Oral micronized progesterone can improve sleep onset and subjective sleep quality. A meta-analysis of 9 randomized controlled trials found it reduced sleep onset latency by approximately 7 minutes. The mechanism is well-characterized: oral progesterone is converted to allopregnanolone, which modulates GABA-A receptors in a manner similar to benzodiazepines. This sedative effect requires oral administration — vaginal progesterone does not produce the same sleep benefit.
Progesterone is increasingly discussed as a sleep-promoting hormone, particularly for perimenopausal and menopausal women. The evidence supports this — but the effect depends on the form, the route, and the dose.
This article covers how progesterone promotes sleep through the allopregnanolone–GABA-A pathway, what the randomized controlled trials show about efficacy, the dose and route that matter, and the paradoxical insomnia some women experience. For the broader hormone replacement picture, see the HRT and sleep article. For the full hormonal landscape, see the pillar.
Progesterone addresses one hormonal pathway. Other contributors — cortisol dysregulation, circadian disruption, inflammatory load — may persist independently. Women who experience 3am waking during menopause or the wired-but-tired pattern often have more than one contributor at play.
How Does Progesterone Promote Sleep?
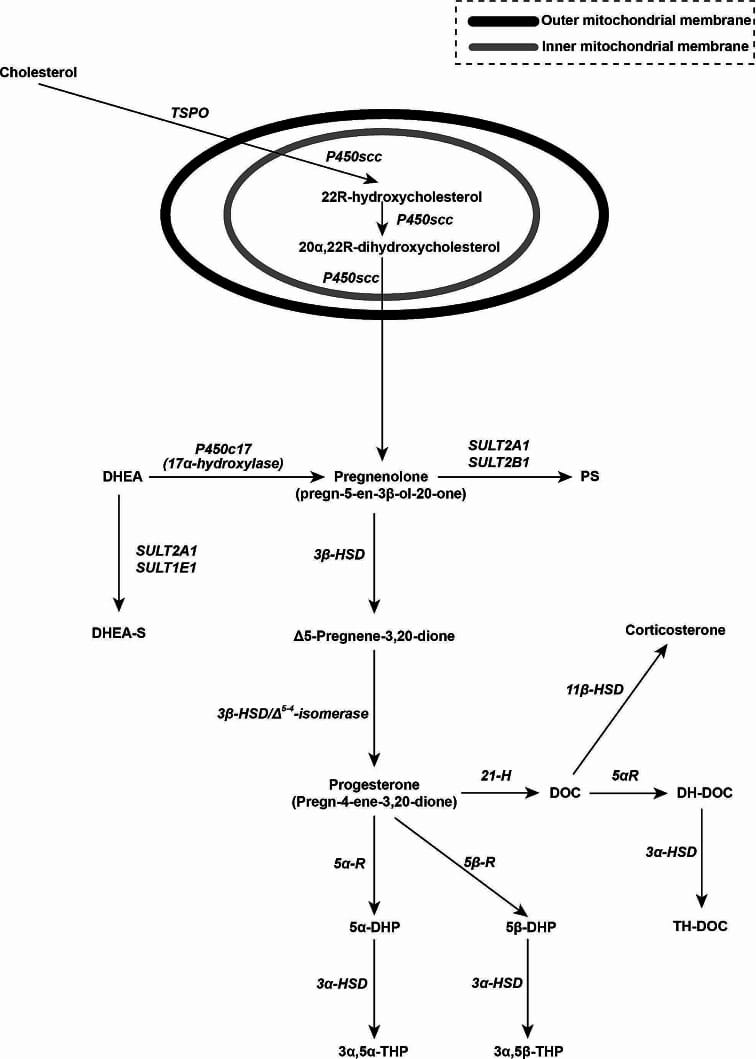
Oral progesterone is metabolized to allopregnanolone, a neurosteroid that positively modulates GABA-A receptors. This produces sedative and anxiolytic effects comparable to benzodiazepines. The effect is route-dependent: oral administration requires first-pass hepatic metabolism to produce allopregnanolone, while vaginal progesterone bypasses this conversion and does not produce the same sleep benefit.
When you take progesterone orally, it passes through the liver before reaching the bloodstream. During this first-pass metabolism, enzymes convert progesterone into several neurosteroid metabolites — the one that matters for sleep being allopregnanolone (also called 3alpha-5alpha-tetrahydroprogesterone).
