NAD+ IV therapy is a $250-1,000/session wellness service marketed for energy, sleep, and anti-aging. Clinics position it as a direct route to raising cellular NAD+ levels. This article is for people considering IV NAD+ who want to know what the published evidence supports for sleep outcomes — separate from marketing claims.
This article covers the human evidence for IV NAD+ and sleep, pharmacokinetic data on how IV NAD+ is metabolized, safety and tolerability, pricing, and what oral NAD+ precursors (NMN, NR) have demonstrated by comparison. It does not cover addiction/withdrawal applications or anti-aging longevity claims.
NAD+ depletion is one of several metabolic contributors to sleep disruption. For the full overview of how metabolic contributors drive 3am wakeups, see Metabolic Sleep Disruption.
What Does the Research Show for IV NAD+ and Sleep?
The Gindri 2024 review searched six databases and identified 10 randomized trials encompassing 489 participants across chronic fatigue, Parkinson’s, and Alzheimer’s populations. Oral NADH supplementation was associated with reduced fatigue and decreased anxiety. Sleep quality was evaluated as an endpoint in the CFS group, but improvement was not specifically concluded by the review. Every trial in the review used oral delivery, not IV. The review concluded that evidence is insufficient for firm recommendations due to heterogeneity in dosing, delivery route, and outcome measures (Gindri et al., 2024).
The Reyna 2026 retrospective pilot is the closest study to have collected IV NAD+ sleep-related data, though sleep outcomes were not reported in the published findings. This study compared commercially administered IV NAD+ (500 mg/day for 4 consecutive days) versus IV NR (500 mg/day for 4 days) in a wellness clinic. Sleep-related impairment was measured using validated PROMIS instruments at baseline and 30-day follow-up. The limitations matter: the study was retrospective, had no placebo arm, was not randomized, had a small sample, and was conducted at a single clinic (Reyna et al., 2026).
Two 2025 reviews — Zhang et al. in Nature Aging and Vinten et al. in Nature Metabolism — both call for large-scale studies with standardized dosing and administration routes. Neither review identifies any completed IV NAD+ sleep trial. Zhang et al. emphasize the need for further large-scale studies to determine optimal dose, administration routes, and frequency (Zhang et al., 2025).
The bottom line for consumers: IV NAD+ clinics are offering a service that has not been tested in a controlled trial for the outcome — sleep improvement — that many clients seek.
What Happens to NAD+ During an IV Infusion?
The Grant 2019 pilot study (n=8, 3 controls) infused NAD+ intravenously at 3 micromol/min for 6 hours while monitoring plasma and urine NAD+ metabolomes continuously. The results raise questions about the efficiency of IV NAD+ delivery.
No measurable change in plasma NAD+ or its metabolites appeared until after 2 hours of continuous infusion. The metabolite profile at 2 hours was consistent with enzymatic catabolism — NAD+ glycohydrolase and NAD+ pyrophosphatase were breaking down the infused NAD+ before it could circulate as an intact molecule. By 6 hours, increased urinary excretion of methylnicotinamide indicated that breakdown products were being processed through the salvage pathway (Grant et al., 2019).
