Perimenopause affects 80-90% of women with disruptive effects (Troia et al., 2025), and sleep disturbances are reported by 16-47% of perimenopausal women. The hormonal mechanisms — estrogen and progesterone reduction affecting sleep architecture — are well documented. But menopause also accelerates a separate metabolic pathway: NAD+ depletion. Ovarian tissue has its own NAD+ economy, and its collapse during menopause compounds hormonal sleep disruption in a way that estrogen-focused approaches do not address.
This article covers what happens to NAD+ metabolism during menopause, why all three synthesis pathways decrease together, and what the current evidence says — and does not say — about NAD+ precursors for menopausal sleep. Estrogen and progesterone sleep mechanisms are covered in the Hormonal Women cluster, not here.
NAD+ depletion is one of several metabolic contributors to sleep disruption. For the full overview of how metabolic factors drive sleep disruption, see Metabolic Sleep Disruption.
What Happens to NAD+ During Menopause?
NAD+ (nicotinamide adenine dinucleotide) is a coenzyme required for cellular energy production, DNA repair, and cell survival. Every person loses NAD+ with age. But women face an additional, tissue-specific depletion tied to reproductive aging.
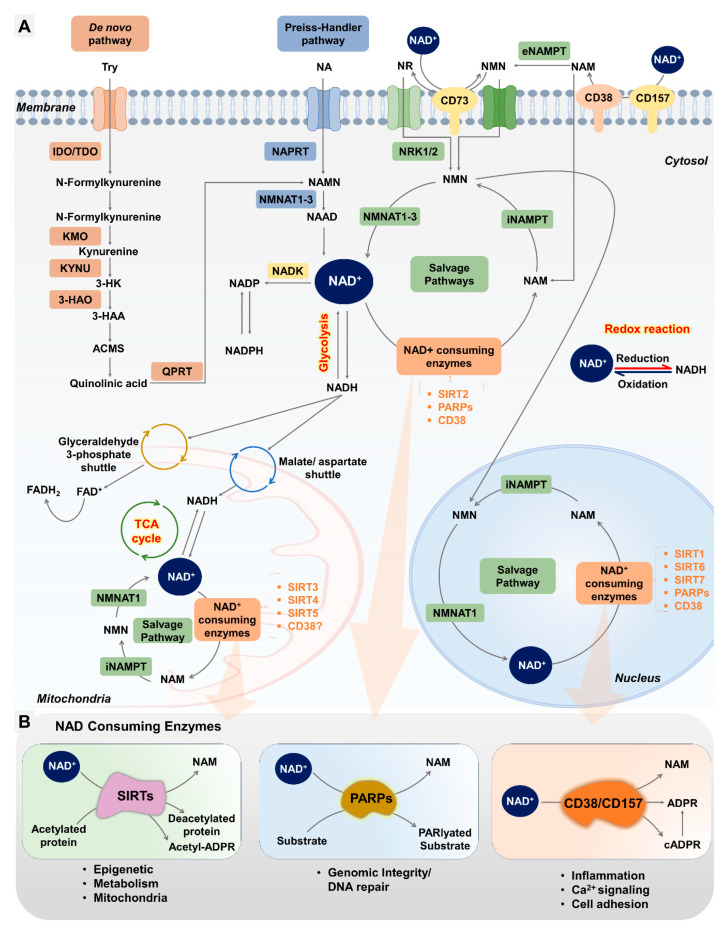
A 2024 review by Ahmed et al. mapped out the ovarian NAD+ depletion mechanism. Three major classes of enzymes consume NAD+ in ovarian cells: sirtuins (SIRT1-7, which regulate gene expression and mitochondrial function), PARPs (which repair DNA damage), and CD38 (a glycohydrolase that breaks down NAD+ and increases with age and inflammation). Each of these enzyme classes ramps up activity with age, creating a compounding depletion — more NAD+ consumed at the same time that less is being produced (Ahmed et al., 2024).
The production decrease is equally notable. Di Emidio et al. (2024) demonstrated that all three NAD+ biosynthetic pathways shut down simultaneously in aging oocytes (egg cells). The kynurenine pathway (which converts tryptophan into NAD+), the Preiss-Handler pathway (which uses dietary niacin), and the salvage pathway (which recycles NAD+ breakdown products back into usable NAD+) all show coordinated gene downregulation. This is not a single-pathway issue — the entire NAD+ production infrastructure weakens at the same time (Di Emidio et al., 2024).
