Hormone replacement therapy can improve sleep in menopausal women, but the benefit depends on whether vasomotor episodes are driving the sleep disruption. In women with hot flashes and night sweats, a meta-analysis of over 15,000 women found moderate sleep improvement. The regimen with the largest effect size in head-to-head comparisons was transdermal 17-beta-estradiol combined with oral micronized progesterone. In women without vasomotor episodes, the evidence for sleep improvement is weaker.
Sleep disruption affects 40–60% of women during the menopausal transition, and approximately 40% in the late menopausal transition wake multiple times per night (Baker, 2023). Hormone replacement therapy is among the better-studied approaches for menopausal sleep disruption — and the evidence is dependent on context. The benefit varies by formulation, route of administration, and whether hot flashes and night sweats are the primary driver of insomnia.
This article covers what the meta-analyses and randomized controlled trials show about hormone replacement therapy and sleep, which regimens perform best in head-to-head comparisons, the vasomotor pathway through which hormone replacement therapy improves sleep consolidation, and the timeline from initiation to measurable improvement. For the broader picture of how hormonal changes affect sleep across a woman’s lifespan, see Hormonal Sleep Disruption in Women. For progesterone’s role as an independent sleep-promoting hormone, see the dedicated progesterone article.
Does Hormone Replacement Therapy Improve Sleep Quality in Menopausal Women?
In a meta-analysis of 15 randomized controlled trials enrolling 27,715 women, hormone replacement therapy improved subjective sleep quality with a standardized mean difference of −0.13. The benefit was strongest in women with vasomotor episodes at baseline. A separate meta-analysis of 7 trials and 15,468 women found moderate improvement (SMD −0.54) in women with hot flashes and night sweats.
Two meta-analyses anchor the evidence for hormone replacement therapy and sleep.
Cintron et al. (2017) pooled data from 7 randomized controlled trials enrolling 15,468 women. Among women with vasomotor episodes at baseline — hot flashes and night sweats — hormone replacement therapy produced a moderate improvement in sleep quality (SMD = −0.54; 95% CI: −0.91 to −0.18). In women without vasomotor episodes, no statistically meaningful benefit was observed in subgroup or combined analyses. This finding establishes a critical moderator: whether hot flashes and night sweats are present at baseline determines whether hormone replacement therapy is likely to improve sleep.
Pan et al. (2022) expanded the analysis to 15 randomized controlled trials and 27,715 women (14,058 receiving hormone replacement therapy, 13,657 controls). Overall, hormone replacement therapy improved subjective sleep quality (SMD = −0.13; 95% CI: −0.18 to −0.08; P < 0.00001). The effect size is modest across the full population -- but the subgroup data tell a more nuanced story. Women in the postmenopausal subgroup, women with vasomotor episodes, and women on specific regimens showed larger effect sizes (detailed in the next section).
A 2024 randomized controlled trial by Liu et al. added further support: in 152 menopausal women, combined hormone replacement therapy produced a 96% effectiveness rate on the Self-Rating Sleep Scale, with improvements across sleep latency, duration, depth, and daytime alertness compared to conventional care alone (P = 0.010–0.016 across subscales).
Across these studies, the pattern is consistent. Hormone replacement therapy improves subjective sleep quality in menopausal women, with the largest benefits in women whose sleep disruption is driven by vasomotor episodes.
Which Hormone Replacement Regimen Works Best for Sleep?
Transdermal 17-beta-estradiol produced the largest subjective sleep improvement (SMD −0.35) compared with oral estrogen (SMD −0.10). Estrogen combined with oral micronized progesterone outperformed estrogen combined with medroxyprogesterone acetate. Estrogen alone did not reach statistical significance for sleep improvement.
Pan et al. (2022) is the only head-to-head regimen comparison with sufficient statistical power to differentiate formulations. The subgroup results reveal a hierarchy.
By estrogen type: 17-beta-estradiol produced the largest subjective sleep improvement (SMD = −0.34; 95% CI: −0.51 to −0.17; P < 0.0001). Conjugated equine estrogens showed a smaller but statistically meaningful effect (SMD = −0.10; P < 0.00001). Estradiol valerate showed no positive effect on sleep quality.
