NAD+ supplements — NR (nicotinamide riboside) and NMN (nicotinamide mononucleotide) — are marketed on the assumption that they enter cells and replenish NAD+ through the salvage pathway. Isotope-tracing data from 2020-2026 shows a different route. Both precursors are degraded to nicotinamide and then converted by gut bacteria into nicotinic acid before the majority of NAD+ is synthesized. The composition of someone’s gut microbiome affects how much NAD+ they produce from a given dose.
This article covers the bacterial conversion pathway, which bacterial enzymes are involved, what human trial data shows, and what disrupts or supports this conversion. It does not cover gut inflammation pathways or the gut-vagus-GABA pathway — those mechanisms are covered separately. For how NAD+ connects to circadian regulation and sleep, see the parent overview: Metabolic Sleep Disruption.
How Do Gut Bacteria Convert NAD+ Precursors Into Their Active Form?
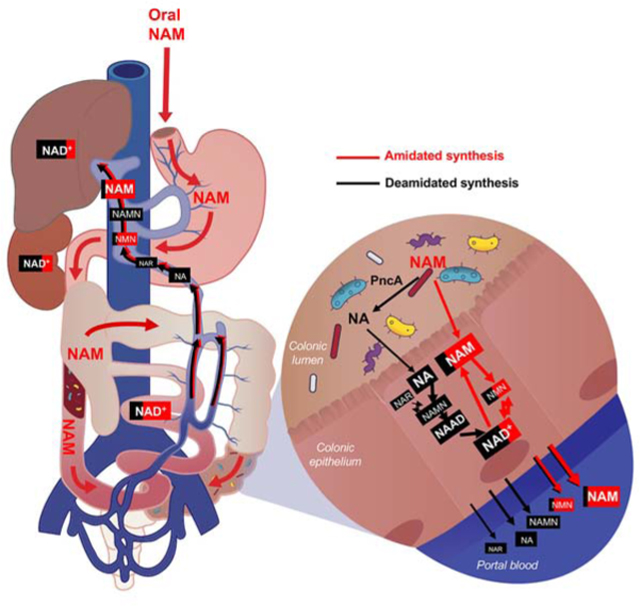
The bacterial enzyme at the center of this pathway is nicotinamidase, encoded by the gene pncA. This enzyme performs a deamidation reaction — it removes an amino group from nicotinamide (NAM) to produce nicotinic acid (NA). A 2020 study by Shats et al. identified pncA as the enzyme responsible for this conversion and demonstrated that gut bacteria carrying pncA are required for full NAD+ elevation from oral precursors (Shats et al., 2020).
Vertebrates lost their own nicotinamidase gene during evolution. Humans cannot perform this NAM-to-NA conversion without gut bacteria. This makes pncA-expressing bacteria the only identified route from nicotinamide to nicotinic acid in the human body. Without this bacterial step, the Preiss-Handler pathway — the enzymatic sequence that builds NAD+ from nicotinic acid in the liver — receives less substrate from this route.
A bioinformatic survey in the same study identified 2,088 bacterial pncA homologs (structurally related versions of the gene) across diverse bacterial phyla. Nicotinamidase activity is not limited to one bacterial species — it is distributed throughout the gut microbiome (Shats et al., 2020).
A 2022 isotope-tracing study by Chellappa et al. added a second piece: the nicotinamide that gut bacteria convert does not come from the supplement itself sitting in the lower gut. Dietary NR is rapidly cleaved to nicotinamide before entering circulation throughout the body and does not reach distal gut bacteria in its intact form. Instead, the host’s own circulating nicotinamide enters the gut lumen, where bacteria convert it to nicotinic acid. The bacteria-derived nicotinic acid is then reabsorbed by the host for NAD+ synthesis. Circulating nicotinic acid levels are maintained even in the absence of dietary nicotinic acid intake — gut bacteria serve as a contributing source (Chellappa et al., 2022).
