Overview: Estrogen, progesterone, and their downstream metabolites regulate multiple pathways that govern sleep — from the brain’s inhibitory tone to body temperature to the neurotransmitters that set the sleep-wake cycle. As these hormones’ function is disrupted during perimenopause and menopause, each pathway is affected in a specific way:
- Progesterone is converted to allopregnanolone, a neurosteroid that activates GABA-A receptors — the same receptor class targeted by sleep medications. Disruption in progesterone function removes this natural sedation pathway.
- Estrogen modulates thermoregulation through hypothalamic KNDy neurons. Disruption in estrogen function narrows the thermoneutral zone, triggering hot flashes and night sweats that fragment sleep.
- Estrogen also modulates serotonin metabolism. When estrogen function is disrupted, serotonin may fall, impairing melatonin synthesis and degrading both slow-wave and REM sleep.
- Estrogen modulates antidiuretic hormone signaling. Disruption in estrogen function increases nocturnal urine production, driving nighttime awakenings to urinate.
- Follicle-stimulating hormone rises sharply during the menopausal transition and may independently affect body composition, cognition, and nighttime awakenings.
- Loss of estrogen and progesterone buffering amplifies HPA axis stress reactivity, producing cortisol-driven early-morning waking.
The result: up to 60% of women during perimenopause and menopause report sleep disruption.
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 29 references cited
What Are the Hormonal Changes in Aging Women?
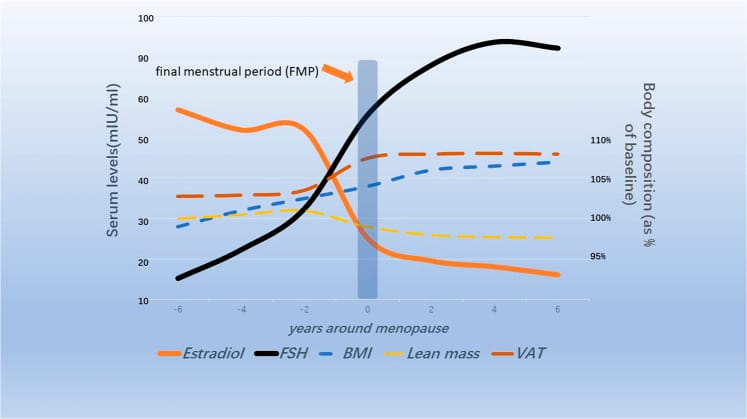
Perimenopause begins 4-8 years before a woman’s final menstrual period. Menopause is defined as 12 consecutive months without a period. Postmenopause is everything after.
During perimenopause, estrogen does not follow a smooth downward curve. Instead, estrogen function fluctuates erratically — sometimes spiking higher than premenopausal levels, sometimes dropping below — before declining permanently. A 2022 systematic review (Haufe et al.) found that hormonal fluctuations were associated with sleep problems during this transition.
Progesterone function changes earlier and more consistently. Progesterone is produced by the corpus luteum after ovulation. As ovulation becomes irregular in perimenopause, progesterone production drops before estrogen does. Progesterone’s roles extend beyond reproduction: it is the precursor to allopregnanolone, a neurosteroid involved in mood regulation and neural inhibition (covered in detail in the mechanisms section below).
Follicle-stimulating hormone begins rising approximately 6 years before the final menstrual period, accelerates rapidly in the 2 years before menopause, and stabilizes roughly 2 years after (Spicer et al., 2025). FSH reaches levels approximately 14 times higher than in men. Emerging research investigates whether FSH has independent metabolic and neural effects beyond marking ovarian reserve depletion.
Three dimensions of hormonal function change simultaneously during this transition. Production declines as ovarian follicles are depleted. Bioavailability changes as sex hormone-binding globulin fluctuates and aromatase activity changes. And receptor sensitivity adjusts in response to the changing hormonal environment. A woman can have lab values within the “normal” reference range while her functional hormonal status has changed in ways that affect multiple downstream pathways.
The well-established consequences of these changes include increased cardiovascular risk, declining bone density, metabolic changes toward central fat deposition, and emerging cognitive vulnerability (Haufe & Leeners, 2023). Up to 60% of women in perimenopause and menopause report sleep disruption (Troia et al., 2025), and perimenopausal women may be up to 1.6 times more likely to experience sleep problems compared to premenopausal women.
