increasing energy expenditure. Legend: WAT – white adipose tissue; BAT – brown adipose tissue; ABCD – adipose-based chronic disease. Source: Hropot et al. (2023), International Journal of Molecular Sciences, 24(10). PMC10218520. CC BY 4.0.
Does Ozempic Muscle Loss Increase Nighttime Cortisol and Sleep Fragmentation
Rapid weight loss can create catabolic pressure, especially when caloric intake, protein intake, and resistance training are low. Cortisol is one pathway the body uses to mobilize fuel, and elevated cortisol during the sleep period can interfere with sleep maintenance. At the same time, less contracting muscle may reduce exercise-linked myokine communication, including BDNF and IL-6 pathways that are involved in sleep regulation.
Muscle Breakdown and Cortisol
Rapid lean mass loss can reflect catabolic pressure. The body can elevate cortisol to supply amino acids for gluconeogenesis – producing glucose from non-carbohydrate sources, including amino acids stripped from muscle. This catabolic state may be amplified in caloric deficit, the standard metabolic environment on semaglutide given the appetite suppression the drug produces.
Cortisol normally reaches its overnight low-activity window during the early sleep period. Elevated nighttime cortisol reduces slow-wave sleep duration, increases the number of awakenings, and shortens total sleep time.
The Bidirectional Relationship Between Muscle Loss and Sleep
Poor sleep reduces growth hormone output – the largest daily growth-hormone pulse is usually linked to early slow-wave sleep – and can elevate cortisol. Both of these hormonal changes can contribute to muscle catabolism. This creates a self-reinforcing relationship: muscle loss may make sleep more vulnerable, and disrupted sleep may make lean-mass preservation harder.
In the context of GLP-1 therapy, where caloric intake is already reduced and lean mass is under catabolic pressure, poor sleep can reduce one recovery pathway. The risk factors for disproportionate lean mass loss – older age, high baseline fat mass, absence of resistance exercise (Neeland et al., 2024) – overlap with the risk factors for poor sleep quality.
Myokine Depletion
Skeletal muscle produces molecules called myokines during contraction. Two myokines relevant to sleep are BDNF (brain-derived neurotrophic factor) and IL-6. Both influence sleep regulation – BDNF is involved in sleep homeostasis and slow-wave activity, and exercise-induced IL-6 has anti-inflammatory effects distinct from the chronic IL-6 elevation seen in obesity.
Less resistance exercise or less contracting muscle may reduce contraction-linked myokine communication. That gives another plausible pathway, separate from thermoregulation and cortisol, through which lean-mass loss and low exercise exposure could affect sleep.
SIRT1 Pathway and Muscle Protection
In preclinical models, GLP-1 receptor agonists protect muscle through the SIRT1 deacetylase pathway – regulating muscle protein homeostasis through mitochondrial biogenesis and anti-inflammatory activity (Xiang et al., 2023a). Pharmacological inhibition of SIRT1 abolished all muscle-protective effects of both liraglutide and semaglutide. However, this protection has not been proven to offset every form of catabolic pressure during large human weight loss. In animal models, semaglutide improved muscle fiber density, type I/type II fiber ratio, and mitochondrial area (Ren et al., 2022) – but these measures come from controlled conditions, not the 15-17% body weight loss seen in human STEP trials.
Can Resistance Training Protect Your Sleep While on Ozempic
Resistance training is a primary modifiable method for preserving lean mass during GLP-1 receptor agonist use. Exercise can help preserve muscle thermogenic capacity, supports the core temperature rhythm required for deep sleep, and sustains myokine production (BDNF, IL-6) that is involved in sleep quality. Reviews of GLP-1 body composition data identify absence of resistance exercise as a modifiable risk factor for disproportionate lean mass loss on semaglutide.
Exercise as the Primary Countermeasure
Neeland et al. (2024) identify resistance training as the strongest modifiable factor for lean mass preservation during GLP-1 receptor agonist use – stronger than dietary protein alone. Absence of resistance exercise is listed as a modifiable risk factor for disproportionate lean mass loss, alongside older age, high baseline fat mass, and sarcopenic status at baseline.
Preserving Thermogenic Capacity
Maintaining muscle mass through resistance exercise preserves the body’s heat-generating capacity. This supports the core temperature oscillation – higher during waking hours, lower at sleep onset – that helps coordinate sleep initiation.
