It depends on which type of sleep disruption you experienced. Drug-dependent effects — especially GI side effects and possible GLP-1-related arousal effects — would be expected to ease as semaglutide exposure declines over about five weeks. Dream changes are reported anecdotally, but PubMed-indexed evidence directly tying semaglutide to vivid dreams or nightmares is limited. Weight-dependent effects — particularly sleep apnea improvement — can recur or worsen if weight is regained, although the relationship is not a one-to-one equation. No PubMed-indexed evidence found in this review suggests semaglutide causes permanent sleep-architecture changes after discontinuation.
“Does the insomnia stop when you stop the drug” appears in GLP-1 discontinuation discussions often, but PubMed-indexed trials do not directly answer this sleep-specific discontinuation question. The answer depends on which sleep mechanism was disrupted and whether that disruption was drug-mediated or weight-mediated.
This article covers the pharmacokinetic timeline for semaglutide clearance, the evidence on weight regain and sleep apnea recurrence, and a framework for predicting recovery speed based on the type of sleep disruption you experienced. GLP-1 drugs may affect sleep through several distinct pathways — each covered in separate articles in this series. This article addresses what happens to each when the drug is removed.
How Long After Stopping Ozempic Does Sleep Normalize
Semaglutide has a half-life of approximately one week. Near-complete washout generally takes about five half-lives, or roughly five weeks after the last injection. Sleep effects that are directly caused by the drug’s pharmacological action — possible GLP-1-related arousal, delayed gastric emptying or reflux, and appetite suppression — would be expected to begin easing as plasma concentrations decline, though individual sleep patterns can lag behind pharmacokinetic washout.
The 5-week timeline comes from semaglutide’s molecular design. A fatty acid chain binds the peptide to albumin in the bloodstream, contributing to slow elimination. This enables once-weekly dosing but means the drug lingers for weeks after the last injection (Yang & Yang, 2024).
During active drug exposure, GLP-1 receptor agonists can excite hypocretin/orexin neurons in mouse hypothalamic slice experiments; these neurons are part of arousal circuitry. In mouse brain slice experiments, exendin-4 (a GLP-1 agonist) increased orexin neuron spike frequency and reduced afterhyperpolarization currents. Sleep-promoting melanin-concentrating hormone neurons showed no response, indicating selectivity for hypocretin/orexin rather than MCH neurons in that model (Acuna-Goycolea & van den Pol, 2004).
When the drug leaves, this activation would be expected to decline if the sleep disruption was driven by receptor-mediated GLP-1 activity. The orexin effect was fully blocked by the GLP-1 antagonist exendin(9-39), establishing that the mechanism is receptor-mediated in the mouse slice model.
Here is what the washout curve looks like in practice:
- Weeks 1-2: Plasma concentration drops to ~25% of peak by the end of week 2. Appetite begins returning, GI side effects subside, reflux-driven and possible GLP-1-related arousal effects may begin improving.
- Weeks 3-4: Plasma falls below 10% of peak. Drug-dependent sleep effects should be noticeably reduced if drug exposure was the primary driver.
- Week 5: Near-complete washout. Drug-dependent sleep disruption should have substantially eased, while weight-dependent sleep changes follow body-weight and airway changes.
One important caveat: weight-related sleep effects, especially sleep apnea improvement, follow a different timeline.
Does Sleep Apnea Come Back When You Regain Weight After Ozempic
The data suggest that sleep apnea risk and severity can rise again if weight is regained. In the STEP 1 trial extension, participants regained approximately two-thirds of lost weight within one year of stopping semaglutide — net weight change fell from -17.3% to -5.6%. In the SURMOUNT-OSA trials, tirzepatide reduced sleep apnea severity by 20-24 events per hour, an improvement that occurred alongside weight loss. Because sleep apnea severity tracks with adiposity — particularly upper-airway and tongue fat — weight regain can increase the likelihood that sleep apnea severity returns.
The STEP 1 trial extension provides the best data on what happens after stopping semaglutide. Of the original 1,961 adults, 327 were followed through one year post-withdrawal. During the active phase, the semaglutide group lost a mean of 17.3% of body weight. One year after stopping, they had regained ~11.6 percentage points — roughly two-thirds of that loss — leaving a net change of only -5.6%. Cardiometabolic improvements (waist circumference, blood pressure, lipids, glycemia) reverted toward baseline (Wilding et al., 2022).