But these hormonal changes don’t only affect cardiovascular and metabolic health. They also disrupt sleep — through at least six distinct mechanisms.
How Does Disruption in Hormonal Function Disrupt Sleep in Women?
Does Rising Follicle-Stimulating Hormone Contribute to Nighttime Awakenings?
FSH’s trajectory across the menopausal transition is well-mapped: it begins rising approximately 6 years before the final menstrual period, accelerates rapidly in the 2 years before menopause, and stabilizes roughly 2 years after (Spicer et al., 2025).
A 2022 study (Coborn et al.) examining reproductive hormone profiles in perimenopausal women found that higher FSH was associated with more frequent nighttime awakenings. The women in the study had a median of 1.5 awakenings per night with approximately 24 minutes of wakefulness after sleep onset.
The question is whether FSH contributes to these awakenings independently — or only as a marker of broader hormonal change. A 2022 review in Frontiers in Physiology (Mao et al.) found that exogenous FSH causes adipose tissue growth and proliferation, while blocking the FSH receptor pathway reduces adipose accumulation. FSH-driven central fat deposition is itself an independent risk factor for sleep disruption through airway collapsibility and increased inflammation.
A 2025 review in Psychiatry Research (Spicer et al.) argued that FSH should be considered more than a marker for menopause — characterizing it as a “frontier for women’s mental health” with potential independent effects on psychiatric vulnerability and cognitive function.
FSH’s direct effects on sleep are less established than the other mechanisms described in this article. The evidence is largely associational. The mechanistic pathway from FSH to sleep fragmentation may work through body composition changes, inflammation, or neural targets that have not yet been fully characterized.
How Does Disruption in Ovarian Hormone Function Amplify Stress Reactivity?
The HPA axis governs how the body responds to stress. Under normal conditions, estrogen and progesterone modulate HPA sensitivity — helping to keep cortisol responses proportional to the stressor. When both hormones’ function is disrupted during perimenopause and menopause, that buffering is reduced.
A 2022 study published in Sleep (Kreitzberg et al.) collected at-home cortisol data from 3,966 individuals — 71% female, with a mean age of 42.8 years. About one in five showed a flattened diurnal cortisol slope, a marker of HPA axis dysregulation. Among the cohort, 145 individuals had bedtime cortisol above the reference range — a pattern directly tied to difficulty falling asleep and staying asleep.
A 2008 randomized trial (Hachul et al.) demonstrated the sleep benefits of restoring hormonal function: combined estrogen-progesterone therapy eliminated nighttime hot flashes (0% prevalence vs 14.2% in the estrogen-only group), reduced arousals from sleep, decreased breathing irregularities, and reduced anxiety.
Body composition changes during menopause amplify HPA stress reactivity. A 2024 study (Gonsalves et al.) found that fat-free mass index — a proxy for muscle mass — showed negative correlations with cortisol response at 15, 30, and 60 minutes post-stimulation (rho = -0.248 to -0.364). Since menopause changes body composition toward higher fat mass and lower lean mass, this amplifies cortisol output in response to stressors.
Chronic HPA dysregulation produces glucocorticoid receptor resistance (Nunez et al., 2025) — the body loses the ability to properly suppress cortisol during sleep, creating a paradoxical pattern where cortisol stays elevated even when it should be at its nadir.
This mechanism overlaps with the cortisol/HPA axis described in the autonomic cause page, but the trigger here is disruption in ovarian hormone function rather than chronic external stress.
How Does Disruption in Progesterone Function Contribute to 3am Wakeups?
Progesterone is produced by the corpus luteum after ovulation. As ovulation becomes irregular in perimenopause, progesterone production drops before estrogen does. Unlike estrogen — which fluctuates erratically during perimenopause — progesterone’s functional disruption tends to be more consistent and earlier in onset.
The sleep relevance of progesterone is not through the hormone itself, but through its metabolite. Progesterone is converted to allopregnanolone via the enzyme 5-alpha-reductase. A 1995 study (Bitran et al.) demonstrated that progesterone’s sedative and anxiolytic effects are not genomic — they work through this conversion to allopregnanolone, which then modulates GABA-A receptors. Blocking 5-alpha-reductase eliminated both the behavioral effect and the rise in circulating allopregnanolone.
