The 2-to-4am wake window is one of the commonly reported sleep complaints during perimenopause and menopause. The prevalence of sleep disorders ranges from 16% to 47% during the perimenopausal phase and increases to 35% to 60% in menopause (Troia et al., 2025). The pattern is distinctive: falling asleep is manageable, but staying asleep past 3am becomes unreliable.
This article covers the cortisol-hormone connection behind that timing — how declining progesterone and estrogen alter the HPA axis in ways that produce a predictable middle-of-the-sleep-period wake. It does not cover the full range of hormonal sleep disruption mechanisms. For an overview of how estrogen, progesterone, and their metabolites affect sleep architecture across the menopause transition, see Hormonal Women Sleep Disruption.
What Causes the 3am Cortisol Spike in Perimenopause?
In a normal cortisol rhythm, cortisol reaches its lowest point (the nadir) around midnight, then begins a gradual pre-dawn rise toward a morning peak between 6am and 8am. This rise is regulated in part by corticotropin-releasing hormone (CRH), which is itself kept in check during sleep by GABA-A receptor activity in the brain.
Progesterone contributes to that GABA-A activity through its metabolite allopregnanolone. Allopregnanolone is a neurosteroid that enhances GABA-A receptor function — GABA-A being the primary inhibitory receptor that dampens excitatory neural activity, including CRH release. When progesterone declines during perimenopause, allopregnanolone declines in parallel. The result is reduced GABA-A inhibition of CRH, and with that, reduced suppression of the HPA axis during sleep (Gordon et al., 2015).
This is the disinhibition model: progesterone decline removes the inhibitory input that keeps cortisol low overnight, allowing cortisol to rise earlier and more steeply than it would in a premenopausal woman.
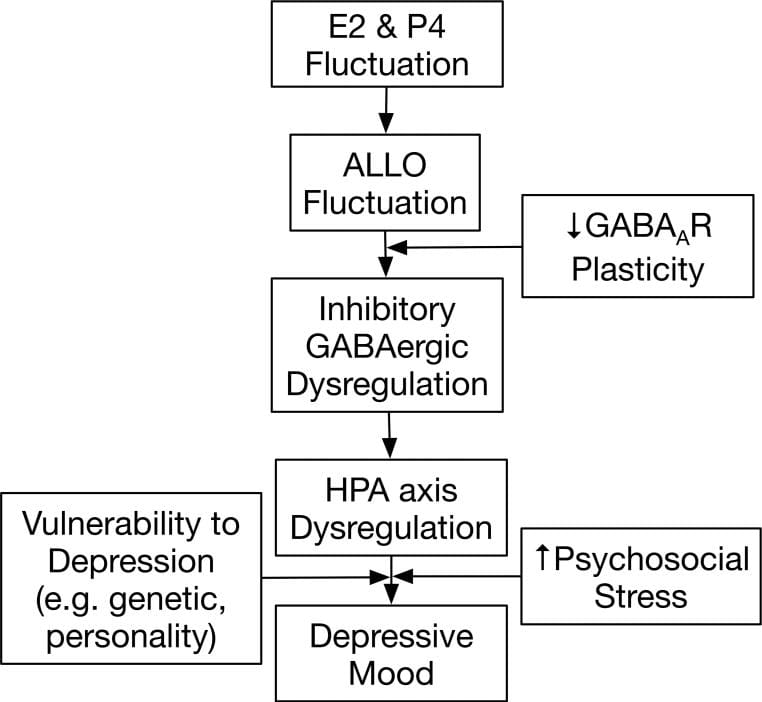
A longitudinal study from the Seattle Midlife Women’s Health Study found that overnight cortisol elevation during the menopause transition was driven by reproductive hormonal changes — not by psychosocial stress (Woods et al., 2009). Women with greater hormonal fluctuation showed higher overnight cortisol independent of perceived stress, life events, or mood. This is an important distinction: the 3am wake has a hormonal substrate, not a psychological one.
More recent data shows that nocturnal wakes are accompanied by measurable cortisol spikes — each arousal event pairs with a pulse of cortisol release (Zhang et al., 2024). In a perimenopausal woman whose HPA axis is already disinhibited, these pulses are larger and more frequent, and each one makes returning to sleep harder.
The reason the wake tends to cluster around 3am is that this is the window where the melatonin-cortisol crossover occurs — melatonin output is declining while cortisol is beginning its pre-dawn rise. When HPA axis regulation is weakened, that crossover happens earlier, and the cortisol rise is steeper. The mismatch can trigger arousal.
Does Menopause Weaken the Body’s Natural Cortisol Regulation?
Two hormonal pathways regulate cortisol during sleep, and both are disrupted during the menopause transition.