This means the common assumption — that NR enters cells and becomes NAD+ through the NRK1/NMNAT enzymes (the salvage pathway) — describes only part of the picture. NR and NMN are absorbed in the upper GI tract, but they are degraded to nicotinamide before the majority of NAD+ is produced. The bacterial deamidation step is where the bulk of the conversion happens.
Does NR or NMN Reach Your Cells — Or Does It Go Through Bacteria First?
A 2025 study by Yaku et al. mapped the full recycling route. NR and NMN administered both orally and intravenously converge on the same gut-bacteria-dependent nicotinic acid intermediate. Even IV-administered NR and NMN — which bypass the gut on initial delivery — are degraded to nicotinamide, released into bile via enterohepatic circulation, and then converted by gut bacteria to nicotinic acid in the intestinal lumen. The liver uses this recycled nicotinic acid for NAD+ synthesis over other available substrates (Yaku et al., 2025).
This finding changes the NR vs. NMN comparison. NR requires two enzymatic steps to become NAD+ through the salvage pathway (NRK1 then NMNAT). NMN requires one step (NMNAT). This biochemical difference is often cited in marketing. But both precursors funnel through the same bacterial conversion bottleneck for the majority of the NAD+ they produce. The shared dependence on gut microbial deamidation matters more than the difference in salvage pathway steps.
For dosing, this has a concrete implication: if the gut microbiome is the rate-limiting factor, taking a higher dose may not compensate for impaired bacterial conversion capacity. Antibiotic use or microbiome disruption may reduce supplementation outcomes regardless of which precursor is used or how much is taken (Yaku et al., 2025). The Chellappa et al. (2022) data support this — even when NR was absorbed in the upper gut, the majority of downstream NAD+ production depended on bacteria converting the host’s circulating nicotinamide in the lower gut.

What Does the Human Trial Evidence Show?
The first head-to-head human comparison of NR, NMN, and nicotinamide came from Christen et al. (2026) in Nature Metabolism. In a randomized, open-label, placebo-controlled trial, 65 healthy participants supplemented for 14 days. Both NR and NMN comparably increased circulatory NAD+ concentrations. Nicotinamide did not produce a sustained chronic increase — it produced only acute, transient effects through the salvage pathway within a 4-hour window after a single dose.
The ex vivo component of the trial demonstrated the bacterial mechanism. Fecal microbiota from healthy adult donors were incubated with NR and NMN. Both compounds enhanced microbial growth and gave rise to nicotinic acid. When tested in whole blood culture assays, nicotinic acid raised NAD+ — while NR, NMN, and nicotinamide alone did not (Christen et al., 2026).
Christen et al. also identified Enterocloster aldensis as a gut bacterial species that metabolizes NR. NR and NMN supplementation increased short-chain fatty acid (SCFA) concentrations during ex vivo fermentation, pointing toward secondary benefits for gut barrier integrity beyond NAD+ production (Christen et al., 2026).
A 2024 review by Benjamin and Crews synthesized human trial evidence on NMN and identified gut microbiota composition as a contributing factor in inter-individual differences in NMN bioavailability. Individual differences in gut microbiome composition may contribute to variable NAD+ outcomes from the same dose. The review noted significant knowledge gaps regarding the interplay between gut microbiota and NAD+ precursor metabolism, suggesting further research is needed (Benjamin & Crews, 2024).
Retrospective evidence also fits the pattern. In 2016, Trammell et al. published the first human NR dose-response data, showing dose-dependent increases in blood NAD+ metabolites across 12 healthy subjects receiving single doses of 100, 300, and 1,000 mg. A preceding pilot study in one individual had observed up to 2.7-fold NAD+ elevation. Blood NAAD — nicotinic acid adenine dinucleotide, a deamidated NAD+ metabolite produced through the Preiss-Handler pathway — emerged as an early biomarker of NAD+ repletion. At the time, the appearance of this deamidated metabolite after NR ingestion was unexplained. In retrospect, it was indirect evidence that gut bacteria were deamidating the precursor before the mechanism was understood (Trammell et al., 2016).