Allopregnanolone acts on both synaptic and extrasynaptic GABA-A receptors. At synaptic receptors (gamma2-containing), it produces phasic inhibition — short bursts of neural calming. At extrasynaptic receptors (delta-containing), it produces tonic inhibition — a steady background current that calms whole neural networks. This dual action is what makes allopregnanolone more effective at supporting natural sleep architecture than medications that target only the synaptic sites.
A 2023 cryo-electron microscopy study (Sun et al., preprint) resolved native murine GABA-A receptor structures and found that endogenous allopregnanolone was bound to the receptor even without external administration — suggesting that this neurosteroid is part of the brain’s baseline inhibitory architecture, not just a transient modulator.
The balance matters. A 2022 review (Wang et al.) showed that allopregnanolone (the 3-alpha-hydroxyl epimer) and epiallopregnanolone (the 3-beta-hydroxyl epimer) produce opposite effects on GABA-A receptors — positive versus negative allosteric modulation — at overlapping binding sites. The net inhibitory tone depends on the ratio of both epimers, which changes when progesterone production changes.
Receptor sensitivity also adapts. A 2015 study (Hedstrom et al.) found that women with polycystic ovary syndrome — who have chronically elevated allopregnanolone — showed blunted GABA-A receptor sensitivity to exogenous allopregnanolone. This has implications for perimenopause: receptors that adapted to decades of cycling neurosteroid levels face a permanent change in exposure, and the mismatch between receptor sensitivity and available allopregnanolone can produce a period of heightened vulnerability to 3am waking.
A 2023 review in the Harvard Review of Psychiatry (Susser) identified allopregnanolone as central to reproductive depression — a condition distinct from major depressive disorder, driven by hormone-level fluctuations in susceptible women. The same progesterone-to-allopregnanolone-to-GABA-A pathway is implicated in premenstrual dysphoric disorder, postpartum depression, and perimenopausal mood and sleep disruption.
How Does Disruption in Estrogen Function Trigger Night Sweats That Fragment Sleep?
KNDy neurons in the arcuate nucleus of the hypothalamus are estrogen-sensitive. Under normal estrogen function, estrogen holds these neurons in check through negative feedback. When estrogen function is disrupted, neurokinin B signaling becomes overactive.
A 2012 study in PNAS (Mittelman-Smith et al.) provided direct evidence: ablation of KNDy neurons in rats blocked estrogen-withdrawal-induced cutaneous vasodilation. Without KNDy neurons, the hot flash response did not occur. A companion study by the same group (Endocrinology, Mittelman-Smith et al., 2012) found that KNDy ablation also prevented the rise in luteinizing hormone that normally follows estrogen withdrawal — demonstrating that the same neurons driving hot flashes also drive the LH surge.
A 2025 mouse model study (Torres Jimenez et al.) used fiber photometry to directly measure KNDy neuron activity. After ovariectomy, KNDy neurons exhibited higher-frequency synchronous firing events, and each event correlated with acute temperature fluctuations — rapid skin temperature elevations with simultaneous core temperature reductions — closely mimicking the hot flashes and night sweats experienced by menopausal women.
The timing of vasomotor events determines their sleep impact. A 2025 ecological momentary assessment study (Law et al., N=99 perimenopausal and early postmenopausal women) found that nocturnal hot flash frequency was strongly associated with worse sleep quality (Est=-0.46, p<0.001), with nocturnal timing showing a larger and more significant effect than daytime hot flashes. Evening hot flash severity predicted longer sleep onset latency that night.
A 2024 systematic review (Cieri-Hutcherson et al.) of NK3R antagonist trials — fezolinetant and elinzanetant — found that these drugs, which target the neurokinin B signaling arm of KNDy neurons, reduced vasomotor symptom frequency and severity and improved sleep quality. This validates the KNDy/NK3R pathway as both the mechanistic explanation and the therapeutic target.
How Does Disruption in Estrogen Function Cause Light, Unrefreshing Sleep?