In a 2020 mouse study, Yang et al. showed that nicotinamide riboside (NR, an NAD+ precursor) supplementation increased ovarian NAD+ levels and reversed several markers of ovarian aging — follicle count, oocyte quality, and mitochondrial membrane potential all improved. In vitro exposure of aged oocytes to NR produced similar improvements, suggesting the mechanism acts at the level of the oocyte itself rather than through whole-body factors (Yang et al., 2020). This established the experimental foundation for NAD+ supplementation in reproductive aging, though no human replication exists.
The takeaway: ovarian tissue faces a double hit during menopause. Production of NAD+ drops across all three synthesis routes, while consumption by sirtuins, PARPs, and CD38 increases. This is a tissue-specific metabolic event on top of the age-related NAD+ reduction that affects all tissues.
Does Menopause Accelerate Aging Beyond Hormone Loss?
A 2025 study in Nature Aging by Jin et al. built a single-nuclei multi-omics atlas of human ovarian aging, analyzing 42,568 nuclei from tissue of young donors (ages 23-29) and reproductively aged donors (ages 49-54). The computational integration of gene expression and chromatin accessibility data revealed coordinated changes across all major ovarian cell types during reproductive aging (Jin et al., 2025).
mTOR (mechanistic target of rapamycin, a nutrient-sensing pathway that regulates cell growth) emerged as a prominent ovary-specific aging pathway, with broad upregulation in aged ovaries. The transcription factor CEBPD showed enhanced activity across multiple cell types — an aging-associated epigenetic change accompanying reproductive senescence. Non-coding genetic variants associated with menopause timing were mapped to specific ovarian cell-type gene regulatory networks, linking genetic risk for earlier menopause to disrupted molecular programs in ovarian tissue (Jin et al., 2025).
This epigenetic acceleration accompanies hormonal change. Troia et al. (2025) documented that perimenopausal sleep disruptions span multiple disorder categories — insomnia, sleep-related breathing disorders, restless legs syndrome, periodic limb movement disorder — each with distinct hormonal correlates. Circadian rhythm disruption, melatonin reduction, and mood disorder comorbidity are additive contributors that compound hormone-driven sleep fragmentation (Troia et al., 2025).
During menopause, at least three concurrent mechanisms converge on sleep disruption: hormonal change (estrogen and progesterone) disrupts sleep architecture through thermoregulation and GABAergic tone; NAD+ depletion weakens SIRT1-mediated clock gene regulation; and mTOR upregulation with inflammatory activity adds metabolic stress. These are independent pathways producing overlapping effects — which helps explain why menopause-related sleep disruption is resistant to single-pathway approaches.
Do NAD+ Precursors Help With Menopause?
The Yoshino et al. (2021) trial, published in Science, enrolled 25 postmenopausal women with prediabetes (NMN group: n=13; placebo: n=12) for 10 weeks of oral NMN supplementation at 250 mg/day. NMN increased insulin-stimulated glucose disposal — a measurable improvement in whole-body insulin sensitivity. Skeletal muscle biopsies showed increased phosphorylated AKT and mTOR protein levels in the NMN group. RNA-sequencing revealed upregulation of PDGFRb (platelet-derived growth factor receptor beta) and genes involved in muscle remodeling (Yoshino et al., 2021).
This trial establishes that NMN is bioavailable and biologically active in postmenopausal tissue. It does not establish that NMN improves sleep, hot flashes, or any other menopause-related outcome. The population was overweight or obese with prediabetes — not representative of all menopausal women. And n=25 is a small sample.
Ahmed et al. (2024) note that while NAD+ biochemistry is well characterized in cancer, neurodegeneration, and cardiovascular disease, its impact on female reproduction "is not well understood." This is a direct gap call from the 2024 review literature — the absence of data is the defining feature of this space.
For NAD+ precursors to improve menopausal sleep, the NAD+/SIRT1/clock gene pathway would need to respond to oral supplementation in women whose ovarian NAD+ economy is in simultaneous production collapse and consumption increase. Whether the standard 250 mg NMN dose addresses this tissue-specific deficit is untested. The Di Emidio et al. (2024) study offers one mechanistic thread: P7C3, a pharmacological activator of NAMPT (the rate-limiting enzyme in the salvage pathway), restored mitochondrial function and spindle assembly in aged oocytes, with SIRT1 identified as the central regulatory node. This is preclinical work in reproductive tissue, not sleep tissue, but it identifies the molecular target that would need to respond to supplementation.