By route of administration: Transdermal delivery outperformed oral delivery (transdermal SMD = −0.35; 95% CI: −0.64 to −0.06 versus oral SMD = −0.10). This difference may reflect more stable serum estradiol levels with transdermal delivery, avoiding the hepatic first-pass metabolism that oral formulations undergo.
By progestogen type: Estrogen combined with micronized progesterone produced the strongest combination effect (SMD = −0.22; 95% CI: −0.37 to −0.06; P = 0.007), compared with estrogen combined with medroxyprogesterone acetate (SMD = −0.10; P < 0.00001). Estrogen alone did not reach statistical significance for sleep improvement (SMD = −0.14; 95% CI: −0.40 to 0.12; P = 0.29).

A review by Haufe et al. (2022), analyzing 86 studies, identified that estrogen modulates serotonergic, noradrenergic, and thermoregulatory hypothalamic pathways — reducing sleep latency and nighttime awakenings while increasing total sleep time. Progesterone adds a second, independent mechanism: its neurosteroid metabolite allopregnanolone acts as a positive allosteric modulator of GABA-A receptors, producing sedative and anxiolytic effects that enhance NREM slow-wave sleep depth. Estrogen addresses vasomotor and serotonergic pathways; progesterone adds direct GABA-A-mediated sedation. The two mechanisms are complementary, which explains the superior combination effect in the meta-analysis data.
Does Hormone Replacement Therapy Reduce Night Sweats and Nighttime Awakenings?
Approximately 69% of nocturnal hot flashes are associated with cortical arousals or awakenings, and hot-flash-related wakefulness accounts for an average of 27% of total nighttime wakefulness in affected women. Reducing vasomotor episodes through hormone replacement therapy removes this source of sleep fragmentation.
Vasomotor activity is the primary mechanism through which hormone replacement therapy improves sleep consolidation. The link between nocturnal hot flashes and sleep fragmentation has been quantified in polysomnography studies.
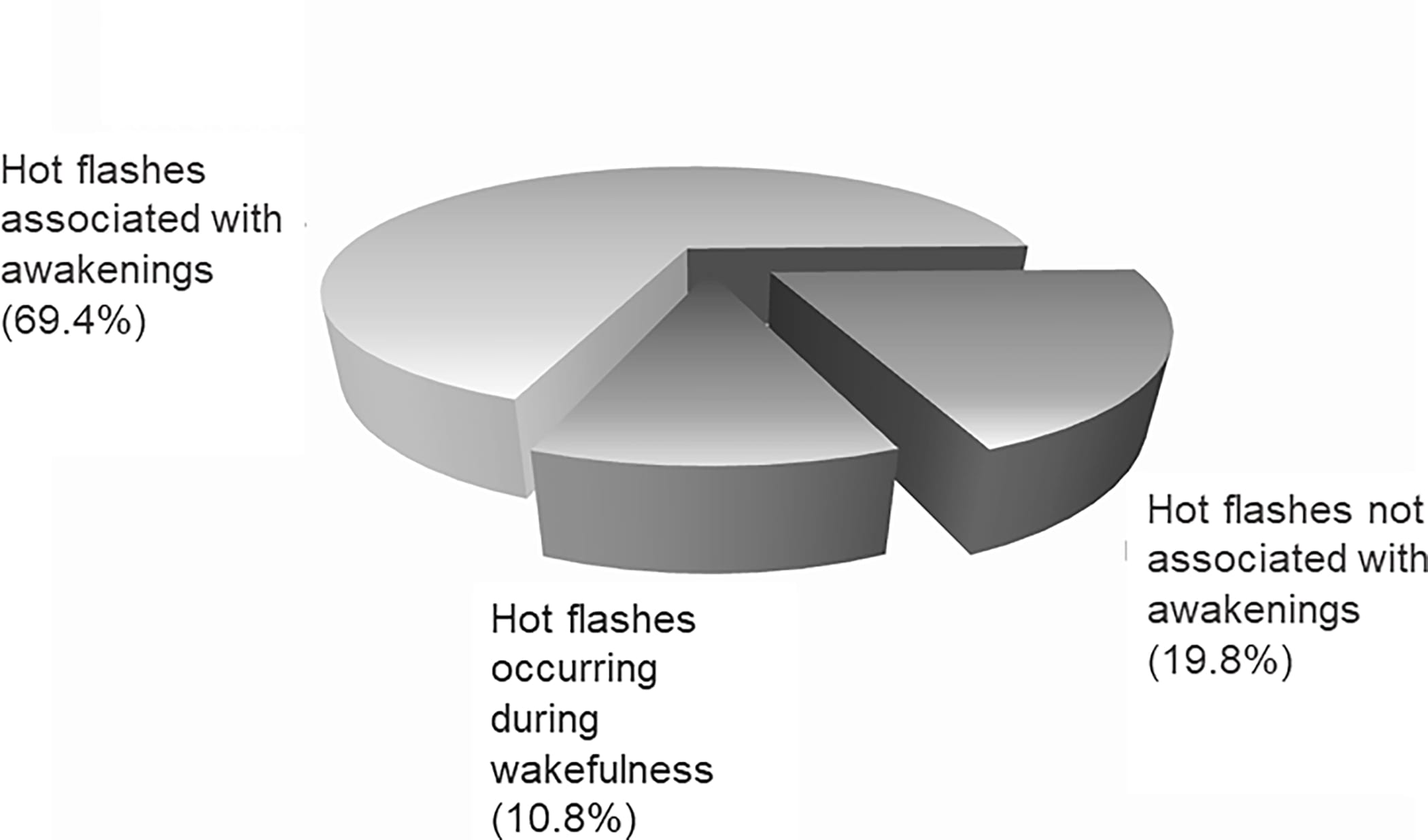