A 2025 review in Frontiers in Molecular Biosciences (Mervosh & Devi) established estrogen’s broad neuroprotective role — including effects on synaptic plasticity, mitochondrial function, and cerebrovascular integrity — all of which are disrupted at menopause. Estrogen modulates serotonin pathways. When estrogen function is disrupted, serotonin degradation may accelerate and norepinephrine levels may rise, moving the arousal balance toward wakefulness.
The serotonin-melatonin connection is direct. Serotonin is converted to melatonin through a two-step enzymatic process (tryptophan -> serotonin -> melatonin). Lower serotonin availability means less substrate for melatonin production, which can delay sleep onset and degrade sleep architecture.
A 2024 study in the Korsakov Journal of Neurology and Psychiatry (Ukraintseva & Saltykov) experimentally fragmented both slow-wave sleep and REM sleep by approximately 50% each. REM sleep fragmentation produced a pronounced melatonin elevation — 1.8 times higher than in the control condition — driven specifically by serotonergic activation, not autonomic changes. This serotonin-melatonin coupling means that when estrogen-related serotonin reduction impairs melatonin regulation, the downstream effect on sleep architecture is measurable.
Polysomnographic data shows the architectural impact. A 2008 study (Kalleinen et al.) compared premenopausal, postmenopausal, and young women. Postmenopausal women had a sleep efficiency of 80.2% compared to 93.4% in young women, with less slow-wave sleep across the board. Premenopausal midlife women already showed slow-wave sleep reduction, suggesting that sleep architecture degradation begins before menopause completes.
A 2007 study (Backhaus et al.) found that middle-aged adults had approximately 70% less slow-wave sleep than young adults (18.4 minutes vs 62.3 minutes during early nocturnal sleep), and this reduction correlated directly with worse declarative memory consolidation. This midlife slow-wave sleep reduction overlaps with perimenopause timing in women.
The norepinephrine component compounds the problem. Disruption in estrogen function may increase norepinephrine levels, raising arousal thresholds during sleep. Combined with reduced serotonin and impaired melatonin production, the result is the light, easily fragmented, unrefreshing sleep that many women in the menopausal transition describe.
How Does Disruption in Estrogen Function Increase Nighttime Urination?
Estrogen receptors (ER-alpha and ER-beta) are expressed in brain regions that regulate vasopressin release — the organum vasculosum of the lamina terminalis, the subfornical organ, and the median preoptic nucleus. These regions connect with the hypothalamic nuclei that produce antidiuretic hormone. When estrogen function is disrupted, antidiuretic hormone secretion loses its normal day-night rhythm, and the kidneys produce excess urine during the sleep period — a condition called nocturnal polyuria.
A 2021 pilot trial (Pauwaert et al.) of 245 postmenopausal women found that hormone replacement therapy reduced nocturia prevalence from 27.7% to 16.4% among women with 2 or more nocturnal voids per night. Combined estrogen-progesterone therapy and tissue-selective estrogen complex both produced reductions in nocturnal voiding frequency (p=0.018 for each). The mechanism ran primarily through sleep improvement rather than bladder effects — directly implicating disruption in estrogen function, rather than bladder capacity, as the driver.
A 2015 study (Mun et al.) tested desmopressin — a synthetic vasopressin analogue — in elderly women with nocturnal polyuria. Nocturia episodes dropped from 3.63 to 2.00 per night (p=0.01), nocturnal urine volume decreased, and sleep quality improved across multiple domains (sleep index p=0.003, sleep disturbance p=0.001). This demonstrates that insufficient endogenous vasopressin activity is a core mechanism in nocturnal polyuria in postmenopausal women.
A 2024 review (Chang & Kuo) established that nocturia involves three overlapping mechanisms: nocturnal frequency, nocturnal polyuria, and sleep disruption itself. The relationship is bidirectional — poor sleep worsens nocturia, and nocturia worsens sleep.
Disruption in estrogen function drives nocturia through two pathways: free-water diuresis from impaired antidiuretic hormone secretion, and salt-predominant diuresis from diminished activation of the renin-angiotensin-aldosterone axis. Both converge on the same outcome — excess urine production during the hours when the body should be producing its least.
What Triggers or Worsens Hormonal Sleep Disruption in Women?
Does Alcohol Worsen Hormonal Sleep Disruption in Women?