Sustaining Myokine Production
Resistance exercise directly stimulates BDNF and IL-6 release from contracting muscle fibers. This maintains the sleep-supportive molecules that are linked to contraction and exercise exposure. Even if total muscle mass decreases on semaglutide, regular contraction-driven myokine release can partially sustain sleep-relevant biochemical activity.
Protein Intake
Dietary protein intake of at least 1.2 g per kg body weight is commonly recommended alongside resistance training during intentional weight loss and GLP-1-based therapy (Neeland et al., 2024). Since semaglutide suppresses appetite, caloric reduction often means protein reduction unless intake is deliberately managed.
Exercise Timing and Temperature
Evening resistance exercise raises core body temperature, which then drops during the 1-2 hour post-exercise cooling period. This temperature drop can amplify the thermal gradient that initiates deep sleep – aligning the exercise countermeasure directly with the thermogenic mechanism.
Functional Preservation Data
In the Xiang et al. (2023b) study, participants on semaglutide maintained handgrip strength when the drug was combined with lifestyle changes over 24 weeks. In the SEMALEAN study, handgrip strength improved by 4.5 kg at 12 months despite lean mass decline (Alissou et al., 2026). These findings indicate that functional muscle capacity can be preserved with appropriate support, even when absolute lean mass decreases.
Muscle loss is one of several mechanisms through which GLP-1 drugs may affect sleep quality. Semaglutide also affects orexin-driven arousal, blood sugar regulation, appetite-related neurotransmitter production, and circadian timing – each of which may compound the effects of declining lean mass. Sleep disruption from metabolic causes often overlaps with hormonal, circadian, or autonomic factors.
Find out which causes might be driving your 3am wakeups ->
Frequently Asked Questions
Does Ozempic Cause More Muscle Loss Than Diet Alone
The proportion of lean mass lost on semaglutide – near 0% to about 40% of total weight lost across the semaglutide trials reviewed by Bikou et al. – is broadly consistent with what occurs during caloric restriction without medication. The pooled review by Bikou et al. (2024) found that semaglutide’s body composition profile is generally favorable, with participants ending the study period at a higher lean-to-fat ratio than they started with. The concern is the magnitude and speed of weight loss: losing 15-17% of body weight in a year means the absolute amount of lean tissue lost can be larger than in typical dieting, even if the proportion is similar.
The rate of weight loss, and the size of the total weight loss appear to be major drivers, while drug-specific effects on muscle remain under study. In the STEP trials, participants lost 15-17% of body weight over 68 weeks. At the upper end of the lean-mass-loss range, that pace translates to several kilograms of muscle tissue – consequential for thermogenesis, cortisol regulation, and sleep architecture.
Does Poor Sleep on Ozempic Accelerate Muscle Loss
Sleep deprivation reduces growth hormone release – which peaks during slow-wave sleep – and can elevate cortisol, both of which push the body toward muscle catabolism. In the context of GLP-1 therapy, where caloric intake is already reduced and lean mass is under pressure, poor sleep can reduce one recovery pathway. Muscle loss may make sleep more vulnerable, and disrupted sleep may make lean-mass preservation harder.
The largest daily growth-hormone pulse is usually linked to early slow-wave sleep. When slow-wave sleep is reduced – whether by thermoregulatory impairment, cortisol elevation, or other GLP-1-related mechanisms – growth hormone output can fall. Growth hormone promotes muscle protein synthesis and opposes cortisol-driven catabolism. Sleep quality is relevant to muscle preservation during GLP-1 therapy (Neeland et al., 2024).
Does Protein Intake on Ozempic Affect Both Muscle and Sleep
Dietary protein provides the amino acids required for muscle protein synthesis and also supplies tryptophan – the precursor to serotonin and melatonin. Reduced food intake on semaglutide often means reduced protein intake, which can simultaneously make it harder to support muscle preservation and sleep-promoting neurotransmitter production. Reviews recommend at least 1.2 g protein per kg body weight during GLP-1 therapy to protect lean mass.
Protein serves dual roles here. Amino acid availability determines whether muscle protein synthesis can keep pace with breakdown. Tryptophan – an essential amino acid in protein-rich foods – is the biochemical precursor to serotonin, which is then converted to melatonin. Lower protein intake may reduce tryptophan substrate availability for serotonin and melatonin production. Given semaglutide’s appetite suppression, protein intake requires active management (Neeland et al., 2024).