The progesterone-allopregnanolone-GABA pathway. Progesterone is metabolized into allopregnanolone, which enhances GABA-A receptor activity. GABA-A receptors are the primary inhibitory mechanism in the central nervous network — they reduce neural excitability, including the excitatory activity that drives CRH release from the hypothalamus. When progesterone declines, allopregnanolone declines with it, and the GABA-A-mediated suppression of the HPA axis weakens (Gordon et al., 2015).
The estrogen-serotonin pathway. Estrogen supports the synthesis and receptor sensitivity of serotonin, a neurotransmitter involved in both mood regulation and sleep-wake transitions. When estrogen declines, serotonin availability drops, and the downstream effects include altered sleep continuity, increased wake propensity, and reduced capacity to sustain sleep through normal arousal events (Fidecicchi et al., 2024).
These two pathways compound each other. Reduced GABA-A-mediated inhibition means cortisol rises more freely during sleep. Reduced serotonergic regulation means the brain is less able to manage the arousal that cortisol produces. The result is a dual vulnerability — both the cortisol rise and the arousal response to it are amplified.
A 2024 review of neuroendocrine mechanisms during the menopause transition identified four disrupted neurotransmitter and neurosteroid pathways: GABA, serotonin, allopregnanolone, and kisspeptin (Fidecicchi et al., 2024). Sleep disorder prevalence ranges from 16% to 47% during the perimenopausal phase and increases to 35% to 60% in menopause (Troia et al., 2025).
For a deeper look at how the allopregnanolone-GABA-A mechanism affects sleep architecture in the menopause transition, see Hormonal Women Sleep Disruption.
Why Does Your Mind Race at 3am During Menopause?
The experience is consistent across the individuals I work with: you are asleep, and then you are not. The mind turns on — thoughts arrive without invitation, heart rate elevates, and the body feels alert. Falling back asleep feels out of reach, not because of worry, but because the body is in a state of activation.
This is cortisol-driven sympathetic arousal. When cortisol spikes during sleep, it activates the sympathetic branch of the autonomic nervous response. Sympathetic activation raises heart rate, increases norepinephrine release (which sharpens alertness and vigilance), and suppresses the parasympathetic tone that would normally keep the body in a resting state. The racing thoughts are a downstream effect of norepinephrine-mediated cortical activation — the brain is responding to a chemical arousal cue, not to an external stimulus or an emotional trigger.
A 2016 study by de Zambotti and colleagues found that perimenopausal women with insomnia showed no recovery of parasympathetic tone during sleep following a stress exposure. Where premenopausal women returned to parasympathetic dominance (lower heart rate, reduced arousal) once asleep, the perimenopausal group remained in sympathetic activation throughout sleep (de Zambotti et al., 2016). The autonomic nervous response stayed in an aroused state — elevated heart rate, reduced heart rate variability — rather than returning to the restorative parasympathetic mode that characterizes normal sleep.
Declining estrogen may also increase amygdala reactivity — the amygdala being the brain region that processes alertness-related stimuli and generates arousal responses. With greater amygdala reactivity, low-level stimuli that would not have caused waking in earlier reproductive stages — a sound, a temperature change, a brief arousal — can trigger a full cortisol-mediated wake event (Fidecicchi et al., 2024).
This distinction matters: the 3am racing mind during perimenopause is not “anxiety” in the psychological sense. It has a hormonal and autonomic basis. The cortisol spike comes first; the mental activation follows.
Does Poor Sleep Raise Cortisol Further?
The relationship between cortisol and sleep is bidirectional. Elevated cortisol fragments sleep, and fragmented sleep elevates cortisol. Once this cycle begins, it can sustain itself independent of the original hormonal trigger.
Cohn and colleagues (2023) demonstrated this in a controlled experimental study that used a pharmacologic menopause model. Participants underwent sleep fragmentation under two estradiol conditions — normal and suppressed. Sleep fragmentation alone raised bedtime cortisol by 27%. It also suppressed the cortisol awakening response (CAR) — the normal morning cortisol surge that supports alertness and daytime function — by 57%. When estradiol was also suppressed (mimicking menopause), bedtime cortisol was elevated further.