Alcohol enhances GABA-A receptor activity acutely, producing the sedative effect that makes it easier to fall asleep. But as the body metabolizes alcohol during sleep, a withdrawal effect fragments sleep in the second half — the same hours when cortisol is beginning its pre-waking rise and when women with disrupted hormonal function are already at their highest vulnerability.
Alcohol also increases aromatase activity, the enzyme that converts androgens to estrogen, disrupting the balance of circulating estrogen metabolites. And alcohol impairs the thermoregulatory responses that are already destabilized by KNDy neuron hyperactivation — compounding the temperature swings that trigger night sweats.
How Does Gut Health Affect Hormonal Sleep Disruption in Women?
Estrobolome bacteria produce beta-glucuronidase, an enzyme that deconjugates estrogen and returns it to circulation in its active form. When gut dysbiosis reduces estrobolome function, less estrogen is recycled — amplifying all estrogen-dependent sleep pathways: thermoregulation via KNDy neurons, serotonin-melatonin production, antidiuretic hormone signaling, and HPA axis buffering.
The relationship is bidirectional: sleep disruption itself alters gut microbiome composition, potentially creating a self-reinforcing cycle where poor sleep degrades estrobolome function, which further reduces bioavailable estrogen, which further disrupts sleep.
How Do Body Composition Changes Affect Sleep in Menopausal Women?
A 2024 study (Gonsalves et al.) found that fat-free mass index showed negative correlations with cortisol output — individuals with lower lean mass produced more cortisol in response to stimulation. Since menopause moves body composition toward higher fat and lower lean mass, this amplifies HPA stress reactivity at the same time that hormonal HPA buffering is being reduced.
FSH may contribute directly to these body composition changes. Mao et al. (2022) found that FSH promotes adipose tissue growth and proliferation. And Mittelman-Smith et al. (2012) showed that KNDy neuron ablation prevented the weight gain normally caused by ovariectomy — suggesting that body composition changes during menopause are partly driven by the same neuronal population causing hot flashes.
How Does Chronic Stress Compound Hormonal Sleep Disruption?
A 2000 study (Bergendahl et al.) demonstrated that even in sex steroid-replete midluteal-phase women, fasting stress amplified cortisol secretion by 1.7-fold and disrupted the coupling between cortisol and other hormonal axes (LH, GH, leptin). The HPA axis is fully stress-responsive regardless of hormonal status. In menopause, the buffering that normally modulates that response is reduced — meaning the same stressor produces a larger and longer-lasting cortisol elevation.
Can Medications Interfere with Hormonal Sleep Pathways?
SSRIs affect serotonin reuptake, which can alter the serotonin-to-melatonin conversion pathway that is already under pressure from disrupted estrogen function. Medications that inhibit 5-alpha-reductase — the enzyme that converts progesterone to allopregnanolone — can compound the GABA-A receptor sedation loss that perimenopause is already producing. Anticholinergic medications (covered in detail in the autonomic cause page) weaken the same cholinergic and parasympathetic pathways that hormonal changes are affecting.
How Do You Know If Disruption in Hormonal Function Is Affecting Your Sleep as a Woman?
Night sweats and hot flashes are the easiest-to-recognize indicators, but they are absent in many women with hormonal sleep disruption. The other five mechanisms described in this article can fragment sleep without producing overt temperature events.
Second-half-of-night waking — particularly between 2am and 4am — is characteristic of both HPA stress reactivity amplification (cortisol-driven waking) and progesterone/GABA-A disruption (reduced tonic inhibition in the second half of the sleep period).
Light, unrefreshing sleep — even with adequate hours in bed, mornings feel unrestored. This points to the serotonin-melatonin pathway and degraded sleep architecture: reduced slow-wave sleep and reduced REM sleep.
Heightened stress reactivity — everyday stressors feel more activating than they used to, and sleep is more easily disrupted by worry or minor events that previously would not have caused waking. This reflects reduced HPA axis buffering from disrupted ovarian hormone function.
Nocturia — waking to urinate two or more times per night, particularly if this started or worsened with perimenopause. This points to disrupted estrogen modulation of antidiuretic hormone signaling.
Timeline correlation is a particularly informative indicator: sleep disruption that started or worsened in the mid-40s to early 50s, correlating with menstrual changes — irregular periods, heavier or lighter flow, skipped cycles, or the cessation of periods.