Baker (2023) reported data from 34 perimenopausal women with physiologically detected hot flashes during overnight polysomnography recordings (222 hot flash events total). An awakening occurred within a 3-minute window around the onset of the hot flash in 69.4% of events. Sleep was undisturbed in only 19.8% of hot flash events, and the remaining 10.8% occurred when the woman was already awake. Women with moderate-to-severe hot flashes were nearly three times more likely to report sleep disruption than women without vasomotor episodes.
Hot-flash-related wakefulness accounted for an average of 27% of total nighttime wakefulness in affected women. In a woman who is awake for 90 minutes overnight, approximately 24 minutes of that wakefulness is directly attributable to hot flashes triggering cortical arousals.
Hormone replacement therapy reduces nocturnal vasomotor episodes, fewer vasomotor episodes mean fewer cortical arousals, and fewer cortical arousals produce more consolidated sleep. Troìa et al. (2025) reported that vasomotor episodes, insomnia, and sleep-related breathing disturbances represent the three primary sleep disorder categories in perimenopause — with vasomotor episodes being the category that responds to hormonal restoration.
Troìa et al. (2025) also identified additional contributing factors: estrogen decline reduces upper-airway tone (worsening sleep-disordered breathing), decreases melatonin secretion, and interacts bidirectionally with anxiety and depression. Hormone replacement therapy can address the vasomotor and some of the neuroendocrine components, but not all of these pathways respond to a single approach.
How Long Does It Take for Hormone Replacement Therapy to Improve Sleep?
Sleep improvements from hormone replacement therapy typically emerge within weeks to 3 months. In a pilot study, subjective sleep quality improved at 1 month and remained improved at 3 months. The 2023 review positions the timeline at weeks for vasomotor relief, with full sleep benefit consolidating over 8–12 weeks.
Improvement from initiation to measurable sleep benefit is gradual.
Ogawa et al. (2025) conducted a pilot study in 15 peri- and postmenopausal women receiving combined transdermal estradiol and oral micronized progesterone. PSQI global scores improved from 7.8 at baseline to 6.1 at 1 month and 6.5 at 3 months. The sleep quality subscale improved from 1.7 at baseline to 1.0 at 1 month. At baseline, 86.7% of participants (13 of 15) qualified as poor sleepers (PSQI global score > 5).
