NAD+ levels decline with age, and CD38 is a primary driver of that decline. Separately, apigenin has gained widespread attention as a sleep supplement, largely driven by Andrew Huberman’s recommendations. What connects these two stories is the same molecule: apigenin acts on both pathways at once.
This article covers what CD38 does to NAD+ as you age, how apigenin inhibits it, the separate GABA-A mechanism through which apigenin promotes sleep, and what the human evidence shows for dosing and efficacy.
NAD+ depletion and sleep disruption are two threads within a broader metabolic picture. The full overview of how metabolic impairment disrupts sleep is in the parent article: Metabolic Sleep Disruption.
What Is CD38 and Why Does It Drain Your NAD+ With Age?
A 2016 study published in Cell Metabolism identified CD38 as a major driver of age-related NAD+ depletion across mammalian tissues. The researchers measured CD38 expression in young (3 month) versus old (32 month) mice and found it increased in liver, adipose tissue, spleen, and skeletal muscle during normal aging (Camacho-Pereira et al., 2016).
The evidence for causality is direct: CD38 knockout mice — animals genetically engineered to lack the CD38 enzyme — maintained higher NAD+ levels throughout their lifespan and displayed preserved mitochondrial function compared to wild-type littermates.
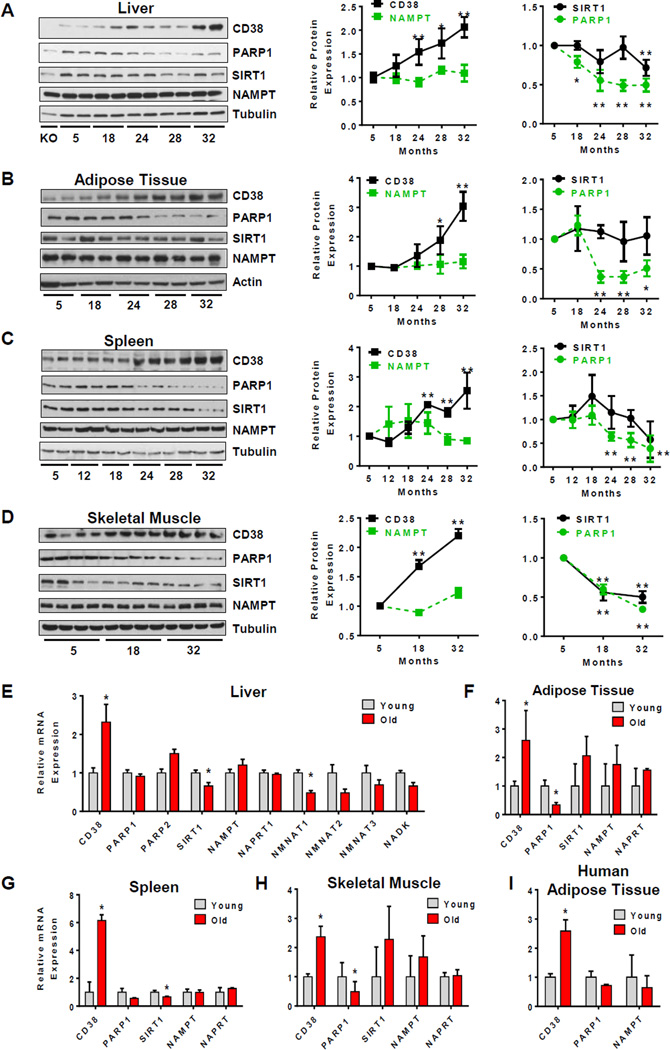
SIRT3, a mitochondrial sirtuin (NAD+-dependent deacetylase enzyme), is central to this mechanism. When CD38 depletes NAD+ below the threshold needed for SIRT3 activity, mitochondrial protein acetylation increases and oxidative phosphorylation — the process by which mitochondria produce ATP — becomes less efficient. CD38 knockout mice showed improved glucose tolerance and mitochondrial respiration even in old age, linking NAD+ preservation directly to metabolic healthspan.
One finding with specific implications for supplementation: CD38 degrades nicotinamide mononucleotide (NMN), a key NAD+ precursor. This means that in aged tissues where CD38 expression is elevated, supplemental NMN may be partially consumed by CD38 before it can be converted to NAD+. The enzyme does not just deplete existing NAD+ — it also intercepts the raw materials used to make more.
NAD+ is required for sirtuin-dependent circadian clock regulation and for mitochondrial energy production during sleep. When CD38 depletes NAD+ below functional thresholds, both of these downstream sleep-regulatory pathways are compromised. This creates a link between age-related CD38 upregulation and the sleep maintenance problems that become more common with age.
How Does Apigenin Block CD38 and Raise NAD+ Levels?