Relevant biomarkers include FSH, estradiol, and progesterone levels — with the caveat that “normal” reference ranges do not capture functional status — as well as cortisol patterns and nocturnal polyuria index.
Hormonal changes often overlap with other causes of sleep disruption — autonomic dysregulation, inflammatory load, metabolic changes. Many women in midlife have 2-3 causes interacting, and addressing only one may not resolve the full picture.
Find out which causes are driving your 3am wakeups
Frequently Asked Questions
Is Hormonal Sleep Disruption Permanent After Menopause?
The KNDy neuron hyperactivation that drives hot flashes and night sweats tends to attenuate over time as the hypothalamus adapts to lower estrogen levels. For many women, vasomotor events become less frequent and less severe in the years following menopause.
The progesterone/allopregnanolone pathway and the serotonin-melatonin cascade, however, are not solely tied to the acute transition. Their effects may persist because the hormonal changes that produced them are permanent. The slow-wave sleep reductions measured in midlife (Backhaus et al., 2007; Kalleinen et al., 2008) reflect both hormonal and aging contributions and may not fully reverse.
Does Hormone Therapy Improve Sleep?
Oral micronized progesterone may have particular sleep benefits because the oral route undergoes first-pass hepatic metabolism, which increases conversion to allopregnanolone — the neurosteroid that produces tonic inhibition at GABA-A receptors. This is one reason why the form and route of progesterone administration may matter for sleep outcomes.
Can You Have Hormonal Sleep Disruption Without Hot Flashes?
The KNDy/thermoregulatory pathway is one of six mechanisms described in this article. The other five work independently of temperature regulation and can produce fragmented, unrefreshing sleep without any obvious external indicators.
Why Does Sleep Disruption Sometimes Start Years Before Menopause?
A woman can be in perimenopause — with progesterone production declining and FSH rising — while still having monthly periods. The sleep disruption can precede the menstrual irregularity that many women use as their marker for “starting menopause.”
Do Men and Women Experience Hormonal Sleep Disruption Differently?
Related Reading
- Why Do You Wake Up at 3am During Menopause?
- Can Insomnia Be the First Sign of Perimenopause — Even Without Hot Flashes?
- Why Did You Become a Light Sleeper in Perimenopause?
- Autonomic Sleep Disruption
- Circadian Sleep Disruption
- Hormonal Sleep Disruption in Men
- Inflammatory Sleep Disruption
- Metabolic Sleep Disruption
References
Epidemiology and Hormonal Changes
- Troia, L., Garassino, M., Volpicelli, A. I., Fornara, A., Libretti, A., Surico, D., & Remorgida, V. (2025). Sleep disturbance and perimenopause: A narrative review. Journal of Clinical Medicine, 14(5), 1479. https://pubmed.ncbi.nlm.nih.gov/40094961/
- Haufe, A., & Leeners, B. (2023). Sleep disturbances across a woman’s lifespan: What is the role of reproductive hormones? Journal of the Endocrine Society, 7(5), bvad036. https://pubmed.ncbi.nlm.nih.gov/37091307/
- Haufe, A., Baker, F. C., & Leeners, B. (2022). The role of ovarian hormones in the pathophysiology of perimenopausal sleep disturbances: A systematic review. Sleep Medicine Reviews, 66, 101710. https://pubmed.ncbi.nlm.nih.gov/36356400/
Follicle-Stimulating Hormone and Sleep
- Coborn, J., de Wit, A., Crawford, S., Nathan, M., Rahman, S., Finkelstein, L., Wiley, A., & Joffe, H. (2022). Disruption of sleep continuity during the perimenopause: Associations with female reproductive hormone profiles. The Journal of Clinical Endocrinology and Metabolism, 107(10), e4144-e4153. https://pubmed.ncbi.nlm.nih.gov/35878624/