Five of 15 participants (33.3%) were classified as responders (>=25% reduction in PSQI global score at 1 month), while 10 were non-responders. Responders had poorer habitual sleep efficiency at baseline compared to non-responders (P = 0.028) — suggesting that women with more disrupted sleep may derive larger benefits from this regimen. No differences in menopausal episode severity were observed between responders and non-responders, raising the possibility that some of the sleep improvement reflects the direct neurosteroid effects of allopregnanolone (progesterone’s active metabolite at GABA-A receptors) rather than vasomotor relief alone.
Liu et al. (2024) used three 28-day courses of combined hormone replacement therapy, with measurable sleep improvements accumulating over that timeframe. Baker (2023) positions the general timeline at weeks for initial vasomotor relief, with full sleep benefit consolidating over 8–12 weeks.
Women with worse baseline sleep quality tend to show stronger responses, and the full effect may take 2–3 months to consolidate. This is consistent with the time needed for estrogen to modulate serotonergic and thermoregulatory pathways and for progesterone’s neurosteroid metabolite to accumulate and engage GABA-A receptors.
Hormone replacement therapy addresses hormonally driven sleep disruption, but multiple causes often overlap. Cortisol dysregulation, GABA deficiency, inflammatory cytokines, metabolic disruption, and circadian misalignment can each independently fragment sleep — and more than one might be active at the same time. Identifying which causes might be involved is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Does Hormone Replacement Therapy Improve Polysomnography Results or Only Self-Reported Sleep?
In the largest regimen-comparison meta-analysis, objective polysomnography outcomes — total sleep time, sleep latency, sleep efficiency, and arousals — showed no improvements that reached statistical significance, while subjective sleep quality improved across regimens. Single-night polysomnography studies in small samples may not capture the cumulative sleep quality benefits that women experience over weeks.
Pan et al. (2022) measured both subjective and objective sleep outcomes. Subjective sleep quality improved with hormone replacement therapy (SMD = −0.13; P < 0.00001). Objective polysomnography outcomes did not reach significance: total sleep time (SMD = −0.14; P = 0.43), sleep latency (P = 0.23), sleep efficiency (P = 0.54), and arousals (P = 0.69).
This discrepancy is common in sleep research. Single-night polysomnography conducted in a laboratory setting captures one snapshot under artificial conditions. Chronic sleep quality as experienced over weeks and months — the variable that affects daytime functioning, mood, and long-term health — is better captured by validated questionnaires that average across many nights. The subjective improvements reported by women on hormone replacement therapy likely reflect measurable changes in sleep consolidation that a one-night laboratory recording cannot detect.
Does Hormone Replacement Therapy Support Melatonin Production?
Estrogen modulates serotonergic pathways, and serotonin is the precursor to melatonin. The mechanistic review covering 86 studies identified estrogen’s serotonergic and thermoregulatory CNS effects as direct pathways to sleep improvement — separate from the indirect vasomotor relief pathway.
Haufe et al. (2022) documented that estrogen acts directly on central nervous pathways involved in sleep regulation. Among these, estrogen modulates serotonergic activity — and serotonin is the biochemical precursor to melatonin synthesis in the pineal gland. As estrogen declines during menopause, serotonergic activity decreases, which can reduce downstream melatonin availability.
This pathway is separate from the indirect route through which hormone replacement therapy improves sleep by reducing hot flashes. Estrogen’s serotonergic effects may explain why some women experience sleep improvements from hormone replacement therapy even when vasomotor episodes are not the primary driver of their sleep disruption. For more on why melatonin supplementation alone often underperforms during menopause, see Why Doesn’t Melatonin Work for Menopause Insomnia?.
Does Hormone Replacement Therapy Work for All Types of Insomnia?
Hormone replacement therapy addresses sleep disruption driven by vasomotor episodes and hormonal decline. For insomnia independent of vasomotor episodes, cognitive behavioral therapy for insomnia (CBT-I) is recommended, with over 50% insomnia remission rates in an 8-week program for menopausal women.
Cintron et al. (2017) found no sleep benefit in women without vasomotor episodes at baseline. Hormone replacement therapy is not a broad-spectrum insomnia approach — it addresses a specific pathway.
