Chronic inflammation is common in adults over 40 — from autoimmune conditions, metabolic changes, gut permeability, or accumulated physiological stress. When sleep deteriorates alongside it, the two problems are usually addressed separately. But at the molecular level, they share machinery: the same clock genes that time your circadian rhythm are direct targets of inflammatory molecules.
This article covers how specific inflammatory cytokines suppress specific clock genes, which clocks in the body are affected, and whether the damage runs in both directions. It does not cover inflammation’s direct effects on sleep architecture — for that broader context, see the circadian sleep disruption pillar. Circadian clock gene suppression is one mechanism within a larger set of causes that fragment sleep after age 40 — and understanding the molecular pathway matters because it changes what the solutions look like.
?-
How Do Inflammatory Cytokines Suppress Circadian Clock Genes?
The molecular clock runs on a transcription-translation feedback loop that repeats approximately every 24 hours. Two proteins — CLOCK and BMAL1 — bind together and attach to DNA sequences called E-box elements, which activate the genes PER1, PER2, PER3, CRY1, and CRY2. Those PER and CRY proteins accumulate over hours, then feed back to inhibit CLOCK-BMAL1, suppressing their own production. When PER and CRY levels decline, CLOCK-BMAL1 activation resumes, and the cycle restarts. This loop runs in every nucleated cell in the body — it is the molecular basis of circadian timing.
Inflammatory cytokines interfere with this loop at the E-box step. In a 2007 study, Cavadini et al. demonstrated that TNF-alpha suppresses Per1, Per2, Per3 and the clock-controlled genes Dbp, Tef, and Hlf by directly inhibiting CLOCK-BMAL1 activation of E-box elements (Cavadini et al., 2007). IL-1-beta shares this effect. IFN-alpha and IL-6 do not — establishing that clock gene suppression is cytokine-specific, not a general consequence of inflammation.
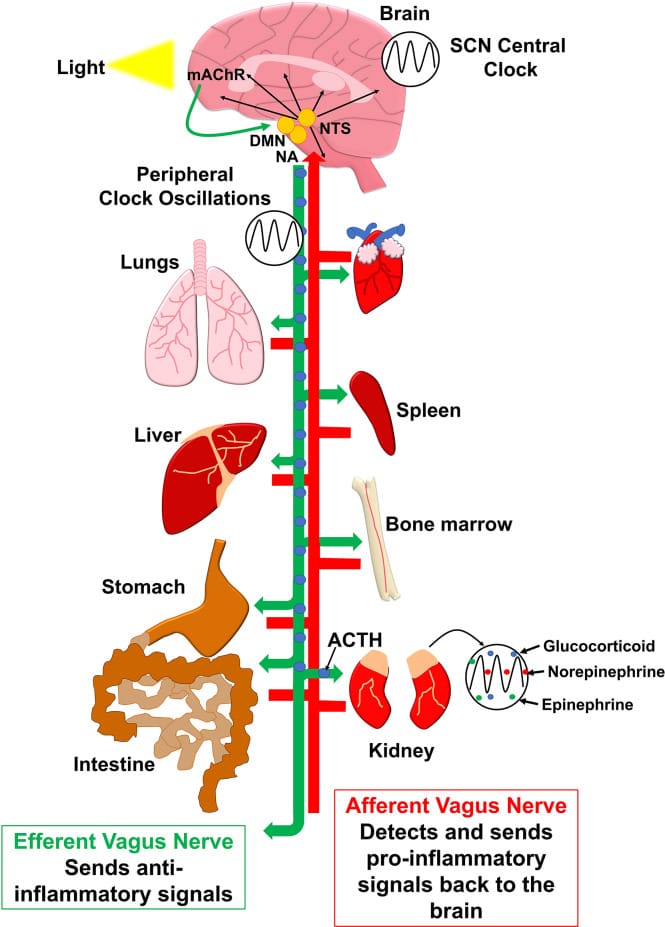
The NF-kB pathway adds a second route of interference. Guo et al. (2015) showed that IL-1-beta disrupts circadian gene expression in cartilage tissue through NF-kB — a protein complex that activates inflammatory genes and, in doing so, functionally interferes with the CLOCK-BMAL1 complex (Guo et al., 2015). When NF-kB is activated by immune molecules, it can interfere with CLOCK-BMAL1 transcriptional activity — reducing the circadian machinery’s ability to drive gene expression (Guo et al., 2015). The result is that the molecular oscillator loses amplitude, and in some cases, stops oscillating.
?-
Which Circadian Clock Genes Does Inflammation Target?