- Spicer, J., Malaspina, D., Blank, S. V., & Goosens, K. A. (2025). Follicle-stimulating hormone: More than a marker for menopause: FSH as a frontier for women’s mental health. Psychiatry Research, 345, 116239. https://pubmed.ncbi.nlm.nih.gov/39892305/
- Mao, L., Wang, L., Bennett, S., Xu, J., & Zou, J. (2022). Effects of follicle-stimulating hormone on fat metabolism and cognitive impairment in women during menopause. Frontiers in Physiology, 13, 1043237. https://pubmed.ncbi.nlm.nih.gov/36545281/
HPA Axis Stress Reactivity
- Kreitzberg, D., Gavin, K., Daumeyer, N., Seixas, A., & Bauer, T. (2022). At-home sample collection for identification of alterations in HPA-axis activity and cortisol area under the curve [Conference abstract]. Sleep, 45(Suppl 1), A170. https://doi.org/10.1093/sleep/zsac079.376
- Hachul, H., Bittencourt, L. R. A., Andersen, M. L., Haidar, M. A., Baracat, E. C., & Tufik, S. (2008). Effects of hormone therapy with estrogen and/or progesterone on sleep pattern in postmenopausal women. International Journal of Gynaecology and Obstetrics, 103(3), 207-212. https://pubmed.ncbi.nlm.nih.gov/18812241/
- Gonsalves, S. G., Ross, A., Steele, M. E., & Saligan, L. N. (2024). Differential impacts of body composition indices on cortisol response and HPA axis activity [Conference abstract]. Journal of the Endocrine Society, 8(Suppl 1), bvae163.168. https://doi.org/10.1210/jendso/bvae163.168
- Nunez, S. G., Rabelo, S. P., Subotic, N., Caruso, J. W., & Knezevic, N. N. (2025). Chronic stress and autoimmunity: The role of HPA axis and cortisol dysregulation. International Journal of Molecular Sciences, 26(20), 9994. https://pubmed.ncbi.nlm.nih.gov/41155288/
Progesterone, Allopregnanolone, and GABA-A Modulation
- Bitran, D., Shiekh, M., & McLeod, M. (1995). Anxiolytic effect of progesterone is mediated by the neurosteroid allopregnanolone at brain GABA(A) receptors. Journal of Neuroendocrinology, 7(3), 171-177. https://pubmed.ncbi.nlm.nih.gov/7606242/
- Susser, L. C. (2023). Clinical implications of the neurosteroid allopregnanolone in reproductive depression. Harvard Review of Psychiatry, 31(1), 37-45. https://pubmed.ncbi.nlm.nih.gov/36608082/
- Sun, C., Zhu, H., Clark, S., & Gouaux, E. (2023). Regulated assembly and neurosteroid modulation constrain GABA-A receptor pharmacology in vivo [Preprint]. bioRxiv. https://pubmed.ncbi.nlm.nih.gov/36824901/
- Hedstrom, H., Backstrom, T., Bixo, M., Nyberg, S., Wang, M., Gideonsson, I., & Turkmen, S. (2015). Women with polycystic ovary syndrome have elevated serum concentrations of and altered GABA(A) receptor sensitivity to allopregnanolone. Clinical Endocrinology, 83(5), 643-650. https://pubmed.ncbi.nlm.nih.gov/25929428/
- Wang, L., Covey, D. F., Akk, G., & Evers, A. S. (2022). Neurosteroid modulation of GABAA receptor function by independent action at multiple specific binding sites. Current Neuropharmacology, 20(5), 886-890. https://pubmed.ncbi.nlm.nih.gov/34856904/
KNDy Neurons and Thermoregulatory Disruption
- Mittelman-Smith, M. A., Williams, H., Krajewski-Hall, S. J., McMullen, N. T., & Rance, N. E. (2012). Role for kisspeptin/neurokinin B/dynorphin (KNDy) neurons in cutaneous vasodilatation and the estrogen modulation of body temperature. Proceedings of the National Academy of Sciences, 109(48), 19846-19851. https://pubmed.ncbi.nlm.nih.gov/23150555/
- Mittelman-Smith, M. A., Williams, H., Krajewski-Hall, S. J., Lai, J., Ciofi, P., McMullen, N. T., & Rance, N. E. (2012). Arcuate kisspeptin/neurokinin B/dynorphin (KNDy) neurons mediate the estrogen suppression of gonadotropin secretion and body weight. Endocrinology, 153(6), 2800-2812. https://pubmed.ncbi.nlm.nih.gov/22508514/