What Disrupts the Gut Bacteria That Process NAD+ Precursors?
The Shats et al. (2020) data on germ-free and antibiotic-exposed mice provide the evidence: mice lacking gut microbiota showed reduced NAD+ responses to orally administered precursors compared to conventionally colonized controls. Without pncA-expressing bacteria, the deamidation step does not occur, and the Preiss-Handler pathway receives less substrate.
Broad-spectrum antibiotic courses reduce microbial diversity and may reduce populations of pncA-expressing bacteria. No human trial has measured NAD+ precursor efficacy before and after antibiotics, but the animal data predicts reduced conversion capacity during and after antibiotic courses.
The relationship between NAD+ precursors and gut bacteria runs in both directions. A 2025 double-blind, randomized, placebo-controlled trial by Schreiber et al. enrolled 900 participants with verified COVID-19 and supplemented them with 1,000 mg nicotinamide daily for 4 weeks. Among participants at risk for severe disease progression, 57.6% of the nicotinamide group recovered from performance decline by week 2 versus 42.6% in the placebo group (P = 0.004). Return to normal activities improved in the nicotinamide arm (P = 0.009). At 6-month follow-up, acute-phase nicotinamide responders reported fewer post-COVID effects than placebo responders (P = 0.010). Gut metagenomic sequencing revealed nicotinamide-associated changes in microbial metabolic signatures that paralleled the recovery improvements (Schreiber et al., 2025).
This means the supplement also reshapes the microbiome. NAD+ precursor supplementation is not a one-directional input — the bacteria metabolize the precursor, and the precursor changes the bacterial community.
Additional data from Christen et al. (2026) reinforces this bidirectional relationship: NR and NMN increased short-chain fatty acid concentrations during ex vivo fecal fermentation. SCFAs — including butyrate, propionate, and acetate — support intestinal epithelial cell integrity and mucosal barrier function. The bacterial metabolism of NAD+ precursors may produce secondary benefits for gut barrier health beyond NAD+ elevation.
Microbiome composition is modifiable through diet, fiber intake, antibiotic stewardship, and fermented food consumption. No trial has tested whether targeted probiotics improve NAD+ precursor conversion rates in humans, though the identification of specific pncA-expressing species and Enterocloster aldensis as an NR-metabolizing species provides targets for future research.
NAD+ precursor metabolism is one piece of the metabolic picture. In adults over 40, blood sugar instability, cortisol rhythm changes, hormonal changes, and inflammatory load may be compounding alongside declining NAD+ — and each of these responds to different approaches.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Does NAD+ Help With Sleep?
SIRT1 is a NAD+-dependent deacetylase that modulates circadian clock gene expression. When NAD+ levels decline — as they do with aging — SIRT1 activity decreases, and circadian gene oscillation can weaken. Restoring NAD+ through precursor supplementation is one route to supporting SIRT1 function and, by extension, circadian regulation.
The trial evidence is mixed. NMN at 250 mg/day improved sleep quality in older adults. NR at higher doses has not demonstrated sleep benefits in placebo-controlled analysis. Differences in gut microbiome composition between study populations may contribute to these variable outcomes — if bacterial conversion capacity differs between individuals, the same precursor dose produces different NAD+ responses (Christen et al., 2026).
For a full comparison of NMN and NR sleep trial data, see: Which Nicotinamide Adenine Dinucleotide Precursor Improves Sleep — Nicotinamide Mononucleotide or Nicotinamide Riboside?
Does NAD+ Supplementation Improve Sleep Quality?
The NMN sleep data comes from Kim et al. (2022) and Morifuji et al. (2024), who found improvements in PSQI scores and reduced daytime drowsiness in older adult populations. The NR data from a placebo-controlled trial at 2,000 mg/day did not reach statistical significance for sleep outcomes (p = 0.69) in a long-COVID population.