The characterization of apigenin as a novel CD38 inhibitor was published by Escande et al. (2013) in Diabetes. The researchers quantified IC50 values — the concentration at which apigenin inhibits 50% of enzymatic activity — for CD38’s two enzymatic functions: cyclic ADP-ribose hydrolase and NAD+ase.
In diet-induced obese mice, apigenin increased liver NAD+ concentrations and reduced global protein acetylation, a marker indicating that sirtuin enzymes had been reactivated by the restored NAD+ supply. The apigenin group showed improved glucose tolerance and favorable changes in lipid metabolism compared to vehicle controls (Escande et al., 2013). Both cell-based experiments (n=3 replicates) and in vivo results were consistent, adding weight to the translational relevance.
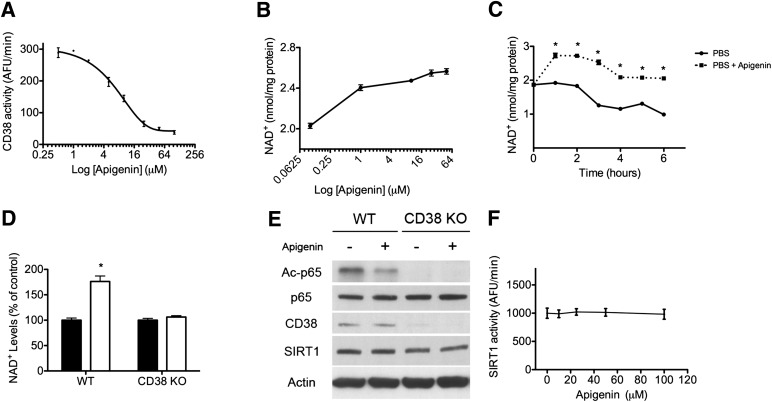
CD38 knockout mice have been shown to be protected against diet-induced obesity, reinforcing the role of CD38 as a metabolic regulator and apigenin’s mechanism of action through that target.
A 2024 review in Frontiers in Nutrition placed apigenin’s CD38 inhibition within the broader sleep-aging intersection (Kramer & Johnson, 2024). The authors reported that dietary apigenin intake correlates with sleep quality in human cohorts and described apigenin’s "electron-rich flavonoid structure" as enabling binding across both aging and sleep targets.
What makes apigenin distinct from other CD38 inhibitors is that apigenin simultaneously acts on GABA-A receptors. Few widely available compounds have this dual mechanism: metabolic preservation through CD38 inhibition and arousal reduction through GABA-A modulation.
Does Apigenin Improve Sleep in Humans?
What does the population data show?
Wang et al. (2023) analyzed dietary flavonoid intake and sleep outcomes in 8,216 adults aged 20 and older drawn from the National Health and Nutrition Examination Survey (NHANES) 2007-2010. After multivariable adjustment, flavones — the subclass that includes apigenin — showed strong protective associations among the flavonoid subclasses measured.
The numbers: adults in the highest flavone intake group had 37% lower odds of insufficient sleep (OR 0.63; 95% CI: 0.41-0.98) and 39% lower odds of sleep disorders (OR 0.61; 95% CI: 0.41-0.90) compared to those with the lowest intake.
Other flavonoid subclasses were also protective — anthocyanidins (OR 0.83), flavonols (OR 0.78), flavan-3-ols (OR 0.91) — but flavones had the numerically largest effect size. The researchers found statistically significant interactions with race/ethnicity, education level, and working status, meaning the protective association varied across subgroups.
This is observational data. It establishes an association between dietary flavone intake and reduced sleep problems at a population level. It does not prove that apigenin caused the improvement.
What do the controlled trials show?
Kazemi et al. (2024) conducted a review of 10 controlled trials with 772 total participants. All trials used chamomile extract — the richest commonly consumed source of apigenin — rather than isolated apigenin.
The primary finding: chamomile supplementation produced a statistically significant reduction in Pittsburgh Sleep Quality Index (PSQI) total scores, with a weighted mean difference (WMD) of -1.88 (95% CI: -3.46 to -0.31) across 5 pooled studies. Three of four eligible studies showed improvements in sleep onset latency — the time it takes to fall asleep.
Improvements were domain-specific. Nighttime awakenings improved in two of three studies, and sleep onset latency improved in three of four studies. Sleep duration and sleep efficiency did not improve across studies. Daytime functioning showed no change in any of the three relevant studies (Kazemi et al., 2024).
No adverse events were documented across all included trials. The authors noted one limitation: only one study verified that blinding was effective, which limits confidence in the precise effect size.
How does this work at the receptor level?
Apigenin modulates GABA-A receptors through a mechanism that appears to be independent of the benzodiazepine binding site (Kramer & Johnson, 2024). Apigenin produces sedative effects through this mechanism.