BMAL1 is the primary transcription factor — the component that initiates the entire feedback loop. IL-1-beta suppresses BMAL1 through the NF-kB pathway. Alhilali et al. (2021) demonstrated in human chondrocytes that IL-1-beta decreases BMAL1 through NF-kB activation while simultaneously increasing PER2 expression through the NMDA receptor/CREB pathway — showing that a single cytokine can disrupt clock gene balance through parallel molecular mechanisms (Alhilali et al., 2021). When BMAL1 is suppressed, the entire transcription-translation loop loses its driving force. The clock does not stop abruptly — its amplitude dampens, meaning the peaks and troughs of gene expression flatten. Sleep-wake boundaries blur because the molecular distinction between “day mode” and “night mode” weakens.
PER1, PER2, and PER3 — the period genes — are the timing components. TNF-alpha blocks their E-box-mediated activation (Cavadini et al., 2007). PER proteins determine when the clock’s negative feedback phase begins, so their suppression produces timing instability: circadian phase becomes unpredictable, and sleep pressure arrives at the wrong times. IL-1-beta increases PER2 expression through the NMDA/CREB pathway (Alhilali et al., 2021), which may further destabilize clock oscillation by disrupting the normal ratio between PER and BMAL1.
CRY1 and CRY2 — the cryptochrome genes — may also be affected when E-box activation is blocked, since CRY genes are E-box-driven — though this has not been directly measured in cytokine studies. CRY proteins help determine circadian period length (how long one full cycle takes). When CRY expression is reduced, the circadian period can shorten or become unstable.
Clock-controlled output genes (Dbp, Tef, Hlf) are also suppressed by TNF-alpha (Cavadini et al., 2007). These genes regulate downstream rhythms in metabolism, hormone release, and immune timing. Their suppression explains why chronic inflammation disrupts not just sleep but the timing of cortisol, body temperature, and immune cell trafficking — everything the clock coordinates.
?-
Does Inflammation Hit Your Central Clock, Your Peripheral Clocks, or Both?
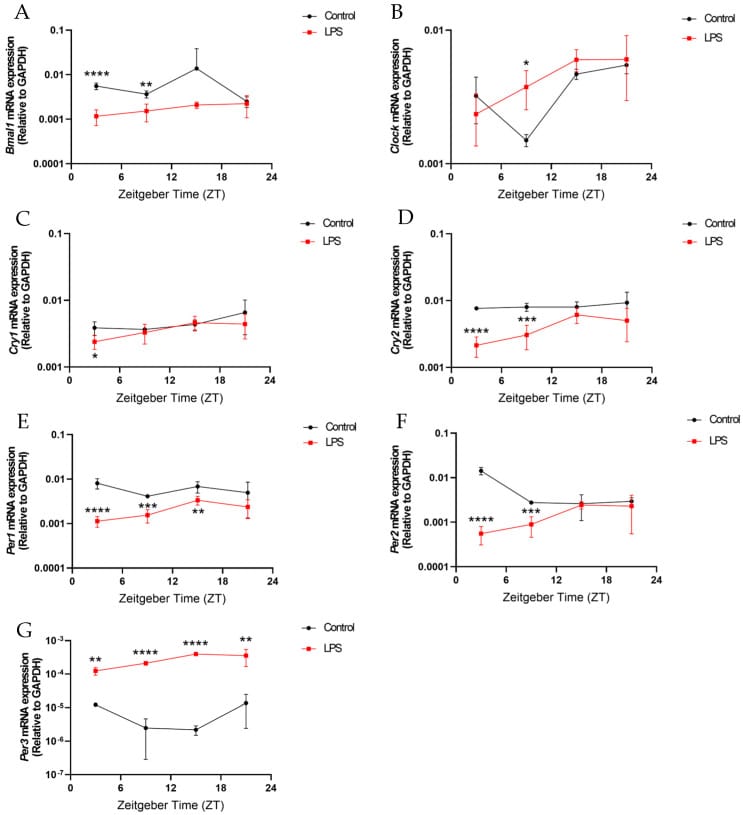
The central clock sits in the suprachiasmatic nucleus (SCN) of the hypothalamus. It coordinates the timing of every peripheral clock in the body and was long considered protected from circulating inflammatory molecules by the blood-brain barrier. But neuroinflammation bypasses that barrier. Cheng et al. (2024) demonstrated in a mouse model that peripheral inflammatory challenge (LPS injection) perturbs Bmal1 oscillations in both the hypothalamus and hippocampus — meaning inflammation originating outside the brain propagates clock disruption into the brain itself (Cheng et al., 2024). In the hippocampus, the diurnal rhythmicity of microglia (the brain’s resident immune cells) was disrupted, which connects to cognitive effects — impaired memory consolidation, reduced concentration — that often accompany chronic inflammation.