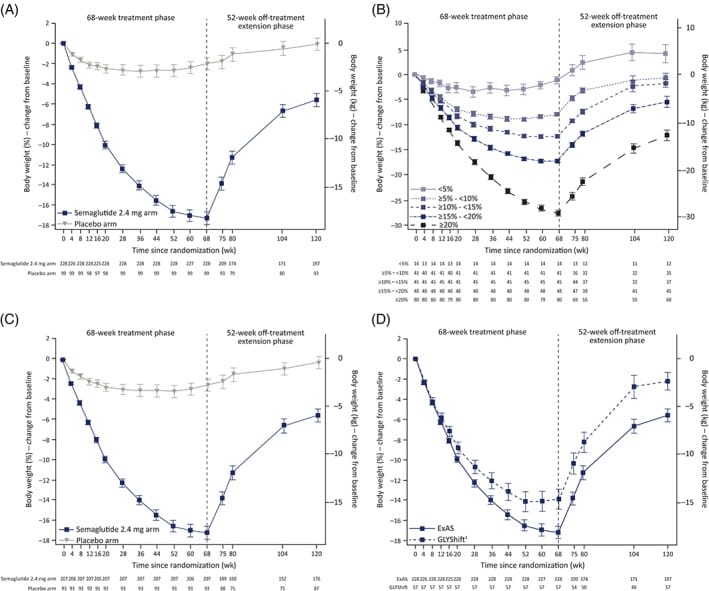
SURMOUNT-OSA trial data show the sleep apnea implications. In two parallel trials (469 adults with moderate-to-severe obstructive sleep apnea and obesity, mean baseline AHI ~50 events/hour), tirzepatide reduced the apnea-hypopnea index by 20.0 and 23.8 events/hour versus placebo at 52 weeks. Because this improvement is at least partly related to weight loss, AHI improvement may be at risk if weight is regained; no trial has directly tested proportional AHI reversal after stopping tirzepatide or semaglutide (Malhotra et al., 2024).
Two 2025 meta-analyses reinforce this pattern. Berg et al. pooled 8 RCTs (2,372 participants) and found 9.69 kg mean regain after stopping semaglutide or tirzepatide, versus 2.20 kg for liraglutide. Kolli et al., in a broader 36-study analysis, found semaglutide produced the largest post-discontinuation weight rebound among the agents/classes included in that analysis (reported mean difference -5.15 kg vs -3.06 kg for exenatide and -1.50 kg for liraglutide).
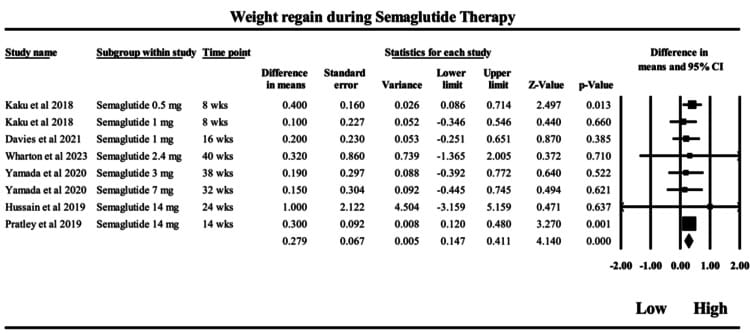
A compounding factor: in 71 adults with prediabetes, higher AHI was independently associated with lower endogenous GLP-1 response (B = -0.242, P = 0.045), even after adjusting for BMI (Reutrakul et al., 2017). This supports a possible metabolic-sleep relationship, but it does not prove that stopping semaglutide suppresses endogenous GLP-1 or creates a self-sustaining sleep-apnea cycle.
Can Ozempic Cause Permanent Changes to Your Sleep
No PubMed-indexed evidence found in this review suggests semaglutide causes permanent sleep-architecture changes after discontinuation. Direct receptor-mediated effects should diminish as semaglutide exposure falls, and the available orexin evidence comes from mouse slice experiments rather than human discontinuation studies.
The 2004 mouse brain slice data support receptor mediation: exendin(9-39), a GLP-1 antagonist, fully blocked the drug’s excitatory effect on orexin neurons (Acuna-Goycolea & van den Pol, 2004).