This receptor-level mechanism may explain the pattern in the trial data: improvements in sleep onset and nighttime awakenings (reduced arousal), but not in sleep duration or efficiency (not increased sleep drive). Apigenin appears to lower the threshold for falling asleep and staying asleep by reducing excitatory neural activity, rather than by extending total sleep time.
What Else Does Apigenin Do to Aging Cells?
A 2025 study published in Advanced Science screened 66 natural compounds across multiple human stromal cell lines to identify senomorphic agents — compounds that suppress the inflammatory output of senescent cells without killing them (Zhang et al., 2025).
Apigenin emerged as a lead candidate. The molecular target: peroxiredoxin 6 (PRDX6). Apigenin blocks PRDX6’s phospholipase A2 activity, which disrupts the ATM/p38MAPK-HSPA8 interaction required for the senescence-associated secretory phenotype (SASP). SASP is the set of inflammatory cytokines that senescent cells release into surrounding tissue, driving chronic inflammation and accelerating aging in neighboring cells.
In prematurely aged mice (n=8 per group), apigenin reduced physical frailty and improved cognitive performance, demonstrating geroprotective effects beyond cell culture (Zhang et al., 2025).
A separate 2024 study screened 143 compounds against human bone marrow stromal stem cells from elderly and young donors (Ali et al., 2024). Apigenin reduced the senescence burden in aged donor cells and restored osteoblastic differentiation — the ability of stem cells to form bone-building cells. Micro-CT imaging showed increased bone volume and cortical thickness in apigenin-exposed samples (n=6 imaging studies).
The multi-target profile is what distinguishes apigenin from single-mechanism sleep supplements. CD38 inhibition preserves NAD+. GABA-A modulation reduces arousal. Senomorphic activity through PRDX6 suppresses the inflammatory secretions of aged cells. These are three independent mechanisms in one compound — addressing both the sleep problem and the underlying aging processes that worsen sleep over time.
Many people experiencing sleep disruption have more than one underlying cause. NAD+ depletion through CD38 might be one factor, but metabolic, hormonal, inflammatory, and circadian mechanisms can overlap — especially after 40. Identifying which causes might be contributing is a useful starting point.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
What Is the Best Apigenin Dose for Sleep?
The gap between the popular 50mg dose and the pharmacologically relevant concentrations in the CD38 research is notable. Escande et al. (2013) quantified IC50 values for CD38 inhibition at concentrations that do not map neatly onto a 50mg oral supplement, especially given apigenin’s low oral bioavailability — it is poorly absorbed in the gastrointestinal tract.
In the Kazemi et al. (2024) meta-analysis, the chamomile trials used whole chamomile extract, which contains apigenin alongside other flavonoids (including apigenin-7-O-glucoside and other glycosylated forms) that may contribute to the observed sleep effects. Isolating apigenin’s specific dose contribution from these extracts is not possible with current data.
Community reports from users following the Huberman sleep stack frequently describe 300-500mg as the range where sleep effects become noticeable. This may reflect the bioavailability constraint: higher doses compensate for low absorption. No controlled dose-response study for isolated apigenin and sleep has been published.
Can Apigenin Replace Melatonin for Sleep?
Melatonin is a circadian phase cue — it tells the brain that darkness has arrived and advances the sleep-wake cycle. Its primary function is timing, not sedation.
Apigenin reduces arousal through GABA-A receptor modulation and preserves NAD+ through CD38 inhibition. These are metabolic and neuroinhibitory mechanisms, not circadian ones.
Use cases differ. Someone whose sleep problem is a delayed circadian phase (falling asleep too late) has a different underlying issue than someone who falls asleep fine but wakes at 3am with a racing mind. Apigenin addresses the latter pattern more directly. In community reports, apigenin is frequently combined with magnesium L-threonate rather than used as a direct melatonin substitute.
Does Apigenin Work Better With Magnesium for Sleep?
The Huberman sleep stack — apigenin + magnesium L-threonate + L-theanine — has become a commonly discussed combination in online sleep supplement communities. The mechanistic rationale is direct: magnesium L-threonate crosses the blood-brain barrier and modulates NMDA receptors, reducing excitatory glutamate activity. Apigenin enhances inhibitory GABAergic activity through GABA-A receptors. These are complementary mechanisms acting on opposing sides of the excitatory-inhibitory balance.
Mechanistic plausibility exists. Controlled evidence does not. No controlled trial has tested apigenin and magnesium L-threonate in combination. The reported efficacy of this stack rests on community anecdotes and the independent mechanistic evidence for each compound.
What Foods Contain High Amounts of Apigenin?
The Wang et al. (2023) NHANES analysis measured dietary flavone intake from food sources. The protective associations with sleep — 37% lower odds of insufficient sleep, 39% lower odds of sleep disorders — were observed at habitual dietary exposure levels, not supplement doses.