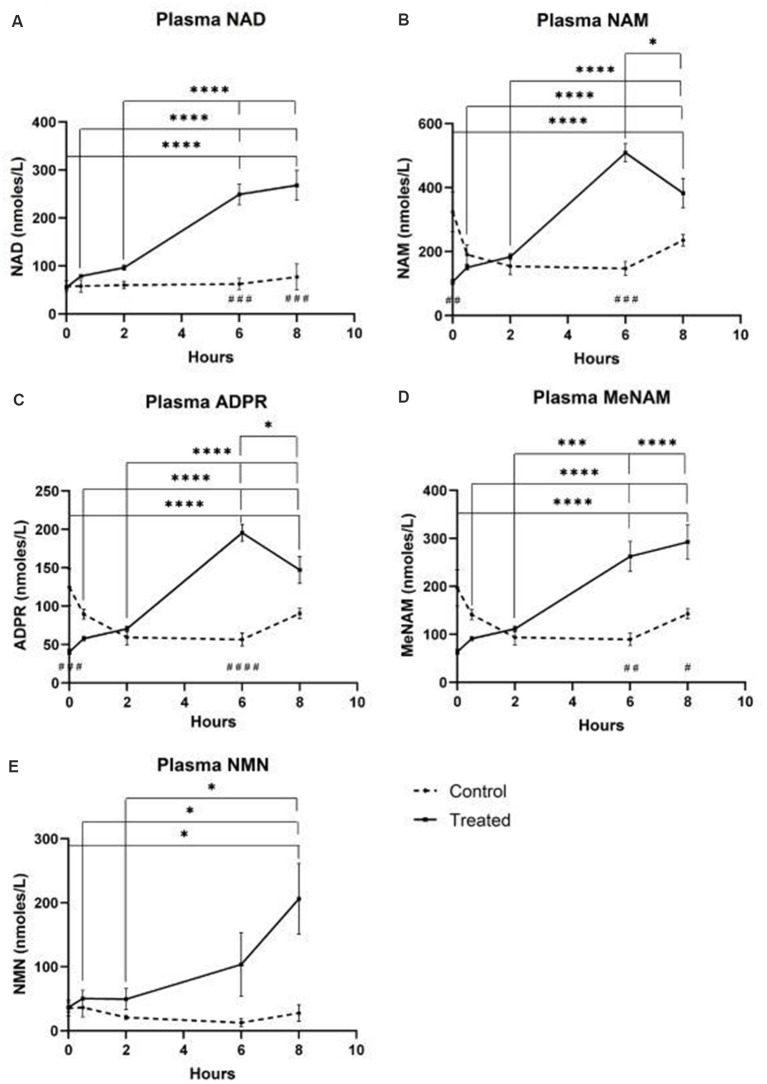
What this means for sleep-related claims: if IV NAD+ is largely catabolized before reaching peripheral tissues, the delivery route may not provide the direct cellular NAD+ elevation that marketing materials describe. The 2025 Nature Aging review identifies administration route as a variable requiring further study to determine its impact on pharmacokinetics and efficacy (Zhang et al., 2025).
Vinten et al. (2025) in Nature Metabolism add a broader caution: direct evidence for age-related NAD+ reduction in human tissues has been "consistently observed only in a limited number of studies." Tissue-specific NAD+ measurement remains sparse in humans, which means the premise that supplementation is necessary across aging populations rests on limited tissue-level data (Vinten et al., 2025).
Whether the metabolites produced from IV NAD+ catabolism — nicotinamide, methylnicotinamide — re-enter the salvage pathway effectively enough to raise NAD+ in sleep-relevant brain tissue has not been studied in humans.
Is NAD+ IV Therapy Safe?
The Reyna 2026 pilot provides the first head-to-head tolerability comparison of IV NAD+ versus IV NR. The IV NAD+ group experienced gastrointestinal distress, elevated heart rate, and chest pressure — effects managed by slowing the infusion rate, extending the average session to 96 minutes. The IV NR group reported only minor tingling and mild cramping, with an average infusion time of 37 minutes (Reyna et al., 2026).
Standard safety biomarkers — ALT, AST, hsCRP, BUN/creatinine, TSH — showed no changes in either group at 30 days. Two findings in the NAD+ group merit mention: alkaline phosphatase decreased (within-group p=0.04; between-group p<0.01) and HDL cholesterol decreased (p=0.02). The practical relevance of these changes from a single small pilot is unclear.
The Gindri 2024 review, covering 10 oral NADH RCTs, found a low incidence of adverse effects with oral delivery — transient muscle pain, headache, nervous disorders, no serious events. However, oral and IV delivery carry different safety profiles. IV delivery introduces infusion-related considerations and the moderate-to-severe tolerability effects observed in the Reyna pilot (Gindri et al., 2024).
The FDA does not recognize NAD+ infusions as approved for fatigue, aging, or sleep. IV NAD+ clinics function as wellness services outside of FDA-regulated indications.
Zhang et al. (2025) identify individual response variability as an unresolved challenge across all NAD+ supplementation routes. Long-term safety data for repeated IV NAD+ infusions does not exist.
How Does IV NAD+ Compare to Oral Supplements for Sleep?
Oral NMN: Kim 2022 (n=108, 250 mg/day, 12 weeks) and Morifuji 2024 (n=60, 250 mg/day, 12 weeks) both evaluated sleep-related endpoints. In Kim 2022, afternoon dosing produced the largest effect sizes but no statistically significant group-by-time interaction was observed (p=0.87). In Morifuji 2024, the NMN group showed reduced daytime dysfunction scores on the PSQI and measurable increases in blood NAD+ metabolites (global PSQI p=0.013, daytime dysfunction p=0.010) (Morifuji et al., 2024). For a full comparison of NMN and NR oral evidence, see Which Nicotinamide Adenine Dinucleotide Precursor Improves Sleep — Nicotinamide Mononucleotide or Nicotinamide Riboside?.