Is Ozempic Muscle Loss Worse in People Over 40
Age is a modifiable risk factor for disproportionate lean mass loss during GLP-1 therapy. The retrospective cohort study in older adults with type 2 diabetes found that semaglutide accelerated sarcopenia markers – including grip strength decline and gait speed reduction – over 24 months. Older adults start with lower muscle reserves, making each kilogram of lean mass lost more consequential for both function and sleep.
The Ren et al. (2025) data showed women experienced continuous grip strength decline, while men improved initially then declined. Baseline muscle mass index was an independent predictor of muscle mass change – people who started with less muscle lost more proportionally.
For adults over 40, age-related muscle decline can compound with semaglutide-associated lean-mass loss. Each kilogram of muscle lost has a larger proportional effect on thermogenic capacity, cortisol regulation, and myokine production when baseline reserves are already lower.
Does Mounjaro Cause Less Muscle Loss Than Ozempic
Tirzepatide (Mounjaro) is a dual GLP-1/GIP receptor agonist. Tirzepatide body-composition data show more fat-mass loss than lean-mass loss, but head-to-head data proving better muscle preservation than semaglutide are limited. Both drugs produce large weight loss, and the same risk factors for lean mass loss – older age, higher baseline fat mass, absence of resistance exercise – apply to both.
Whether tirzepatide’s GIP receptor activation translates to measurably different lean mass outcomes compared to semaglutide remains an open question – there are no published head-to-head trials with body composition as a primary endpoint. The countermeasures are the same regardless of which drug is prescribed: resistance training, adequate protein intake (at least 1.2 g/kg), and attention to sleep quality (Neeland et al., 2024).
Related Reading
- Metabolic Sleep Disruption — The full metabolic sleep framework covering glucose regulation, insulin resistance, mitochondrial decline, NAD+ loss, and fat metabolism
- Does Ozempic Cause Insomnia What GLP-1 Drugs Do to Your Sleep — The broad overview of GLP-1 sleep side effects and mechanisms
- Why Does Ozempic Make You Tired But Keep You Awake — How GLP-1 drugs activate orexin/wakefulness circuitry while appetite suppression can create fatigue
- Why Does Ozempic Wake You Up at 3am — How lower overnight fuel availability and counter-regulatory hormones can contribute to 3am waking
- Does Ozempic Give You Vivid Dreams — How GLP-1 drugs may affect dream intensity through arousal and neuropsychiatric pathways
- Does Eating Too Little on Ozempic Cause Insomnia — How appetite suppression can affect tryptophan, serotonin, melatonin, and overnight glucose stability
- Does Your Ozempic Injection Day Disrupt Your Sleep The 24-48 Hour Pattern — Why sleep disruption can follow a weekly pattern tied to injection timing and peak drug exposure
- Does Your Semaglutide Dose Increase Restart Your Insomnia Why Each Titration Can Disrupt Sleep Again — Why each titration step can restart sleep disruption before adaptation catches up
- Can You Take Melatonin, Magnesium, or Sleep Aids While on Ozempic — How melatonin, magnesium, DORAs, and other sleep aids intersect with GLP-1 physiology
- Does Ozempic Give You Anxiety at Night Why GLP-1 Drugs May Contribute to Nighttime Panic — How orexin activation, HPA-axis signaling, and glucose instability may contribute to nighttime anxiety
- Do GLP-1 Drugs Disrupt Your Body Clock Ozempic, Circadian Rhythms, and Melatonin — How GLP-1 secretion rhythms, food timing, peripheral clocks, and melatonin intersect
- Ozempic Night Sweats: How GLP-1 Drugs May Affect Temperature at Night — How GLP-1 drugs may affect nighttime sweating through autonomic tone and thermoregulation