How Does Hormonal Sleep Disruption Differ From NAD+ Sleep Disruption?
Coborn et al. (2022) tracked 45 perimenopausal women with daily sleep diaries and weekly hormone measurements over 8 weeks. Lower estradiol levels were independently associated with more frequent nighttime awakenings (p=0.007). Higher FSH (follicle-stimulating hormone) showed a similar association (p=0.02). These associations held after controlling for hot flash frequency and depressive mood — a direct hormone-sleep relationship independent of vasomotor and mood pathways. Progesterone levels showed no significant association with sleep continuity measures (Coborn et al., 2022).
The NAD+/SIRT1 pathway works through a separate mechanism. NAD+ fuels SIRT1, which regulates the clock genes BMAL1 and PER2 — the molecular timekeepers that drive circadian rhythm. When NAD+ declines, SIRT1 activity drops and circadian precision degrades. This is a metabolic clock mechanism, independent of estrogen or progesterone.
The distinction matters for approach. Hormone replacement therapy addresses the estrogen/progesterone pathway. It does not restore NAD+ levels or SIRT1 activity. Conversely, NAD+ precursor supplementation does not replace estrogen or progesterone. A woman in menopause experiencing sleep disruption may have both pathways contributing — and addressing only one may produce incomplete results.
Troia et al. (2025) highlight that there is "still a lack of standards on the appropriate management and care of sleep disorders in perimenopausal women." Their review identifies cognitive behavioral therapy for insomnia (CBT-I) as the first-line non-pharmacological approach, with hormone therapy considered according to individual needs and risk factors. NAD+ is not mentioned in any management framework, consistent with the zero-evidence status for this population.
NAD+ depletion during menopause might be one metabolic contributor to sleep disruption — but blood sugar instability, cortisol rhythm changes, and inflammation might be compounding alongside it. Each responds to different approaches.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Can NAD+ Depletion Affect Sleep With Aging?
The mechanism runs through NAD+ → SIRT1 → BMAL1/PER2 clock genes. When NAD+ levels drop, SIRT1 has less fuel to regulate these clock genes, and circadian timing weakens. In menopausal women, Di Emidio et al. (2024) showed all three NAD+ synthesis pathways — kynurenine, Preiss-Handler, and salvage — decrease in coordination in aging oocytes, with SIRT1 identified as the central regulatory node. This dual-hit (age-related reduction plus ovarian depletion) compresses the timeline of NAD+-related circadian degradation in ways that do not apply to men aging at the same rate.
Does NAD+ Help With Menopause?
The Yoshino 2021 RCT remains the single human data point for NAD+ precursors in postmenopausal women — and it measured metabolic outcomes, not menopause-specific effects. Yang et al. (2020) demonstrated ovarian follicle rescue and improved mitochondrial function with NR in aging mice, establishing preclinical proof-of-concept. The gap: no human trial has tested NAD+ precursors for any menopause-related outcome other than insulin sensitivity.
Does NAD+ Help With Sleep?
Both positive NMN trials enrolled mixed-sex older populations — not menopause-specific cohorts. The gap for this specific population remains open. For the full precursor-by-precursor comparison, see Which Nicotinamide Adenine Dinucleotide Precursor Improves Sleep — Nicotinamide Mononucleotide or Nicotinamide Riboside?.
What Causes Sleep Disruption During Perimenopause?
Coborn et al. (2022) established the estradiol/FSH-sleep association independent of hot flashes and mood. Troia et al. (2025) documented the full spectrum of perimenopausal sleep disorders spanning insomnia, breathing disorders, restless legs, and limb movement. NAD+ functions as a concurrent but separate mechanism. The Hormonal Women cluster covers estrogen and progesterone mechanisms in depth — this article covers the NAD+ component.