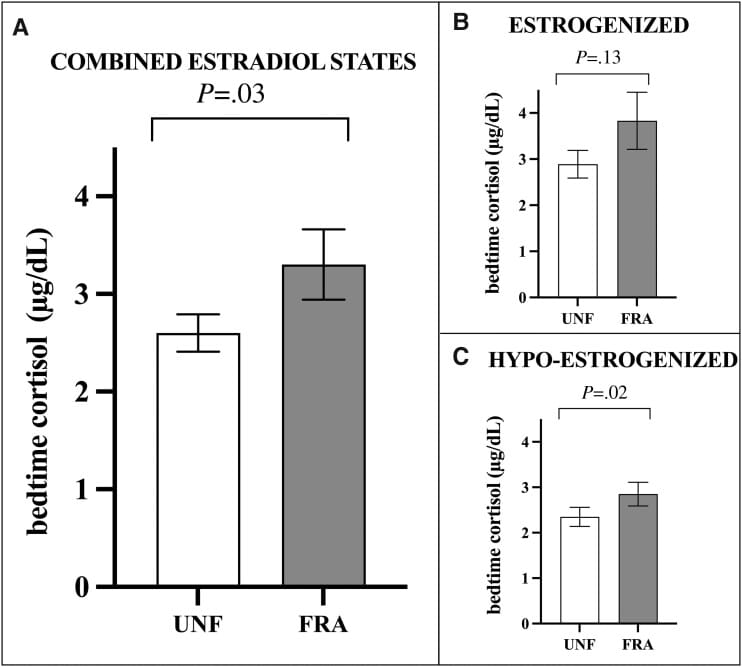
The cortisol curve that results from this pattern is characteristic: elevated at bedtime (producing wakefulness and arousal) and blunted in the morning (producing fatigue and difficulty getting going). This is the pattern many midlife women describe — wired at 3am, exhausted by 8am. For more on this cortisol curve pattern, see the wired-but-tired pattern in menopause.
Sahola and colleagues (2024) found that objective sleep architecture — measured by polysomnography — correlated with cortisol output in menopausal women. Women with less slow-wave sleep (deep sleep) showed higher cortisol levels, reinforcing that sleep quality and cortisol regulation are coupled (Sahola et al., 2024).
The self-reinforcing nature of this cycle is why the 3am wake pattern tends to persist and worsen over time rather than resolve on its own. Each sleep period of fragmented sleep raises the next period’s baseline cortisol, which increases the likelihood of another 3am arousal.
There is evidence that addressing the upstream hormonal cause can interrupt this cycle. In a randomized controlled study, progesterone replacement reduced wake-after-sleep-onset (WASO) by 53% and increased slow-wave sleep by approximately 50% in postmenopausal women (Caufriez et al., 2011). Restoring the missing progesterone — and with it, allopregnanolone and GABA-A-mediated HPA suppression — reduced the cortisol-driven arousals that fragment sleep.
Hormonal changes are one of several causes that can contribute to 3am waking. Cortisol amplification from declining progesterone and estrogen may compound with metabolic, inflammatory, circadian, or autonomic factors. Identifying which causes might be involved is a useful next step.
Find out which causes might be driving your 3am wakeups
Frequently Asked Questions
Is the 3am Wake-Up the Earliest Perimenopause Sleep Change?
Progesterone typically begins declining before estrogen does in the early stages of perimenopause. Because allopregnanolone — the progesterone metabolite responsible for enhancing GABA-A receptor activity during sleep — tracks with progesterone levels, sleep disruption from HPA disinhibition can begin before other commonly recognized perimenopausal changes appear.
Sleep disruption often precedes vasomotor changes (hot flashes and night sweats) by months or years (Troia et al., 2025). A woman in early perimenopause may not yet have irregular cycles or noticeable hot flashes but may already be waking at 3am due to reduced allopregnanolone-mediated cortisol suppression during sleep.
Does Cortisol Spike After a Hot Flash?
The vasomotor and HPA pathways interact. A hot flash activates sympathetic nervous arousal — increasing heart rate, skin temperature, and cortisol release. At the same time, elevated cortisol and the neuroendocrine disruptions of the menopause transition may make the thermoregulatory center in the hypothalamus more reactive, reducing the threshold at which a hot flash is triggered (Fidecicchi et al., 2024).
In practice, this means that a 3am cortisol spike and a 3am hot flash can feed each other. For more on how night sweats interact with sleep architecture, see how night sweats affect deep sleep.
Does Blood Sugar Affect 3am Waking?
When blood glucose falls below a certain threshold during sleep, the body releases cortisol and epinephrine to mobilize stored glucose. This counter-regulatory response produces sympathetic activation — elevated heart rate, alertness, and wakefulness — similar to the HPA-driven 3am wake described in this article.
Hormonal changes during perimenopause can alter insulin sensitivity and glucose metabolism, which may increase the likelihood of overnight blood sugar instability. This is a separate mechanism from the progesterone-cortisol pathway, but it can compound it. For a full explanation of the blood sugar mechanism, see how a blood sugar drop can wake you at 3am.