A 2025 narrative review of 13 RCTs found that weight regain was consistent across all durations of drug use — longer exposure did not produce durable metabolic reprogramming. That review is useful for weight-regain expectations, but it does not directly test sleep architecture or brain arousal after stopping semaglutide (Quarenghi et al., 2025).
Whether GLP-1 sleep benefits are all weight-mediated or also reflect direct drug actions on airway musculature or respiratory drive remains an open question (Mifsud et al., 2025). But even the direct-action hypothesis has not shown persistent sleep effects after discontinuation.
What can persist: if weight loss resolved sleep apnea and you maintain that weight independently through dietary changes, exercise, or transition to another therapy, the sleep apnea benefit can persist as long as the airway and body-composition changes persist. The persistence is in weight maintenance, not in the drug.
Does the Type of Sleep Disruption Determine How Fast It Resolves
Sleep disruption from GLP-1 drugs can be grouped into three categories, each with a different recovery timeline after stopping. Drug-driven disruption (possible GLP-1-related arousal, GI side effects, self-reported vivid dreams) is expected to ease across the 5-week drug clearance window. Weight-dependent disruption (sleep apnea improvement) can change over months as weight and upper-airway anatomy change. Nutritional disruption (lower tryptophan intake, blood sugar instability from undereating) may improve as appetite, protein intake, carbohydrate intake, and meal timing normalize.
Which Drug-Driven Sleep Effects Resolve as Semaglutide Clears
Each of the following is most likely to follow the pharmacokinetic washout curve described above (Yang & Yang, 2024):
- Possible GLP-1-related arousal: GLP-1 receptor activation can increase hypocretin/orexin neuron firing in mouse slice models. When receptor occupancy drops as the drug clears, this arousal pressure would be expected to decline if it is contributing to sleep disruption.
- GI-mediated sleep disruption: Semaglutide delays gastric emptying, which can contribute to nocturnal reflux, regurgitation, or nausea that may fragment sleep. As the drug clears, gastric-motility effects may improve.
- Self-reported vivid dreams: Direct PubMed-indexed evidence tying semaglutide to vivid dreams or nightmares is limited. If dream changes began with GLP-1 exposure and no other driver is present, the conservative expectation is gradual improvement as drug exposure falls.
Which Sleep Effects Reverse Proportionally to Weight Regain
Weight-dependent effects are tied to body composition rather than to the drug itself:
- Sleep apnea: AHI improvement tracks adiposity, particularly upper-airway and tongue fat. Li et al. (2025) pooled 6 trials (1,067 participants) and found GLP-1 drugs reduced AHI by 9.48 events/hour. If weight is regained, AHI can worsen again, but the amount of recurrence varies by airway anatomy and other OSA drivers.
- Possible direct respiratory effects: Mifsud et al. (2025) write that whether GLP-1 benefits are purely weight-mediated or partly direct remains an open question.
Which Sleep Effects Resolve as Appetite and Eating Normalize
Nutritional disruption stems from the appetite suppression that GLP-1 drugs produce:
- Lower tryptophan intake: Reduced food intake can mean less dietary tryptophan — the precursor to serotonin and melatonin. As appetite and protein intake return, tryptophan intake may normalize.
- Blood sugar instability: Undereating can contribute to nocturnal glucose dips; experimental hypoglycemia can trigger awakening and counterregulatory responses during sleep. As caloric and carbohydrate intake return to adequate levels, overnight glucose stability may improve.
Of the three categories, nutritional effects may be among the first to improve, because appetite can return before the drug fully leaves the body.
What Does the Recovery Timeline Look Like for Each Disruption Type
| Disruption type | Cause | Expected resolution timeline | What to monitor |
|—|—|—|—|
| Drug-driven | Possible GLP-1-related arousal, GI reflux, self-reported vivid dreams | 1-5 weeks (follows drug clearance if drug-dependent) | Sleep onset latency, dream intensity, nighttime reflux |
| Weight-dependent | Sleep apnea improvement from fat loss | Months (tracks weight and airway changes) | Snoring recurrence, daytime sleepiness, AHI if tested |
| Nutritional | Lower tryptophan intake, blood sugar instability | Days to weeks (as eating normalizes) | 3am waking frequency, sleep onset difficulty |
Whether you are still on a GLP-1 drug, tapering off, or already stopped, your sleep disruption likely has multiple overlapping causes — some pharmacological, some metabolic, some nutritional. Understanding which pathways are active in your case helps estimate which changes may ease as the drug clears and which ones need a separate plan.