- Torres Jimenez, E., Voliotis, M., Wall, E. G., Lopez, I., Zdon, S. T., Zhuo, J., & Navarro, V. M. (2025). Modeling body temperature rhythms and vasomotor symptoms linked to KNDy neurons in a mouse model of menopause [Conference abstract]. Journal of the Endocrine Society, 9(Suppl 1), bvaf149.1613. https://doi.org/10.1210/jendso/bvaf149.1613
- Law, C., Gunthert, K., Sokolovsky, A. W., & Behar, E. (2025). Subjective menopausal hot flashes in sleep disturbance: Does the timing of hot flashes matter? Menopause, advance online publication. https://pubmed.ncbi.nlm.nih.gov/41401251/
- Cieri-Hutcherson, N. E., Marji, E. K., & Hutcherson, T. C. (2024). Systematic review of neurokinin-3 receptor antagonists for the management of vasomotor symptoms of menopause. Menopause, 31(4), 342-354. https://pubmed.ncbi.nlm.nih.gov/38471077/
Serotonin, Melatonin, and Sleep Architecture
- Kalleinen, N., Polo-Kantola, P., Himanen, S.-L., Alhola, P., Joutsen, A., Urrila, A. S., & Polo, O. (2008). Sleep and the menopause — Do postmenopausal women experience worse sleep than premenopausal women? Menopause International, 14(3), 97-104. https://pubmed.ncbi.nlm.nih.gov/18714074/
- Ukraintseva, Yu. V., & Saltykov, K. A. (2024). Effects of slow-wave sleep fragmentation and rapid eye movement sleep fragmentation on melatonin secretion. S.S. Korsakov Journal of Neurology and Psychiatry, 124(5 Vyp. 2), 26-32. https://pubmed.ncbi.nlm.nih.gov/38934663/
- Backhaus, J., Born, J., Hoeckesfeld, R., Fokuhl, S., Hohagen, F., & Junghanns, K. (2007). Midlife decline in declarative memory consolidation is correlated with a decline in slow wave sleep. Learning and Memory, 14(5), 336-341. https://pubmed.ncbi.nlm.nih.gov/17522024/
- Mervosh, N., & Devi, G. (2025). Estrogen, menopause, and Alzheimer’s disease: Understanding the link to cognitive decline in women. Frontiers in Molecular Biosciences, 12, 1634302. https://pubmed.ncbi.nlm.nih.gov/40661313/
Estrogen, Antidiuretic Hormone, and Nocturia
- Pauwaert, K., Goessaert, A.-S., Ghijselings, L., Bower, W., Mariman, A., Everaert, K., & De Wachter, S. (2021). Hormone therapy as a possible solution for postmenopausal women with nocturia: Results of a pilot trial. Menopause, 28(5), 502-510. https://pubmed.ncbi.nlm.nih.gov/33739310/
- Chang, T.-L., & Kuo, H.-C. (2024). Nocturia, nocturnal polyuria, and nocturnal enuresis in adults: What we know and what we do not know. Tzu Chi Medical Journal, 36(4), 370-376. https://pubmed.ncbi.nlm.nih.gov/39421492/
- Mun, J.-H., Kim, S.-O., Yu, H. S., Chung, H. S., & Kwon, D. (2015). Effects of desmopressin for the treatment of nocturnal polyuria in elderly women: Impact on related sleep quality. Canadian Urological Association Journal, 9(11-12), E770-E774. https://pubmed.ncbi.nlm.nih.gov/26600882/
Stress and Triggers
- Bergendahl, M., Iranmanesh, A., Pastor, C., Evans, W. S., & Veldhuis, J. D. (2000). Homeostatic joint amplification of pulsatile and 24-hour rhythmic cortisol secretion by fasting stress in midluteal phase women: Concurrent disruption of cortisol-growth hormone, cortisol-luteinizing hormone, and cortisol-leptin synchrony. The Journal of Clinical Endocrinology and Metabolism, 85(11), 4028-4035. https://pubmed.ncbi.nlm.nih.gov/11095428/
Additional References
- Corbo, R. M., Gambina, G., Broggio, E., & Scacchi, R. (2011). Influence of variation in the follicle-stimulating hormone receptor gene (FSHR) and age at menopause on the development of Alzheimer’s disease in women. Dementia and Geriatric Cognitive Disorders, 32(1), 63-69. https://pubmed.ncbi.nlm.nih.gov/21865747/