Allopregnanolone binds to the neurosteroid site on the GABA-A receptor — the same receptor targeted by benzodiazepines and barbiturates. When allopregnanolone binds, it enhances the inhibitory action of GABA, the brain’s primary inhibitory neurotransmitter. The result: reduced neural excitability, decreased anxiety, and sedation (Memi et al., 2024).
This is a direct pharmacological effect, not an indirect one. Estrogen influences sleep through different pathways — serotonergic and thermoregulatory circuits that reduce nocturnal hot flashes and awakenings. Progesterone works through GABA-A modulation, producing sedation independent of temperature regulation or serotonin.
A 2004 double-blind, placebo-controlled study by Soderpalm et al. demonstrated this in both men and women. After 200 mg intramuscular progesterone, plasma allopregnanolone increased reliably, and participants showed increased fatigue and impaired smooth pursuit eye movement — a validated marker of central nervous sedation. The effects were comparable between males and females, indicating that the sedative mechanism is driven by the metabolite, not by reproductive context (Soderpalm et al., 2004).
One distinction matters for anyone considering progesterone for sleep: the route of administration determines whether this conversion occurs. Oral micronized progesterone undergoes first-pass hepatic metabolism, producing allopregnanolone. Vaginal progesterone bypasses the liver and does not produce the same neurosteroid conversion — and does not produce the same sedative effect (Memi et al., 2024).
Haufe and Leeners (2023) examined reproductive hormone contributions to sleep across a woman’s lifespan in a narrative review and identified progesterone as the reproductive hormone with the strongest replicated association with sleep promotion. Sleep disruption peaks during the late luteal phase, when progesterone concentrations fall sharply — a pattern that maps directly onto the allopregnanolone–GABA-A pathway described above (Haufe & Leeners, 2023).
What Does the Evidence Show About Oral Micronized Progesterone and Sleep?
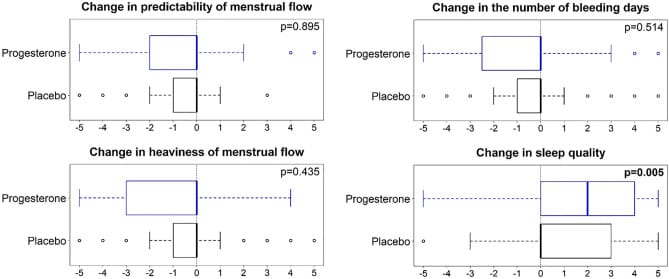
A meta-analysis of 9 randomized controlled trials found oral micronized progesterone reduced sleep onset latency by 7 minutes compared to placebo. Total sleep time trended toward improvement by nearly 21 minutes but did not reach significance. In a Phase III trial, 189 perimenopausal women receiving progesterone at 300 mg at bedtime reported improved sleep quality (P=0.005) and reduced night sweats (P=0.023).
What Did the Meta-Analysis Find?
Nolan et al. (2021) pooled data from 9 randomized controlled trials with 388 participants and found that micronized progesterone reduced sleep onset latency by 7.10 minutes (95% CI: 1.30 to 12.91) — the only parameter reaching statistical significance. Total sleep time and sleep efficiency did not reach significance.
Nolan et al. (2021) conducted a meta-analysis of 9 randomized controlled trials enrolling 388 participants — predominantly postmenopausal women. All studies employing polysomnography used doses of 200–300 mg oral micronized progesterone.
The pooled results:
- Sleep onset latency improved by 7.10 minutes (95% CI: 1.30 to 12.91) — the only parameter reaching statistical significance in the meta-analysis
- Total sleep time trended toward improvement by 20.72 minutes (95% CI: -0.16 to 41.59) but did not reach significance
- Sleep efficiency showed no benefit (effect size 1.31%; 95% CI: -2.09 to 4.70)
- Self-reported sleep quality improved in the majority of individual trials
Progesterone helps people fall asleep faster, with a modest trend toward longer sleep duration. It does not appear to improve the percentage of time spent asleep once in bed (Nolan et al., 2021).
What Did the Phase III Perimenopause Trial Show?
Prior et al. (2023) randomized 189 perimenopausal women (mean age 50, 63% in late perimenopause) to 300 mg oral micronized progesterone at bedtime or placebo for three months. Sleep quality improved with progesterone (P=0.005), and night sweats decreased (P=0.023).
Prior et al. (2023) conducted a Phase III double-blind randomized controlled trial across Canada in 189 perimenopausal women (mean age 50, with 63% in late perimenopause). Participants received 300 mg oral micronized progesterone at bedtime or placebo for three months.