Find out which causes might be driving your 3am wakeups.
Frequently Asked Questions
How Long Does Semaglutide Stay in Your Body After Your Last Injection
Semaglutide has a half-life of approximately one week. One week after your last injection, roughly half the drug remains in your bloodstream. Near-complete washout takes approximately five half-lives, or about 35 days (5 weeks). During this washout period, the drug’s effects on appetite, GI motility, and possible brain arousal circuits gradually diminish rather than stopping abruptly.
A C-18 fatty acid chain binds semaglutide to albumin in plasma, shielding it from enzymatic degradation. This makes once-weekly dosing possible but means the drug takes over a month to nearly clear from your body. Body weight may affect clearance rate, with lower-weight individuals potentially reaching higher relative plasma concentrations per dose (Yang & Yang, 2024).
Will Ozempic Dreams and Nightmares Stop When You Discontinue
Vivid dreams and nightmares are reported by some GLP-1 users, but direct PubMed-indexed evidence tying semaglutide to dream changes is limited. If dream changes began with semaglutide and no other driver is present, they would be expected to ease as semaglutide exposure falls across the roughly five-week washout. This review found no PubMed-indexed evidence that GLP-1 drugs cause permanent changes to dream physiology.
The stronger evidence is pharmacokinetic: semaglutide exposure declines gradually. If a dream change is drug-dependent, the timeline is more likely to be gradual than abrupt (Yang & Yang, 2024).
Does Stopping Ozempic Abruptly Affect Sleep Differently Than Tapering
No established tapering guidelines exist for semaglutide, and no study has compared abrupt versus gradual cessation for sleep outcomes. Quarenghi et al. (2025) reviewed 13 RCTs and found weight regain patterns consistent regardless of drug-use duration, suggesting the metabolic rebound is driven by removing pharmacological appetite suppression, not by withdrawal speed.
Abrupt cessation means semaglutide exposure declines gradually over roughly five weeks. Tapering would theoretically produce a more gradual step-down. No PubMed-indexed study found in this review has tested whether tapering changes sleep outcomes after semaglutide discontinuation (Quarenghi et al., 2025; Yang & Yang, 2024).
Can You Keep the Sleep Benefits of Ozempic Weight Loss After Stopping
Sleep benefits tied to weight loss — specifically sleep apnea improvement — have the highest chance of persisting. If you maintain the weight you lost through dietary changes, exercise, or another therapy, reduced sleep apnea severity can persist as long as the airway and body-composition changes persist because the mechanism (reduced upper-airway fat) is structural. However, STEP 1 showed extension participants regained approximately two-thirds of lost weight within one year without pharmacotherapy.
Drug-dependent benefits (reduced possible GLP-1-related arousal, fewer GI awakenings) are expected to fade when the drug leaves the body. Weight-dependent benefits (sleep apnea reduction, thermoregulation) persist as long as the weight loss persists. Given that STEP 1 showed net -5.6% at one year post-stopping and the Berg et al. (2025) meta-analysis found 9.69 kg mean regain, a weight maintenance plan before discontinuation is likely to be important for sustained sleep-apnea benefit (Wilding et al., 2022; Berg et al., 2025).
Should You Address Sleep Before or After Stopping Ozempic
The first step is to identify which type of sleep disruption you are experiencing — drug-driven, weight-dependent, or nutritional. Drug-driven disruption (possible GLP-1-related arousal, GI reflux) is expected to ease as the drug clears if drug exposure is the main driver. Nutritional disruption (lower tryptophan intake, blood sugar instability) can be addressed by normalizing eating patterns as appetite returns. Weight-dependent disruption may persist or worsen with weight regain and requires a different approach.
Drug-driven effects are expected to fade across the washout period if drug exposure is the main driver. Nutritional disruption from undereating responds to restoring adequate protein and carbohydrate as appetite returns. If sleep apnea worsens with weight regain, the response is weight management and airway assessment (Mifsud et al., 2025; Wang et al., 2020).