One explanation for inter-individual variability: gut microbiota composition differs between people, and people with greater populations of pncA-expressing bacteria may convert more of their NMN or NR dose into nicotinic acid, producing higher NAD+ elevation and stronger downstream effects on circadian regulation (Benjamin & Crews, 2024).
For the full trial-by-trial comparison: Which Nicotinamide Adenine Dinucleotide Precursor Improves Sleep — Nicotinamide Mononucleotide or Nicotinamide Riboside?
Do Probiotics Help NAD+ Levels?
The Christen et al. (2026) trial identified Enterocloster aldensis as a species that metabolizes NR in the gut — a potential target for future probiotic research. The Shats et al. (2020) survey of 2,088 pncA homologs shows the enzymatic capacity is distributed across many bacterial taxa, not concentrated in one species.
The probiotic-NAD+ hypothesis has plausible mechanistic support: if gut bacteria perform the rate-limiting conversion step, then increasing the population of bacteria with that enzymatic capacity could improve conversion rates. No trial has tested this.
This is a different mechanism from the GABA-producing probiotic literature. Probiotics that produce GABA (such as Lactobacillus rhamnosus and Bifidobacterium longum) affect sleep through vagal nerve transmission and central GABA receptor modulation — a separate pathway from NAD+ precursor conversion. For the GABA-probiotic evidence, see: How Your Gut Talks to Your Brain Through the Vagus Nerve — and Why It Matters for Sleep.
Does Gut Health Affect Supplement Absorption?
The gut microbiome affects absorption and metabolism of many supplements and medications. For NAD+ precursors, the bacterial role is enzymatic conversion, not passive absorption. This distinction matters: taking a higher dose does not overcome a microbiome-dependent enzymatic bottleneck the way it might overcome a passive absorption limitation.
NR and NMN are absorbed in the upper gastrointestinal tract. The bacterial conversion happens downstream — circulating nicotinamide enters the gut lumen, bacteria deamidate it to nicotinic acid, and the nicotinic acid is reabsorbed for NAD+ synthesis in the liver. The conversion step depends on the presence of pncA-expressing bacteria. Reduced microbial diversity — from antibiotics, poor dietary fiber intake, or other disruptions — may reduce the bacterial population capable of performing this conversion (Yaku et al., 2025).
Related Reading
- Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep — the pillar article for metabolic sleep disruption, covering glucose regulation, cortisol rhythm, mitochondrial stress, NAD+ loss, and fat metabolism
- Which Nicotinamide Adenine Dinucleotide Precursor Improves Sleep — Nicotinamide Mononucleotide or Nicotinamide Riboside? — NMN and NR sleep evidence, timing, safety, and human trial differences
- Does NMN Cause Insomnia? What Timing and Dose Matter — NMN timing, dose, circadian effects, and sleep continuity data
- Does Apigenin Protect Your NAD+ and Improve Sleep Through CD38 Inhibition? — CD38 inhibition, NAD+ depletion with age, and apigenin sleep evidence
- Does Your Body Divert Tryptophan From Melatonin to Make NAD+? — tryptophan allocation between melatonin, kynurenine, inflammation, and NAD+ synthesis
- Why Does Sleep Repair Your DNA — And Burn Through Your NAD+ to Do It? — PARP1, sleep pressure, DNA repair, and NAD+ demand during sleep
- Does NAD+ IV Therapy Improve Sleep? What the Evidence Shows — IV NAD+ evidence, safety, infusion effects, and comparison with oral precursors