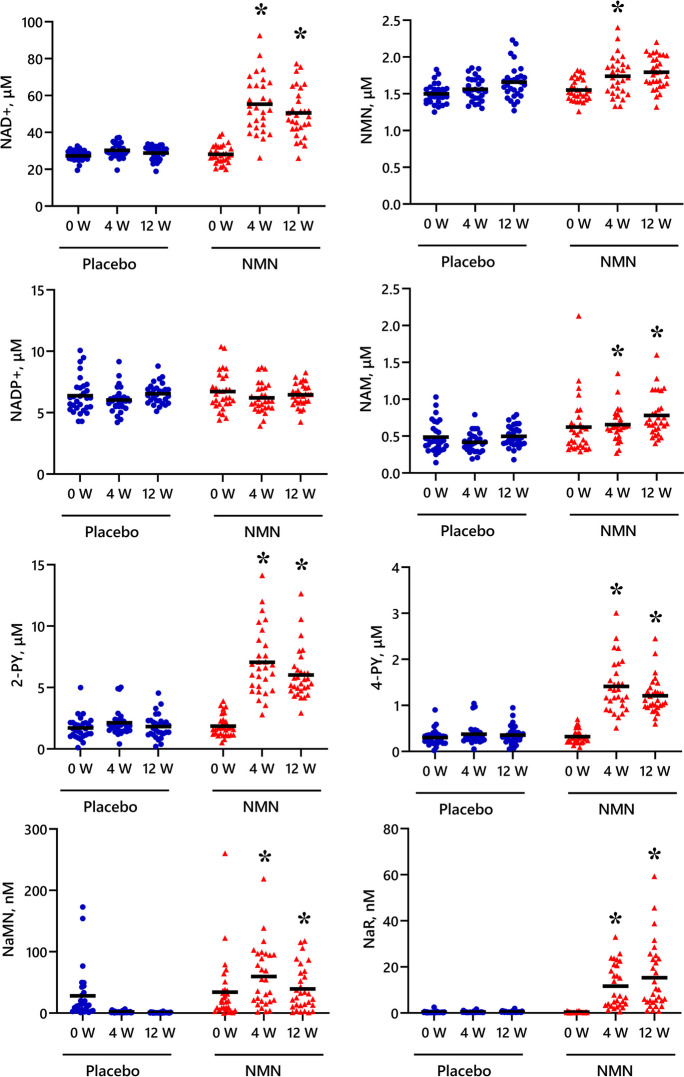
Oral NR: NR combined with pterostilbene raised blood NAD+ by 40-90% in the Dellinger 2017 trial (NRPT combination, not NR alone), but the only controlled trial reporting a sleep-specific p-value — Wu 2025, a long-COVID trial, reported p=0.69 for the primary between-group sleep comparison, though post-hoc analysis found p=0.03 after 10 weeks of NR. Whether raising blood NAD+ levels is sufficient on its own for sleep improvement remains unclear.
IV NAD+: Zero controlled sleep trials. The Reyna 2026 pilot collected sleep data but did not report sleep outcomes, and was retrospective and uncontrolled. Tolerability is lower than oral delivery — 96-minute infusions with gastrointestinal distress versus oral supplements taken at home.
Cost comparison: Oral NMN at 250 mg/day costs approximately $40-80/month. IV NAD+ sessions range from $250-1,000 per infusion, with many clinics recommending a series of 4-10 sessions for initial loading. A standard 4-session series costs $1,000-4,000 — equivalent to roughly 12-100 months of oral NMN supply. Neither option is covered by insurance.
Preclinical mechanism context: Chen 2024 showed NAD+ supplementation reduced neuroinflammation caused by chronic sleep restriction in animal and cell models. NAD+ inhibited reactive oxygen species production and promoted conversion of microglia from pro-inflammatory (M1) to anti-inflammatory (M2) states, via the cGAS-STING/NF-kB pathway. This provides a plausible mechanism linking NAD+ repletion to sleep-related neuroprotection, but it does not distinguish between delivery routes (Chen et al., 2024).
NAD+ depletion is one of several metabolic contributors that might be disrupting sleep — but it is rarely the only one. Blood sugar instability, cortisol rhythm changes, hormonal changes, and inflammatory load might each be compounding with the metabolic pathway, and identifying which causes might be active is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Can NAD+ IV Therapy Help with Insomnia?