The results relevant to sleep:
- Sleep quality improved with progesterone compared to placebo (P=0.005)
- Night sweats decreased with progesterone (P=0.023)
- Perimenopausal life interference decreased more with progesterone (P=0.017)
- Completion rate was 93%, with 91.4% medication adherence — indicating the regimen was well-tolerated
The primary endpoint — objective vasomotor score at month three — did not reach statistical significance (P=0.222). Sleep quality improvement, however, was robust (Prior et al., 2023).
What Did the 2025 Pilot Study Find?
Ogawa et al. (2025) studied 15 peri- and postmenopausal Japanese women receiving estradiol and oral micronized progesterone. At baseline, 86.7% were classified as poor sleepers. PSQI global scores improved from 7.8 at baseline to 6.1 at month one and 6.5 at month three.
Ogawa et al. (2025) conducted a prospective pilot study in 15 peri- and postmenopausal Japanese women receiving estradiol and oral micronized progesterone. At baseline, 86.7% were classified as poor sleepers (Pittsburgh Sleep Quality Index global score of 6 or higher).
PSQI global scores improved from a baseline mean of 7.8 to 6.1 at month one and 6.5 at month three. Women who responded best — those achieving at least 25% improvement in PSQI score at month one — had worse baseline sleep efficiency than non-responders. Menopausal complaint severity at baseline did not differ between responders and non-responders, suggesting that sleep improvement was not secondary to vasomotor relief (Ogawa et al., 2025).
Across these trials, the 300 mg bedtime dose is the best-studied regimen.
Can Progesterone Cause Insomnia?
Some women report worsened sleep on progesterone. The mechanism may involve individual variation in allopregnanolone metabolism or GABA-A receptor sensitivity. Progesterone has biphasic effects — sustained levels promote sleep, but rapid elevation or abrupt changes can increase wake time after sleep onset.
This is a common question with an uncommon answer. Progesterone is a sedative through the GABA-A pathway — and yet some women report that it keeps them awake or worsens their sleep.
Soderpalm et al. (2004) provide one piece of the explanation. While progesterone reliably increased allopregnanolone levels and produced sedation across participants, the sedative effects were not proportional to plasma allopregnanolone concentrations. This suggests that GABA-A receptor sensitivity varies between individuals. Two women with identical allopregnanolone levels could have different sedative responses — one feeling drowsy, the other feeling agitated (Soderpalm et al., 2004).
Biphasic effects of progesterone add a second dimension. During the luteal phase of the menstrual cycle, sustained progesterone levels are associated with improved sleep. But rapid progesterone elevation — or abrupt withdrawal, as occurs premenstrually — can increase wake time after sleep onset. The GABA-A mechanism that promotes sleep under stable conditions may produce paradoxical arousal when receptor exposure changes quickly.
Dose may also matter. Prior (2018) reviewed evidence from a double-blind randomized controlled trial in 133 postmenopausal women using 300 mg oral micronized progesterone at bedtime. At this dose, progesterone enhanced deep sleep quality and did not cause depression. Lower doses may not produce sufficient allopregnanolone to engage the GABA-A receptor meaningfully, and the relationship between dose and effect is not linear (Prior, 2018).
For women who report insomnia after starting progesterone, several factors are relevant: the dose (200 mg versus 300 mg), the timing relative to sleep onset, and whether the progesterone is micronized and taken orally. Women who experience melatonin underperforming during menopause may have additional upstream contributors that progesterone alone does not address.
The paradox has a plausible mechanism. It does not mean progesterone is the wrong approach for those women — it may mean the dose, timing, or route needs adjustment.
Progesterone addresses one pathway to sleep disruption. Many women have more than one contributor — cortisol dysregulation, circadian disruption, inflammatory load, and metabolic factors can each independently fragment sleep and may persist regardless of progesterone status. Identifying which causes might be involved is a useful next step.
Find out which causes might be driving your 3am wakeups
Frequently Asked Questions
What Dose of Progesterone Is Used for Sleep?
The dose with the strongest trial support is 300 mg oral micronized progesterone at bedtime. The meta-analysis by Nolan et al. (2021) included studies using 200–300 mg. The Phase III randomized controlled trial by Prior et al. (2023) used 300 mg. The sedative effect comes from first-pass metabolism producing allopregnanolone, so timing at bedtime aligns peak sedation with the sleep window (Nolan et al., 2021; Prior, 2018).