Related Reading
- Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep — the pillar article for metabolic sleep disruption, covering glucose regulation, cortisol rhythm, mitochondrial stress, NAD+ loss, and fat metabolism
- Which Nicotinamide Adenine Dinucleotide Precursor Improves Sleep — Nicotinamide Mononucleotide or Nicotinamide Riboside? — NMN and NR sleep evidence, timing, safety, and human trial differences
- Does NMN Cause Insomnia? What Timing and Dose Matter — NMN timing, dose, circadian effects, and sleep continuity data
- Does Apigenin Protect Your NAD+ and Improve Sleep Through CD38 Inhibition? — CD38 inhibition, NAD+ depletion with age, and apigenin sleep evidence
- Does Your Body Divert Tryptophan From Melatonin to Make NAD+? — tryptophan allocation between melatonin, kynurenine, inflammation, and NAD+ synthesis
- Why Does Sleep Repair Your DNA — And Burn Through Your NAD+ to Do It? — PARP1, sleep pressure, DNA repair, and NAD+ demand during sleep
- Does Your Gut Decide Whether Your NAD+ Supplement Works? — gut microbiome conversion of NAD+ precursors and why supplement response varies
- Does NAD+ IV Therapy Improve Sleep? What the Evidence Shows — IV NAD+ evidence, safety, infusion effects, and comparison with oral precursors
References
Ahmed, M., Riaz, U., Lv, H., & Yang, L. (2024). A Molecular Perspective and Role of NAD+ in Ovarian Aging. International Journal of Molecular Sciences, 25(9), 4680. https://pubmed.ncbi.nlm.nih.gov/38731898/
Coborn, J., de Wit, A., Crawford, S., Nathan, M., Rahman, S., Finkelstein, L., Wiley, A., & Joffe, H. (2022). Disruption of Sleep Continuity During the Perimenopause: Associations with Female Reproductive Hormone Profiles. The Journal of Clinical Endocrinology and Metabolism, 107(10), e4144-e4153. https://pubmed.ncbi.nlm.nih.gov/35878624/
Di Emidio, G., Vergara, T., Konstantinidou, F., Flati, I., Stuppia, L., Artini, P. G., Gatta, V., Falone, S., & Tatone, C. (2024). NAD+ Metabolism and Mitochondrial Activity in the Aged Oocyte: Focus on the Effects of NAMPT Stimulation. Aging and Disease, 15(6), 2828-2851. https://pubmed.ncbi.nlm.nih.gov/39325941/
Jin, C., Wang, X., Yang, J., Kim, S., Hudgins, A. D., Gamliel, A., Pei, M., Contreras, D., Devos, M., Guo, Q., Vijg, J., Conti, M., Hoeijmakers, J. H. J., Campisi, J., Lobo, R., Williams, Z., Rosenfeld, M. G., & Suh, Y. (2025). Molecular and genetic insights into human ovarian aging from single-nuclei multi-omics analyses. Nature Aging, 5(2), 275-290. https://pubmed.ncbi.nlm.nih.gov/39578560/
Kim, M., Seol, J., Sato, T., Fukamizu, Y., Sakurai, T., & Okura, T. (2022). Effect of 12-Week Intake of Nicotinamide Mononucleotide on Sleep Quality, Fatigue, and Physical Performance in Older Japanese Adults: A Randomized, Double-Blind Placebo-Controlled Study. Nutrients, 14(4), 755. https://pubmed.ncbi.nlm.nih.gov/35215405/
Morifuji, M., Higashi, S., Ebihara, S., & Nagata, M. (2024). Ingestion of β-nicotinamide mononucleotide increased blood NAD levels, maintained walking speed, and improved sleep quality in older adults in a double-blind randomized, placebo-controlled study. GeroScience, 46(5), 4671-4688. https://pubmed.ncbi.nlm.nih.gov/38789831/
Troia, L., Garassino, M., Volpicelli, A. I., Fornara, A., Libretti, A., Surico, D., & Remorgida, V. (2025). Sleep Disturbance and Perimenopause: A Narrative Review. Journal of Clinical Medicine, 14(5), 1479. https://pubmed.ncbi.nlm.nih.gov/40094961/
Yang, Q., Cong, L., Wang, Y., Luo, X., Li, H., Wang, H., Zhu, J., Dai, S., Jin, H., Yao, G., Shi, S., Hsueh, A. J., & Sun, Y. (2020). Increasing ovarian NAD+ levels improve mitochondrial functions and reverse ovarian aging. Free Radical Biology and Medicine, 156, 1-10. https://pubmed.ncbi.nlm.nih.gov/32492457/
Yoshino, M., Yoshino, J., Kayser, B. D., Patti, G. J., Franczyk, M. P., Mills, K. F., Sindelar, M., Pietka, T., Patterson, B. W., Imai, S. I., & Klein, S. (2021). Nicotinamide mononucleotide increases muscle insulin sensitivity in prediabetic women. Science, 372(6547), 1224-1229. https://pubmed.ncbi.nlm.nih.gov/33888596/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 9 references cited