- Why Does NAD+ Drop Faster in Women After 40 — And What Does That Mean for Sleep? — menopause, ovarian aging, NAD+ decline, and sleep disruption after 40
References
Shats, I., Williams, J. G., Liu, J., Makarov, M. V., Wu, X., Lih, F. B., Deterding, L. J., Lim, C., Xu, X., Randall, T. A., Lee, E., Li, W., Fan, W., Li, J. L., Sokolsky, M., Kabanov, A. V., Li, L., Migaud, M. E., Locasale, J. W., & Li, X. (2020). Bacteria boost mammalian host NAD metabolism by engaging the deamidated biosynthesis pathway. Cell Metabolism, 31(3), 564-579.e7. https://pubmed.ncbi.nlm.nih.gov/32130883/
Chellappa, K., McReynolds, M. R., Lu, W., Zeng, X., Makarov, M., Hayat, F., Mukherjee, S., Bhat, Y. R., Lingala, S. R., Shima, R. T., Descamps, H. C., Cox, T., Ji, L., Jankowski, C., Chu, Q., Davidson, S. M., Thaiss, C. A., Migaud, M. E., Rabinowitz, J. D., & Baur, J. A. (2022). NAD precursors cycle between host tissues and the gut microbiome. Cell Metabolism, 34(12), 1947-1959.e5. https://pubmed.ncbi.nlm.nih.gov/36476934/
Yaku, K., Palikhe, S., Iqbal, T., Hayat, F., Watanabe, Y., Fujisaka, S., Izumi, H., Yoshida, T., Karim, M., Uchida, H., Nawaz, A., Tobe, K., Mori, H., Migaud, M. E., & Nakagawa, T. (2025). Nicotinamide riboside and nicotinamide mononucleotide facilitate NAD+ synthesis via enterohepatic circulation. Science Advances, 11(12), eadr1538. https://pubmed.ncbi.nlm.nih.gov/40117359/
Christen, S., Redeuil, K., Goulet, L., Giner, M. P., Breton, I., Rota, R., Frezal, A., Nazari, A., Van den Abbeele, P., Godin, J. P., Nutten, S., & Cuenoud, B. (2026). The differential impact of three different NAD+ boosters on circulatory NAD and microbial metabolism in humans. Nature Metabolism, 8(1), 62-73. https://pubmed.ncbi.nlm.nih.gov/41540253/
Trammell, S. A. J., Schmidt, M. S., Weidemann, B. J., Redpath, P., Jaksch, F., Dellinger, R. W., Li, Z., Abel, E. D., Migaud, M. E., & Brenner, C. (2016). Nicotinamide riboside is uniquely and orally bioavailable in mice and humans. Nature Communications, 7, 12948. https://pubmed.ncbi.nlm.nih.gov/27721479/
Benjamin, C., & Crews, R. (2024). Nicotinamide mononucleotide supplementation: Understanding metabolic variability and clinical implications. Metabolites, 14(6), 341. https://pubmed.ncbi.nlm.nih.gov/38921475/
Schreiber, S., Waetzig, G. H., Lopez-Agudelo, V. A., Geisler, C., Schlicht, K., Franzenburg, S., di Giuseppe, R., Pape, D., Bahmer, T., Krawczak, M., Kokott, E., Penninger, J. M., Harzer, O., Kramer, J., von Schrenck, T., Sommer, F., Zacharias, H. U., Millet Pascual-Leone, B., Forslund, S. K., Heyckendorf, J., Aden, K., Hollweck, R., Laudes, M., & Rosenstiel, P. (2025). Nicotinamide modulates gut microbial metabolic potential and accelerates recovery in mild-to-moderate COVID-19. Nature Metabolism, 7(6), 1136-1149. https://pubmed.ncbi.nlm.nih.gov/40355744/
Kim, M., Seol, J., Sato, T., Fukamizu, Y., Sakurai, T., & Okura, T. (2022). Effect of 12-week intake of nicotinamide mononucleotide on sleep quality, fatigue, and physical performance in older Japanese adults: A randomized, double-blind placebo-controlled study. Nutrients, 14(4), 755. https://pubmed.ncbi.nlm.nih.gov/35215405/
Morifuji, M., Higashi, S., Ebihara, S., & Nagata, M. (2024). Ingestion of beta-nicotinamide mononucleotide increased blood NAD levels, maintained walking speed, and improved sleep quality in older adults in a double-blind randomized, placebo-controlled study. GeroScience, 46(5), 4671-4688. https://pubmed.ncbi.nlm.nih.gov/38789831/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 9 references cited