For menopausal women whose insomnia is independent of hot flashes and night sweats, cognitive behavioral therapy for insomnia (CBT-I) has stronger evidence. Baker (2023) reported that an 8-week telephone-delivered CBT-I program achieved insomnia remission in more than 50% of participants, with improvements in daytime functioning, work performance, and depressive mood maintained through 6-month follow-up. For the “wired but tired” pattern — exhaustion combined with hyperarousal at night — the mechanisms may involve HPA axis dysregulation and autonomic imbalance rather than estrogen decline alone. See What Causes the “Wired but Tired” Feeling in Menopause? for that pathway.
Baker (2023) emphasizes a stepped-care model: match the approach to the predominant driver. If vasomotor episodes are primary, hormone replacement therapy addresses the upstream cause. If insomnia persists without vasomotor involvement, CBT-I or other approaches may be more appropriate.
Is Hormone Replacement Therapy Safe for Women with Breast Cancer History?
Hormone replacement therapy safety requires individualized evaluation. Oral micronized progesterone carries a more favorable safety profile than synthetic progestins regarding breast cancer risk, metabolic parameters, and venous thromboembolism. Troìa et al. (2025) recommend non-pharmacological approaches as first-line, with hormone replacement therapy individualized according to contraindications.
The safety profile of hormone replacement therapy varies by formulation. Troìa et al. (2025) position non-pharmacological approaches — particularly CBT-I — as first-line for perimenopausal sleep disruption, with hormone replacement therapy individualized according to contraindications and the specific profile of each woman.
Within hormone replacement therapy options, oral micronized progesterone carries a more favorable safety profile than synthetic progestins (such as medroxyprogesterone acetate) regarding breast cancer risk, metabolic parameters, and venous thromboembolism. This is an ongoing area of research, and individualized evaluation with a physician is appropriate for women with breast cancer history or other contraindications.
Related Reading
- Hormonal Sleep Disruption in Women — How estrogen, progesterone, and other hormones disrupt sleep across a woman’s lifespan
- Why Do You Wake Up at 3am During Menopause? — How cortisol and progesterone loss drive nocturnal waking in menopause
- Why Doesn’t Melatonin Work for Menopause Insomnia? — Why melatonin supplementation underperforms when estrogen decline is the upstream cause
- What Causes the “Wired but Tired” Feeling in Menopause? — How HPA axis changes and autonomic imbalance produce exhaustion with hyperarousal
- Why Can’t You Sleep Before Your Period? — How the progesterone crash in the late luteal phase fragments sleep and raises core body temperature
- Does Progesterone Help You Sleep? — The GABA-A receptor mechanism behind progesterone’s sleep-promoting effects and why dose timing matters
References
1. Cintron, D., et al. (2017). Efficacy of menopausal hormone therapy on sleep quality: systematic review and meta-analysis. Endocrine, 55(3), 702–711. https://pubmed.ncbi.nlm.nih.gov/27515805/
2. Pan, Z., et al. (2022). Different regimens of menopausal hormone therapy for improving sleep quality: a systematic review and meta-analysis. Menopause, 29(5), 627–635. https://pubmed.ncbi.nlm.nih.gov/35102100/
3. Haufe, A., Baker, F. C., & Leeners, B. (2022). The role of ovarian hormones in the pathophysiology of perimenopausal sleep disturbances: A systematic review. Sleep Medicine Reviews, 66, 101710. https://pubmed.ncbi.nlm.nih.gov/36356400/
4. Baker, F. C. (2023). Optimizing sleep across the menopausal transition. Climacteric, 26(3), 198–205. https://pubmed.ncbi.nlm.nih.gov/37011660/
5. Liu, Q., Huang, Z., & Xu, P. (2024). Effects of hormone replacement therapy on mood and sleep quality in menopausal women. World Journal of Psychiatry, 14(7), 1087–1094. https://pubmed.ncbi.nlm.nih.gov/39050194/
6. Troìa, L., et al. (2025). Sleep disturbance and perimenopause: A narrative review. Journal of Clinical Medicine, 14(5), 1479. https://pubmed.ncbi.nlm.nih.gov/40094961/
7. Ogawa, M., et al. (2025). Changes in sleep quality after hormone replacement therapy with micronized progesterone in Japanese menopausal women: A pilot study. Journal of Menopausal Medicine, 31(1), 45–50. https://pubmed.ncbi.nlm.nih.gov/40347165/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