Related Reading
- Metabolic Sleep Disruption — The full metabolic sleep framework covering glucose regulation, insulin resistance, mitochondrial decline, NAD+ loss, and fat metabolism
- Does Ozempic Cause Insomnia What GLP-1 Drugs Do to Your Sleep — The broad overview of GLP-1 sleep side effects and mechanisms
- Why Does Ozempic Make You Tired But Keep You Awake — How GLP-1 drugs activate orexin/wakefulness circuitry while appetite suppression can create fatigue
- Why Does Ozempic Wake You Up at 3am — How lower overnight fuel availability and counter-regulatory hormones can contribute to 3am waking
- Does Ozempic Give You Vivid Dreams — How GLP-1 drugs may affect dream intensity through arousal and neuropsychiatric pathways
- Does Eating Too Little on Ozempic Cause Insomnia — How appetite suppression can affect tryptophan, serotonin, melatonin, and overnight glucose stability
- Does Your Ozempic Injection Day Disrupt Your Sleep The 24-48 Hour Pattern — Why sleep disruption can follow a weekly pattern tied to injection timing and peak drug exposure
- Does Your Semaglutide Dose Increase Restart Your Insomnia Why Each Titration Can Disrupt Sleep Again — Why each titration step can restart sleep disruption before adaptation catches up
- Can You Take Melatonin, Magnesium, or Sleep Aids While on Ozempic — How melatonin, magnesium, DORAs, and other sleep aids intersect with GLP-1 physiology
- Does Ozempic Give You Anxiety at Night Why GLP-1 Drugs May Contribute to Nighttime Panic — How orexin activation, HPA-axis signaling, and glucose instability may contribute to nighttime anxiety
- Do GLP-1 Drugs Disrupt Your Body Clock Ozempic, Circadian Rhythms, and Melatonin — How GLP-1 secretion rhythms, food timing, peripheral clocks, and melatonin intersect
- Ozempic Night Sweats: How GLP-1 Drugs May Affect Temperature at Night — How GLP-1 drugs may affect nighttime sweating through autonomic tone and thermoregulation
- Ozempic Nausea and Sleep: How Stomach Side Effects Keep You Awake at Night — How delayed gastric emptying, reflux, and nausea can fragment sleep
- Does Ozempic Muscle Loss Affect Your Sleep Sarcopenia, Temperature, and Deep Sleep — How lean-mass loss, thermoregulation, and deep sleep can interact during GLP-1 weight loss
- Could Ozempic Contribute to Restless Legs at Night Iron, B12, and the Dopamine Connection — How iron, B12, dopamine, and rapid weight loss may intersect with restless legs at night
- Does Compounded Semaglutide Affect Sleep Differently Than Brand-Name Ozempic — How dose variability, formulation differences, and adverse-event tracking can affect sleep side effects
- Ozempic and Sleep After 40: Why GLP-1 Side Effects Can Feel Different as You Age — Why GLP-1 sleep effects can land differently after 40 because of muscle, hormones, and glucose regulation
References
- Acuna-Goycolea, C., & van den Pol, A. (2004). Glucagon-like peptide 1 excites hypocretin/orexin neurons by direct and indirect mechanisms: Implications for viscera-mediated arousal. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience, 24(37), 8141-8152. https://pubmed.ncbi.nlm.nih.gov/15371515/
- Berg, S., Stickle, H., Rose, S. J., & Nemec, E. C. (2025). Discontinuing glucagon-like peptide-1 receptor agonists and body habitus: A systematic review and meta-analysis. Obesity Reviews: An Official Journal of the International Association for the Study of Obesity, 26(8), e13929. https://pubmed.ncbi.nlm.nih.gov/40186344/