- Ozempic Nausea and Sleep: How Stomach Side Effects Keep You Awake at Night — How delayed gastric emptying, reflux, and nausea can fragment sleep
- Will Your Sleep Go Back to Normal When You Stop Ozempic — What is known and not known about sleep after semaglutide discontinuation
- Could Ozempic Contribute to Restless Legs at Night Iron, B12, and the Dopamine Connection — How iron, B12, dopamine, and rapid weight loss may intersect with restless legs at night
- Does Compounded Semaglutide Affect Sleep Differently Than Brand-Name Ozempic — How dose variability, formulation differences, and adverse-event tracking can affect sleep side effects
- Ozempic and Sleep After 40: Why GLP-1 Side Effects Can Feel Different as You Age — Why GLP-1 sleep effects can land differently after 40 because of muscle, hormones, and glucose regulation
References
- Alissou, M., Demangeat, T., Folope, V., Van Elslande, H., Lelandais, H., Blanchemaison, J., Cailleaux, P. E., Guney, S., Aupetit, A., Aubourg, A., Rapp, C., Petit, A., Godin, M., Vignal, L., Grigioni, S., Dechelotte, P., Colange, G., Coeffier, M., & Achamrah, N. (2026). Impact of Semaglutide on fat mass, lean mass and muscle function in patients with obesity: The SEMALEAN study. Diabetes, Obesity & Metabolism, 28(1), 112-121. https://pubmed.ncbi.nlm.nih.gov/41068996/
- Balbo, M., Leproult, R., & Van Cauter, E. (2010). Impact of sleep and its disturbances on hypothalamo-pituitary-adrenal axis activity. International Journal of Endocrinology, 2010, 759234. https://pubmed.ncbi.nlm.nih.gov/20628523/
- Beiroa, D., Imbernon, M., Gallego, R., Senra, A., Herranz, D., Villarroya, F., Serrano, M., Ferno, J., Salvador, J., Escalada, J., Dieguez, C., Lopez, M., Fruhbeck, G., & Nogueiras, R. (2014). GLP-1 agonism stimulates brown adipose tissue thermogenesis and browning through hypothalamic AMPK. Diabetes, 63(10), 3346-3358. https://pubmed.ncbi.nlm.nih.gov/24917578/
- Bikou, A., Dermiki-Gkana, F., Penteris, M., Constantinides, T. K., & Kontogiorgis, C. (2024). A systematic review of the effect of semaglutide on lean mass: Insights from clinical trials. Expert Opinion on Pharmacotherapy, 25(5), 611-619. https://pubmed.ncbi.nlm.nih.gov/38629387/
- Cohn, A. Y., Grant, L. K., Nathan, M. D., Wiley, A., Abramson, M., Harder, J. A., Crawford, S., Klerman, E. B., Scheer, F. A. J. L., Kaiser, U. B., Rahman, S. A., & Joffe, H. (2023). Effects of sleep fragmentation and estradiol decline on cortisol in a human experimental model of menopause. The Journal of Clinical Endocrinology and Metabolism, 108(11), e1347-e1357. https://pubmed.ncbi.nlm.nih.gov/37207451/
- Ditzenberger, G. L., Lake, J. E., Kitch, D. W., Kantor, A., Muthupillai, R., Moser, C., Belaunzaran-Zamudio, P. F., Brown, T. T., Corey, K., Landay, A. L., Avihingsanon, A., Sattler, F. R., & Erlandson, K. M. (2025). Effects of Semaglutide on muscle structure and function in the SLIM LIVER study. Clinical Infectious Diseases, 80(2), 389-396. https://pubmed.ncbi.nlm.nih.gov/39046173/
- Faraguna, U., Vyazovskiy, V. V., Nelson, A. B., Tononi, G., & Cirelli, C. (2008). A causal role for brain-derived neurotrophic factor in the homeostatic regulation of sleep. The Journal of Neuroscience, 28(15), 4088-4095. https://pubmed.ncbi.nlm.nih.gov/18400908/
- Haghayegh, S., Khoshnevis, S., Smolensky, M. H., Diller, K. R., & Castriotta, R. J. (2019). Before-bedtime passive body heating by warm shower or bath to improve sleep: A systematic review and meta-analysis. Sleep Medicine Reviews, 46, 124-135. https://pubmed.ncbi.nlm.nih.gov/31102877/
- Hropot, T., Herman, R., Janez, A., Lezaic, L., & Jensterle, M. (2023). Brown adipose tissue: A new potential target for glucagon-like peptide 1 receptor agonists in the treatment of obesity. International Journal of Molecular Sciences, 24(10), 8592. https://pubmed.ncbi.nlm.nih.gov/37239935/