The Gao 2023 multicenter RCT is the first trial positioning an NAD+ precursor for chronic insomnia — 400 participants, oral NMN at 320 mg/day for 60 days, with PSQI (Pittsburgh Sleep Quality Index) as the primary endpoint. No IV NAD+ insomnia trial exists at any stage (Gao et al., 2023).
Anecdotal reports diverge. Some IV NAD+ recipients describe improved sleep continuity and circadian regularity. Others report difficulty sleeping the night of infusion. This divergence may relate to individual differences in NAD+ catabolism rates and timing of infusion relative to sleep onset — but no study has investigated these variables.
How Much Is NAD+ IV Therapy?
The pricing structure breaks down as follows: per-session fees range from $250-1,000 depending on the NAD+ dose (typically 250-750 mg), clinic pricing, and geographic market. Many clinics recommend an initial loading series of 4-10 consecutive or closely spaced sessions, followed by monthly or quarterly maintenance infusions.
For comparison: oral NMN at the dose used in the Morifuji 2024 sleep RCT (250 mg/day) costs approximately $40-80/month. Oral NR costs approximately $30-60/month. A standard 4-session IV NAD+ series at mid-range pricing ($500/session = $2,000 total) buys 25-50 months of oral NMN at the RCT dose.
Price alone does not determine value. But when the higher-priced option has zero controlled sleep evidence and the lower-priced option has one positive RCT and one trial with suggestive effect sizes, the price comparison becomes relevant to decision-making (Zhang et al., 2025).
What Are the Side Effects of NAD+ IV Therapy?
The Reyna 2026 pilot provides the clearest tolerability comparison. IV NAD+ required an average of 96 minutes per infusion to manage side effects, compared to 37 minutes for IV NR — the NR group experienced only minor tingling and mild cramping.
In the IV NAD+ group, HDL cholesterol decreased (p=0.02) and alkaline phosphatase decreased (within-group p=0.04; between-group p<0.01). Whether these changes are meaningful or artifacts of a small, short-duration pilot is unknown (Reyna et al., 2026).
Long-term safety data for repeated IV NAD+ infusions — the kind of ongoing schedule many clinics recommend — does not exist.
Does Raising NAD+ Levels Improve Sleep?
The disconnect between NAD+ elevation and sleep outcomes suggests that the route NAD+ reaches sleep-relevant tissue may matter, or that other bottlenecks exist beyond raw NAD+ availability. SIRT1 expression levels, clock gene responsiveness to NAD+-mediated activation, and tissue-specific conversion rates may all mediate the gap between blood NAD+ concentrations and circadian function (Vinten et al., 2025).
Oral NMN raised blood NAD+ metabolites in both Kim 2022 and Morifuji 2024; Morifuji 2024 showed statistically significant sleep improvement, while Kim 2022 showed the largest effect sizes with PM dosing but no significant interaction. Oral NR raised blood NAD+ by 40-90% in combination with pterostilbene (Dellinger 2017) but did not improve sleep in the primary analysis of a long-COVID trial (Wu 2025). IV NAD+ raises blood NAD+ (after a 2-hour delay in the Grant 2019 pilot) but has never been tested for sleep in a controlled design. The relationship between raising blood NAD+ and improving sleep is more nuanced than marketing materials suggest — delivery route, timing, and individual metabolic context all appear to matter (Morifuji et al., 2024).