- Bravo, R., Matito, S., Cubero, J., Paredes, S. D., Franco, L., Rivero, M., Rodriguez, A. B., & Barriga, C. (2013). Tryptophan-enriched cereal intake improves nocturnal sleep, melatonin, serotonin, and total antioxidant capacity levels and mood in elderly humans. Age, 35(4), 1277-1285. https://pubmed.ncbi.nlm.nih.gov/22622709/
- Chang, M. G., Ripoll, J. G., Lopez, E., Krishnan, K., & Bittner, E. A. (2024). A scoping review of GLP-1 receptor agonists: Are they associated with increased gastric contents, regurgitation, and aspiration events Journal of Clinical Medicine, 13(21), 6336. https://pubmed.ncbi.nlm.nih.gov/39518474/
- Jauch-Chara, K., Hallschmid, M., Gais, S., Oltmanns, K. M., Peters, A., Born, J., & Schultes, B. (2007). Awakening and counterregulatory response to hypoglycemia during early and late sleep. Diabetes, 56(7), 1938-1942. https://pubmed.ncbi.nlm.nih.gov/17400929/
- Kolli, R. T., Aoutla, S., Jyothi, N., Mohamed Kalifa, M. R. H., Raju, A., & Cheenikkal Muralidharan, K. (2025). Rebound or retention: A meta-analysis of weight regain after the discontinuation of glucagon-like peptide-1 (GLP-1) receptor agonists and other anti-obesity drugs. Cureus, 17(10), e94926. https://pubmed.ncbi.nlm.nih.gov/41116804/
- Li, M., Lin, H., Yang, Q., Zhang, X., Zhou, Q., Shi, J., & Ge, F. (2025). Glucagon-like peptide-1 receptor agonists for the treatment of obstructive sleep apnea: A meta-analysis. Sleep, 48(4), zsae280. https://pubmed.ncbi.nlm.nih.gov/39626095/
- Malhotra, A., Grunstein, R. R., Fietze, I., Weaver, T. E., Redline, S., Azarbarzin, A., Sands, S. A., Schwab, R. J., Dunn, J. P., Chakladar, S., Bunck, M. C., Bednarik, J., & SURMOUNT-OSA Investigators. (2024). Tirzepatide for the treatment of obstructive sleep apnea and obesity. The New England Journal of Medicine, 391(13), 1193-1205. https://pubmed.ncbi.nlm.nih.gov/38912654/
- Mifsud, C. S., Kolla, B. P., Rushlow, D. R., & Mansukhani, M. P. (2025). The impact of GLP-1 agonists on sleep disorders: Spotlight on sleep apnea. Expert Opinion on Pharmacotherapy, 26(14-15), 1529-1538. https://pubmed.ncbi.nlm.nih.gov/41114602/
- Quarenghi, M., Capelli, S., Galligani, G., Giana, A., Preatoni, G., & Turri Quarenghi, R. (2025). Weight regain after liraglutide, semaglutide or tirzepatide interruption: A narrative review of randomized studies. Journal of Clinical Medicine, 14(11), 3791. https://pubmed.ncbi.nlm.nih.gov/40507553/
- Reutrakul, S., Sumritsopak, R., Saetung, S., Chanprasertyothin, S., & Anothaisintawee, T. (2017). The relationship between sleep and glucagon-like peptide 1 in patients with abnormal glucose tolerance. Journal of Sleep Research, 26(6), 756-763. https://pubmed.ncbi.nlm.nih.gov/28560837/
- Wang, S. H., Keenan, B. T., Wiemken, A., Zang, Y., Staley, B., Sarwer, D. B., Torigian, D. A., Williams, N., Pack, A. I., & Schwab, R. J. (2020). Effect of weight loss on upper airway anatomy and the apnea-hypopnea index: The importance of tongue fat. American Journal of Respiratory and Critical Care Medicine, 201(6), 718-727. https://pubmed.ncbi.nlm.nih.gov/31918559/
- Wilding, J. P. H., Batterham, R. L., Davies, M., Van Gaal, L. F., Kandler, K., Konakli, K., Lingvay, I., McGowan, B. M., Oral, T. K., Rosenstock, J., Wadden, T. A., Wharton, S., Yokote, K., Kushner, R. F., & STEP 1 Study Group. (2022). Weight regain and cardiometabolic effects after withdrawal of semaglutide: The STEP 1 trial extension. Diabetes, Obesity & Metabolism, 24(8), 1553-1564. https://pubmed.ncbi.nlm.nih.gov/35441470/
- Yang, X. D., & Yang, Y. Y. (2024). Clinical pharmacokinetics of semaglutide: A systematic review. Drug Design, Development and Therapy, 18, 2555-2570. https://pubmed.ncbi.nlm.nih.gov/38952487/
Written by Kat Fu, M.S., M.S. – Last reviewed: May 2026 – 14 references cited