Peripheral clocks in the liver, gut, muscle, and cartilage are directly exposed to circulating cytokines and are more immediately vulnerable. Guo et al. (2015) showed in cartilage tissue that IL-1-beta disrupts the clock through NF-kB (Guo et al., 2015). In inflammatory bowel disease, BMAL1 expression may be suppressed in colon tissue, disrupting local immune timing and tissue repair cycles. These peripheral clocks control organ-specific functions: the liver clock times metabolic processes, the gut clock times immune surveillance, and the muscle clock times repair.
Neuroinflammatory signaling may amplify this process — when microglia are chronically activated, they produce pro-inflammatory cytokines including TNF-alpha that can further suppress clock gene expression in surrounding neural tissue (Zielinski & Gibbons, 2022; Cheng et al., 2024). When organ clocks desynchronize from each other and from the SCN, the result extends beyond sleep fragmentation. Metabolism, immune function, and tissue repair all lose coordinated timing. A person with chronic inflammation may experience not just disrupted sleep but misaligned hunger, temperature regulation, and immune responses — because the peripheral clocks that time those functions are running on different schedules.
?-
Is the Damage Bidirectional — Does a Disrupted Clock Also Make Inflammation Worse?
Pferdehirt et al. (2022) provided direct experimental evidence of the bidirectional relationship using engineered tissue. They showed that IL-1-induced inflammation disrupts the circadian clock — and preserving clock function through IL-1 blockade prevents tissue damage. When they deployed three different synthetic gene circuits to block IL-1 and preserve clock function, circadian oscillation amplitude and period were maintained, and tissue damage was prevented (Pferdehirt et al., 2022). This indicates the clock is not incidental to inflammation — it is an active component of how disease progresses. (Note: this was demonstrated in engineered murine cartilage tissue; the same bidirectional dynamic in human inflammatory disease is supported by the lupus data below but has not been tested with the same gene-circuit approach in humans.)
Nakabo et al. (2024) extended this finding to autoimmune disease. In lupus, BMAL1 loss does not just result from autoimmune inflammation — it drives disease progression. In a mouse model, myeloid-specific Bmal1 deficiency promoted autoantibody production, immune complex deposition, and disrupted neutrophil homeostasis. In a human lupus cohort, BMAL1 expression was inversely correlated with disease activity: the more active the disease, the lower the BMAL1 levels, and the lower the BMAL1 levels, the more the disease progressed (Nakabo et al., 2024).
This bidirectional relationship reframes chronic inflammation and poor sleep from two separate problems to one self-reinforcing cycle with a shared molecular mechanism. It also means that supporting the circadian clock — through consistent light exposure timing, meal timing, and sleep regularity — may function as an anti-inflammatory strategy, not just a sleep strategy. For adults over 40 managing conditions like rheumatoid arthritis, inflammatory bowel disease, or lupus, circadian regularity may be more relevant than it initially appears — because the clock is part of the disease machinery, not a bystander.
?-
What Is Nuclear Factor Kappa B and How Does It Affect the Circadian Clock?
NF-kB has a dual role: it is essential for immune defense, activating the genes that produce inflammatory cytokines in response to infection or tissue damage. But when chronically activated — as it is in persistent low-grade inflammation — that same activity interferes with circadian timing. The interference happens at the molecular level: NF-kB activation reduces the ability of CLOCK-BMAL1 to drive the transcription-translation feedback loop (Guo et al., 2015). This means NF-kB does not destroy the clock components — it interferes with their transcriptional activity, reducing the amplitude of circadian gene expression rather than eliminating it.
?-
Do Inflammatory Cytokines Suppress Clock Gene Expression?
This cytokine specificity is an important distinction. Not all inflammation disrupts the clock equally. Cavadini et al. (2007) tested TNF-alpha, IL-1-beta, IFN-alpha, and IL-6 for their effects on clock gene expression. TNF-alpha and IL-1-beta both suppressed E-box-mediated transcription. IFN-alpha and IL-6 did not (Cavadini et al., 2007). This matters for understanding why some inflammatory conditions fragment sleep more than others: conditions dominated by TNF-alpha or IL-1-beta (such as rheumatoid arthritis, inflammatory bowel disease, and obesity-related inflammation) may have stronger effects on circadian clock integrity than conditions driven by other cytokine profiles.
?-
What Is the Connection Between Neuroinflammation and Circadian Rhythm?
When microglia remain in an activated state — as occurs in neurodegenerative conditions, or sustained peripheral inflammation that propagates into the brain — they produce the same TNF-alpha and IL-1-beta that suppress clock genes in peripheral tissue (Zielinski & Gibbons, 2022). But the consequences are amplified because the central clock in the SCN coordinates every other clock in the body. Cheng et al. (2024) showed in mice that peripheral inflammation disrupts hippocampal microglial rhythmicity — meaning the brain’s immune cells lose their own circadian timing, which may contribute to sustained disruption of clock gene expression in surrounding neural tissue (Cheng et al., 2024). The experience of feeling exhausted but unable to sleep well can stem from this central clock disruption: the brain’s sleep-wake coordination weakens when the molecular machinery that times it is suppressed by the brain’s own immune cells.