Related Reading
- Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep — the pillar article for metabolic sleep disruption, covering glucose regulation, cortisol rhythm, mitochondrial stress, NAD+ loss, and fat metabolism
- Which Nicotinamide Adenine Dinucleotide Precursor Improves Sleep — Nicotinamide Mononucleotide or Nicotinamide Riboside? — NMN and NR sleep evidence, timing, safety, and human trial differences
- Does NMN Cause Insomnia? What Timing and Dose Matter — NMN timing, dose, circadian effects, and sleep continuity data
- Does Apigenin Protect Your NAD+ and Improve Sleep Through CD38 Inhibition? — CD38 inhibition, NAD+ depletion with age, and apigenin sleep evidence
- Does Your Body Divert Tryptophan From Melatonin to Make NAD+? — tryptophan allocation between melatonin, kynurenine, inflammation, and NAD+ synthesis
- Why Does Sleep Repair Your DNA — And Burn Through Your NAD+ to Do It? — PARP1, sleep pressure, DNA repair, and NAD+ demand during sleep
- Does Your Gut Decide Whether Your NAD+ Supplement Works? — gut microbiome conversion of NAD+ precursors and why supplement response varies
- Why Does NAD+ Drop Faster in Women After 40 — And What Does That Mean for Sleep? — menopause, ovarian aging, NAD+ decline, and sleep disruption after 40
References
Chen, Q., Xiao, J., Lin, Z., Xu, X., & Chen, J. (2024). NAD+ supplement relieved chronic sleep restriction (CSR)-induced microglial proinflammation in vivo and in vitro. Journal of Neuroimmunology, 397, 578469. https://pubmed.ncbi.nlm.nih.gov/39520937/
Dellinger, R. W., Santos, S. R., Morris, M., Evans, M., Alminana, D., Guarente, L., & Marcotulli, E. (2017). Repeat dose NRPT (nicotinamide riboside and pterostilbene) increases NAD+ levels in humans safely and sustainably: a randomized, double-blind, placebo-controlled study. NPJ Aging and Mechanisms of Disease, 3, 17. https://pubmed.ncbi.nlm.nih.gov/29184669/
Gao, X., Li, J., Xu, S., Li, X., Wang, X., Li, Y., Huang, Y., Liu, S., & Zeng, Q. (2023). Oral nicotinamide mononucleotide (NMN) to treat chronic insomnia: protocol for the multicenter, randomized, double-blinded, placebo-controlled trial. Trials, 24(1), 340. https://pubmed.ncbi.nlm.nih.gov/37202819/
Gindri, I. M., Ferrari, G., Pinto, L. P. S., Bicca, J., Dos Santos, I. K., Dallacosta, D., & Roesler, C. R. M. (2024). Evaluation of safety and effectiveness of NAD in different clinical conditions: a systematic review. American Journal of Physiology. Endocrinology and Metabolism, 326(4), E417-E427. https://pubmed.ncbi.nlm.nih.gov/37971292/
Grant, R., Berg, J., Mestayer, R., Braidy, N., Bennett, J., Broom, S., & Watson, J. (2019). A pilot study investigating changes in the human plasma and urine NAD+ metabolome during a 6 hour intravenous infusion of NAD. Frontiers in Aging Neuroscience, 11, 257. https://pubmed.ncbi.nlm.nih.gov/31572171/
Kim, M., Seol, J., Sato, T., Fukamizu, Y., Sakurai, T., & Okura, T. (2022). Effect of 12-week intake of nicotinamide mononucleotide on sleep quality, fatigue, and physical performance in older Japanese adults: a randomized, double-blind placebo-controlled study. Nutrients, 14(4), 755. https://pubmed.ncbi.nlm.nih.gov/35215405/
Morifuji, M., Higashi, S., Ebihara, S., & Nagata, M. (2024). Ingestion of beta-nicotinamide mononucleotide increased blood NAD levels, maintained walking speed, and improved sleep quality in older adults in a double-blind randomized, placebo-controlled study. GeroScience, 46(5), 4671-4688. https://pubmed.ncbi.nlm.nih.gov/38789831/
Reyna, K., Heinzen, G., Patel, N., Ritter, M., Siojo, A., Legere, H., & Pojednic, R. (2026). Intravenous infusion of nicotinamide adenine dinucleotide (NAD+) versus nicotinamide riboside (NR): a retrospective tolerability pilot study in a real-world setting. Frontiers in Aging, 7, 1652582. https://pubmed.ncbi.nlm.nih.gov/41704678/
Vinten, K. T., Tretowicz, M. M., Coskun, E., van Weeghel, M., Canto, C., Zapata-Perez, R., Janssens, G. E., & Houtkooper, R. H. (2025). NAD+ precursor supplementation in human ageing: clinical evidence and challenges. Nature Metabolism, 7(10), 1974-1990. https://pubmed.ncbi.nlm.nih.gov/41083806/
Wu, C.-Y., Reynolds, W. C., Abril, I., McManus, A. J., Brenner, C., Gonzalez-Irizarry, G., Gutierrez-Martinez, L., Sun, O., Rosand, J., Tanzi, R. E., Arnold, S. E., & Guzman-Velez, E. (2025). Effects of nicotinamide riboside on NAD+ levels, cognition, and symptom recovery in long-COVID: a randomized controlled trial. EClinicalMedicine, 89, 103633. https://pubmed.ncbi.nlm.nih.gov/41357333/
Zhang, J., Wang, H. L., Lautrup, S., et al. (2025). Emerging strategies, applications and challenges of targeting NAD+ in the clinic. Nature Aging, 5(9), 1704-1731. https://pubmed.ncbi.nlm.nih.gov/40926126/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 11 references cited