Related Reading
- Hormonal Sleep Disruption in Women: What It Is, How It Fragments Sleep, and How to Recognize It — Full overview of how estrogen, progesterone, and their metabolites affect sleep architecture
- Why Doesn’t Melatonin Work for Menopause Insomnia? — The estrogen-serotonin-melatonin chain and why supplemental melatonin misses the upstream hormonal cause
- What Causes the ‘Wired but Tired’ Feeling in Menopause? — HPA axis dysregulation and hyperarousal that leave you exhausted but unable to sleep
- Why Can’t You Sleep Before Your Period? — How the progesterone crash in the late luteal phase fragments sleep and raises core body temperature
- Does Hormone Replacement Therapy Help with Sleep in Menopause? — What the clinical evidence shows about HRT’s effect on sleep architecture and hot-flash-driven waking
- Does Progesterone Help You Sleep? — The GABA-A receptor mechanism behind progesterone’s sleep-promoting effects and why dose timing matters
- Does Low Progesterone Cause Insomnia in Perimenopause? — Sleep maintenance impairment as the characteristic complaint of progesterone decline (publishing soon)
- Can Night Sweats Disrupt Your Deep Sleep? — The thermoregulatory pathway from estrogen withdrawal to sleep fragmentation (publishing soon)
- Does Menopause Reduce Your Deep Sleep? — Estrogen-dependent changes in slow-wave and REM sleep architecture (publishing soon)
- Does PMDD Cause Insomnia? — Cyclical sleep disruption from hormone sensitivity (publishing soon)
- Does Menopause Cause Sleep Apnea in Women? — The underdiagnosed connection between hormone decline and airway changes (publishing soon)
- Does FSH Affect Your Sleep During Menopause? — Emerging research on FSH as an active sleep disruptor (publishing soon)
References
1. Caufriez A, Leproult R, L’Hermite-Baleriaux M, Kerkhofs M, Copinschi G. (2011). Progesterone prevents sleep disturbances and modulates GH, TSH, and melatonin secretion in postmenopausal women. Journal of Clinical Endocrinology & Metabolism, 96(4):E614-E623. https://pubmed.ncbi.nlm.nih.gov/21289261/
2. Cohn AY, Grant LK, Nathan MD, Wiley A, Abramson M, Harder JA, Crawford S, Klerman EB, Scheer FAJ, Kaiser UB, Rahman SA, Joffe H. (2023). Effects of sleep fragmentation and estradiol decline on cortisol in a human experimental model of menopause. Journal of Clinical Endocrinology & Metabolism, 108(11). https://pmc.ncbi.nlm.nih.gov/articles/PMC10584010/
3. de Zambotti M, Sugarbaker D, Trinder J, Colrain IM, Baker FC. (2016). Acute stress alters autonomic modulation during sleep in women approaching menopause. Psychoneuroendocrinology, 66:1-10. https://pubmed.ncbi.nlm.nih.gov/26766119/
4. Fidecicchi T, Giannini A, Chedraui P, Luisi S, Battipaglia C, Genazzani AR, Genazzani AD, Simoncini T. (2024). Neuroendocrine mechanisms of mood disorders during menopause transition: A narrative review and future perspectives. Maturitas, 188:108099. https://pubmed.ncbi.nlm.nih.gov/39111089/
5. Gordon JL, Girdler SS, Meltzer-Brody SE, Stika CS, Thurston RC, Clark CT, Prairie BA, Moses-Kolko E, Joffe H, Wisner KL. (2015). Ovarian hormone fluctuation, neurosteroids, and HPA axis dysregulation in perimenopausal depression: A novel heuristic model. American Journal of Psychiatry, 172(3):227-236. https://pmc.ncbi.nlm.nih.gov/articles/PMC4513660/
6. Sahola N, Toffol E, Kalleinen N, Polo-Kantola P. (2024). Worse sleep architecture but not self-reported insomnia and sleepiness is associated with higher cortisol levels in menopausal women. Maturitas, 187:108057. https://pubmed.ncbi.nlm.nih.gov/38909441/
7. Troia L, Garassino M, Volpicelli AI, Fornara A, Libretti A, Surico D, Remorgida V. (2025). Sleep disturbance and perimenopause: A narrative review. Journal of Clinical Medicine, 14(5). https://pmc.ncbi.nlm.nih.gov/articles/PMC11901009/
8. Woods NF, Mitchell ES, Smith-DiJulio K. (2009). Cortisol levels during the menopausal transition and early postmenopause: Observations from the Seattle Midlife Women’s Health Study. Menopause, 16(4):708-718. https://pmc.ncbi.nlm.nih.gov/articles/PMC2749064/
9. Zhang XX, et al. (2024). Changed nocturnal levels of stress-related hormones couple with sleep-wake states in the patients with chronic insomnia disorder: A clinical pilot study. Sleep Medicine, 117:177-183. https://pubmed.ncbi.nlm.nih.gov/38554533/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 9 references cited