- Krauchi, K., & Wirz-Justice, A. (2001). Circadian clues to sleep onset mechanisms. Neuropsychopharmacology, 25(5 Suppl), S92-S96. https://pubmed.ncbi.nlm.nih.gov/11682282/
- Leproult, R., Copinschi, G., Buxton, O., & Van Cauter, E. (1997). Sleep loss results in an elevation of cortisol levels the next evening. Sleep, 20(10), 865-870. https://pubmed.ncbi.nlm.nih.gov/9415946/
- Look, M., Dunn, J. P., Kushner, R. F., Cao, D., Harris, C., Gibble, T. H., Stefanski, A., & Griffin, R. (2025). Body composition changes during weight reduction with tirzepatide in the SURMOUNT-1 study of adults with obesity or overweight. Diabetes, Obesity & Metabolism, 27(5), 2720-2729. https://pubmed.ncbi.nlm.nih.gov/39996356/
- Neeland, I. J., Linge, J., & Birkenfeld, A. L. (2024). Changes in lean body mass with glucagon-like peptide-1-based therapies and mitigation strategies. Diabetes, Obesity & Metabolism, 26 Suppl 4, 16-27. https://pubmed.ncbi.nlm.nih.gov/38937282/
- Piovezan, R. D., Abucham, J., Dos Santos, R. V., Mello, M. T., Tufik, S., & Poyares, D. (2015). The impact of sleep on age-related sarcopenia: Possible connections and clinical implications. Ageing Research Reviews, 23(Pt B), 210-220. https://pubmed.ncbi.nlm.nih.gov/26216211/
- Ren, Q., Chen, S., Chen, X., Niu, S., Yue, L., Pan, X., Li, Z., & Chen, X. (2022). An effective glucagon-like peptide-1 receptor agonists, semaglutide, improves sarcopenic obesity in obese mice by modulating skeletal muscle metabolism. Drug Design, Development and Therapy, 16, 3723-3735. https://pubmed.ncbi.nlm.nih.gov/36304787/
- Ren, Q., Zhi, L., & Liu, H. (2025). Semaglutide therapy and accelerated sarcopenia in older adults with type 2 diabetes: A 24-month retrospective cohort study. Drug Design, Development and Therapy, 19, 5645-5652. https://pubmed.ncbi.nlm.nih.gov/40631351/
- Rubio-Arias, J. A., Rodriguez-Fernandez, R., Andreu, L., Martinez-Aranda, L. M., Martinez-Rodriguez, A., & Ramos-Campo, D. J. (2019). Effect of sleep quality on the prevalence of sarcopenia in older adults: A systematic review with meta-analysis. Journal of Clinical Medicine, 8(12), 2156. https://pubmed.ncbi.nlm.nih.gov/31817603/
- St-Onge, M. P., Zuraikat, F. M., & Neilson, M. (2023). Exploring the role of dairy products in sleep quality: From population studies to mechanistic evaluations. Advances in Nutrition, 14(2), 283-294. https://pubmed.ncbi.nlm.nih.gov/36774251/
- Tan, X., van Egmond, L. T., Cedernaes, J., & Benedict, C. (2020). The role of exercise-induced peripheral factors in sleep regulation. Molecular Metabolism, 42, 101096. https://pubmed.ncbi.nlm.nih.gov/33045432/
- Van Cauter, E., & Copinschi, G. (2000). Interrelationships between growth hormone and sleep. Growth Hormone & IGF Research, 10 Suppl B, S57-S62. https://pubmed.ncbi.nlm.nih.gov/10984255/
- Van Cauter, E., Leproult, R., & Plat, L. (2000). Age-related changes in slow wave sleep and REM sleep and relationship with growth hormone and cortisol levels in healthy men. JAMA, 284(7), 861-868. https://pubmed.ncbi.nlm.nih.gov/10938176/
- Xiang, J., Qin, L., Zhong, J., Xia, N., & Liang, Y. (2023). GLP-1RA liraglutide and semaglutide improves obesity-induced muscle atrophy via SIRT1 pathway. Diabetes, Metabolic Syndrome and Obesity, 16, 2433-2446. https://pubmed.ncbi.nlm.nih.gov/37602204/
- Xiang, J., Ding, X. Y., Zhang, W., Zhang, J., Zhang, Y. S., Li, Z. M., Xia, N., & Liang, Y. Z. (2023). Clinical effectiveness of semaglutide on weight loss, body composition, and muscle strength in Chinese adults. European Review for Medical and Pharmacological Sciences, 27(20), 9908-9915. https://pubmed.ncbi.nlm.nih.gov/37916360/
Written by Kat Fu, M.S., M.S. – Last reviewed: May 2026 – 23 references cited