?-
Circadian clock gene disruption from chronic inflammation may compound with other causes of sleep fragmentation — hormonal changes, metabolic imbalance, autonomic imbalance, or medication effects. In adults over 40 managing inflammatory conditions, multiple causes often overlap. Identifying which causes might be contributing is a useful next step.
Find out which causes might be driving your 3 AM wakeups →
?-
Related Reading
- Circadian Rhythm Disruption: How a Misaligned Internal Clock Fragments Sleep, Drives 3AM Wakeups, and Reduces Sleep Depth — the full overview of circadian sleep disruption, including melatonin, cortisol, body temperature, peripheral clocks, and early-morning waking
- Does Your Circadian Clock Control When Insulin Works? Prediabetes, the Dawn Phenomenon, and Sleep — how circadian insulin timing, glucose rhythm, and dawn physiology affect sleep continuity
- Can Fixing Your Gut Fix Your Sleep? How Gut Bacteria Entrain Your Body Clock — how gut bacteria, the vagus nerve, and microbial rhythms interact with sleep timing
- Can Your Body Temperature Keep You From Falling Asleep? — how thermoregulation, vasodilation, and temperature rhythm affect sleep onset and depth
- Why Are You Still Exhausted After Months on CPAP? — how intermittent hypoxia can suppress clock genes even after breathing normalizes
- Do Beta Blockers Suppress Melatonin? What Blood Pressure Medication Does to Your Body Clock — how beta blockers, melatonin suppression, and medication timing affect circadian sleep
References
- Alhilali, M., Hearn, J. I., Rong, J., Jain, L., Bolam, S. M., Monk, A. P., Munro, J. T., Dalbeth, N., & Poulsen, R. C. (2021). IL-1β induces changes in expression of core circadian clock components PER2 and BMAL1 in primary human chondrocytes through the NMDA receptor/CREB and NF-κB signalling pathways. Cellular Signalling, 87, 110143. https://pubmed.ncbi.nlm.nih.gov/34481895/
- Cavadini, G., Petrzilka, S., Kohler, P., Jud, C., Tobler, I., Birchler, T., & Fontana, A. (2007). TNF-α suppresses the expression of clock genes by interfering with E-box-mediated transcription. Proceedings of the National Academy of Sciences, 104(31), 12843–12848. https://pubmed.ncbi.nlm.nih.gov/17646651/
- Cheng, W. Y., Chan, P. L., Ong, H. Y., Wong, K. H., & Chang, R. C. C. (2024). Systemic inflammation disrupts circadian rhythms and diurnal neuroimmune dynamics. International Journal of Molecular Sciences, 25(13), 7458. https://pubmed.ncbi.nlm.nih.gov/39000563/
- Guo, B., Yang, N., Borysiewicz, E., Dudek, M., Williams, J. L., Li, J., Maywood, E. S., Adamson, A., Hastings, M. H., Bateman, J. F., White, M. R. H., Boot-Handford, R. P., & Meng, Q. J. (2015). Catabolic cytokines disrupt the circadian clock and the expression of clock-controlled genes in cartilage via an NF-κB-dependent pathway. Osteoarthritis and Cartilage, 23(11), 1981–1988. https://pubmed.ncbi.nlm.nih.gov/26521744/
- Nakabo, S., Sandoval-Heglund, D., Hanata, N., Brooks, S., Hoffmann, V., Zhang, M., Ambler, W., Manna, Z., Poncio, E., Hasni, S., Islam, S., Dell’Orso, S., & Kaplan, M. J. (2024). The circadian clock gene BMAL1 modulates autoimmunity features in lupus. Frontiers in Immunology, 15, 1465185. https://pubmed.ncbi.nlm.nih.gov/39664388/
- Pferdehirt, L., Damato, A. R., Dudek, M., Meng, Q. J., Herzog, E. D., & Guilak, F. (2022). Synthetic gene circuits for preventing disruption of the circadian clock due to interleukin-1–induced inflammation. Science Advances, 8(21), eabj8892. https://pubmed.ncbi.nlm.nih.gov/35613259/
- Zielinski, M. R., & Gibbons, A. J. (2022). Neuroinflammation, sleep, and circadian rhythms. Frontiers in Cellular and Infection Microbiology, 12, 853096. https://pubmed.ncbi.nlm.nih.gov/35392608/
Written by Kat Fu, M.S., M.S. ? Last reviewed: May 2026 ? 7 references cited