Chamomile tea is the bridge between the epidemiological evidence and the supplement market. It is the richest commonly consumed source of apigenin and was the material used in all 10 trials in the Kazemi et al. (2024) review.
Parsley contains high concentrations of apigenin per gram of dry weight, though typical serving sizes are small. Celery and certain citrus fruits provide lower but measurable amounts.
For context: supplement doses of isolated apigenin (50-500mg) exceed what is achievable through dietary intake alone. The epidemiological evidence supports a benefit at food-level exposures. Whether higher supplement doses produce proportionally larger effects is unknown — no dose-response data exists for isolated apigenin and sleep outcomes.
Related Reading
- Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep — the pillar article for metabolic sleep disruption, covering glucose regulation, cortisol rhythm, mitochondrial stress, NAD+ loss, and fat metabolism
- Which Nicotinamide Adenine Dinucleotide Precursor Improves Sleep — Nicotinamide Mononucleotide or Nicotinamide Riboside? — NMN and NR sleep evidence, timing, safety, and human trial differences
- Does NMN Cause Insomnia? What Timing and Dose Matter — NMN timing, dose, circadian effects, and sleep continuity data
- Does Your Body Divert Tryptophan From Melatonin to Make NAD+? — tryptophan allocation between melatonin, kynurenine, inflammation, and NAD+ synthesis
- Why Does Sleep Repair Your DNA — And Burn Through Your NAD+ to Do It? — PARP1, sleep pressure, DNA repair, and NAD+ demand during sleep
- Does Your Gut Decide Whether Your NAD+ Supplement Works? — gut microbiome conversion of NAD+ precursors and why supplement response varies
- Does NAD+ IV Therapy Improve Sleep? What the Evidence Shows — IV NAD+ evidence, safety, infusion effects, and comparison with oral precursors
- Why Does NAD+ Drop Faster in Women After 40 — And What Does That Mean for Sleep? — menopause, ovarian aging, NAD+ decline, and sleep disruption after 40
References
Escande, C., Nin, V., Price, N. L., Capellini, V., Gomes, A. P., Barbosa, M. T., O’Neil, L., White, T. A., Sinclair, D. A., & Chini, E. N. (2013). Flavonoid apigenin is an inhibitor of the NAD+ ase CD38: implications for cellular NAD+ metabolism, protein acetylation, and treatment of metabolic syndrome. Diabetes, 62(4), 1084-1093. https://pubmed.ncbi.nlm.nih.gov/23172919/
Camacho-Pereira, J., Tarragó, M. G., Chini, C. C. S., Nin, V., Escande, C., Warner, G. M., Puranik, A. S., Schoon, R. A., Reid, J. M., Galina, A., & Chini, E. N. (2016). CD38 Dictates Age-Related NAD Decline and Mitochondrial Dysfunction through an SIRT3-Dependent Mechanism. Cell Metabolism, 23(6), 1127-1139. https://pubmed.ncbi.nlm.nih.gov/27304511/
Wang, L., Gui, J., Ding, R., Yang, X., Yang, J., Luo, H., Huang, D., Han, Z., & Jiang, L. (2023). Dietary Intake of Flavonoids Associated with Sleep Problems: An Analysis of Data from the National Health and Nutrition Examination Survey, 2007-2010. Brain Sciences, 13(6), 873. https://pubmed.ncbi.nlm.nih.gov/37371353/
Kramer, D. J., & Johnson, A. A. (2024). Apigenin: a natural molecule at the intersection of sleep and aging. Frontiers in Nutrition, 11, 1359176. https://pubmed.ncbi.nlm.nih.gov/38476603/
Ali, D., Okla, M., Abuelreich, S., Vishnubalaji, R., Ditzel, N., Hamam, R., Kowal, J. M., Sayed, A., Aldahmash, A., Alajez, N. M., & Kassem, M. (2024). Apigenin and Rutaecarpine reduce the burden of cellular senescence in bone marrow stromal stem cells. Frontiers in Endocrinology, 15, 1360054. https://pubmed.ncbi.nlm.nih.gov/38638133/
Kazemi, A., Shojaei-Zarghani, S., Eskandarzadeh, P., & Hashempur, M. H. (2024). Effects of chamomile (Matricaria chamomilla L.) on sleep: A systematic review and meta-analysis of clinical trials. Complementary Therapies in Medicine, 84, 103071. https://pubmed.ncbi.nlm.nih.gov/39106912/
Zhang, H., Xu, Q., Jiang, Z., Sun, R., Wang, Q., Liu, S., Luan, X., Campisi, J., Kirkland, J. L., Zhang, W., & Sun, Y. (2025). Targeting Senescence with Apigenin Improves Chemotherapeutic Efficacy and Ameliorates Age-Related Conditions in Mice. Advanced Science, 12(20), e2412950. https://pubmed.ncbi.nlm.nih.gov/40265973/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 7 references cited