How Quickly Does Progesterone Make You Drowsy?
Oral micronized progesterone produces drowsiness within 30–60 minutes of ingestion, aligning with the time required for hepatic conversion to allopregnanolone. This is why bedtime dosing is standard — the sedative effect peaks as the person is falling asleep (Memi et al., 2024).
Does Progesterone Help with Sleep Apnea?
Progesterone has respiratory stimulant properties that may benefit upper airway tone during sleep. However, the evidence for progesterone as a standalone approach for sleep apnea is limited. In the context of menopausal sleep-disordered breathing, combined hormone replacement (estrogen + progesterone) addresses the loss of estrogen’s upper airway protective effects alongside progesterone’s respiratory and sedative contributions (Troìa et al., 2025).
Is It Estrogen or Progesterone That Helps Sleep More?
Estrogen and progesterone improve sleep through different pathways. Estrogen modulates serotonergic and thermoregulatory circuits, reducing nocturnal hot flashes and awakenings. Progesterone produces direct sedation through GABA-A receptor modulation via allopregnanolone. In meta-analysis, estrogen combined with micronized progesterone outperformed estrogen alone for sleep quality (Pan et al., 2022; Haufe & Leeners, 2023).
Related Reading
- Hormonal Sleep Disruption in Women — How estrogen, progesterone, and other hormones affect sleep across a woman’s lifespan
- Why Do You Wake Up at 3am During Menopause? — How cortisol and progesterone loss drive nocturnal waking in menopause
- Why Doesn’t Melatonin Work for Menopause Insomnia? — Why melatonin supplementation underperforms when estrogen decline is the upstream cause
- What Causes the “Wired but Tired” Feeling in Menopause? — How HPA axis changes and autonomic imbalance produce exhaustion with hyperarousal
- Why Can’t You Sleep Before Your Period? — How the progesterone crash in the late luteal phase fragments sleep and raises core body temperature
- Does Hormone Replacement Therapy Help with Sleep in Menopause? — What the clinical evidence shows about HRT’s effect on sleep architecture and hot-flash-driven waking
References
1. Haufe, A., & Leeners, B. (2023). Sleep disturbances across a woman’s lifespan: What is the role of reproductive hormones? Journal of the Endocrine Society, 7(5), bvad036. https://pubmed.ncbi.nlm.nih.gov/37091307/
2. Memi, E., et al. (2024). Diagnostic and therapeutic use of oral micronized progesterone in endocrinology. Reviews in Endocrine and Metabolic Disorders, 25, 751–772. https://pubmed.ncbi.nlm.nih.gov/38652231/
3. Nolan, B. J., Liang, B., & Cheung, A. S. (2021). Efficacy of micronized progesterone for sleep: A systematic review and meta-analysis of randomized controlled trial data. Journal of Clinical Endocrinology & Metabolism, 106(4), 942–951. https://pubmed.ncbi.nlm.nih.gov/33245776/
4. Ogawa, M., et al. (2025). Changes in sleep quality after hormone replacement therapy with micronized progesterone in Japanese menopausal women: A pilot study. Journal of Menopausal Medicine, 31(1), 45–50. https://pubmed.ncbi.nlm.nih.gov/40347165/
5. Pan, Z., et al. (2022). Different regimens of menopausal hormone therapy for improving sleep quality: A systematic review and meta-analysis. Menopause, 29(5), 627–635. https://pubmed.ncbi.nlm.nih.gov/35102100/
6. Prior, J. C. (2018). Progesterone for treatment of symptomatic menopausal women. Climacteric, 21(4), 358–365. https://pubmed.ncbi.nlm.nih.gov/29962247/
7. Prior, J. C., et al. (2023). Oral micronized progesterone for perimenopausal night sweats and hot flushes: A Phase III Canada-wide randomized placebo-controlled 4-month trial. Scientific Reports, 13, 9082. https://pubmed.ncbi.nlm.nih.gov/37277418/
8. Soderpalm, A. H., et al. (2004). Administration of progesterone produces mild sedative-like effects in men and women. Psychoneuroendocrinology, 29(3), 339–354. https://pubmed.ncbi.nlm.nih.gov/14644065/
9. Troìa, L., et al. (2025). Sleep disturbance and perimenopause: A narrative review. Journal of Clinical Medicine, 14(5), 1479. https://pubmed.ncbi.nlm.nih.gov/40094961/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 9 references cited
