Premenstrual insomnia has three converging causes. Progesterone drops in the late luteal phase, withdrawing GABAergic sedation that maintained sleep earlier in the cycle. Core body temperature rises approximately 0.4 °C after ovulation, blunting the nocturnal temperature drop that normally initiates sleep. Melatonin onset is delayed by up to 90 minutes in the luteal phase, and progesterone creates resistance to melatonin’s temperature-lowering effect. These three changes overlap in the days before menstruation.
Among cycle-tracked populations, 29% of women show perimenstrual increases in sleep difficulty, while 25% experience mid-cycle increases and 46% show no menstrual phase relationship (Alzueta & Baker, 2023). For those who do experience cyclical insomnia, the disruption is predictable to the day — arriving in the same premenstrual window, cycle after cycle — yet its hormonal drivers are distinct from the causes addressed by general sleep recommendations.
This article covers the three mechanisms that converge before menstruation: progesterone withdrawal and the loss of GABA-mediated sedation, luteal-phase body temperature elevation, and melatonin disruption. For the broader landscape of how reproductive hormones affect sleep across a woman’s lifespan, see the full hormonal sleep disruption article.
Cyclical hormonal insomnia is one of several causes of sleep disruption in women. Cortisol dysregulation, inflammatory load, and circadian misalignment may also be involved — and each may compound during the premenstrual window.
Does Progesterone Withdrawal Before Your Period Cause Insomnia?
Progesterone’s metabolite allopregnanolone acts on GABA-A receptors to promote sleep during the mid-luteal phase. When progesterone drops before menstruation, this GABAergic sedation withdraws. Research shows the rate of progesterone rise — not the peak level — predicts how much sleep fragments.
The mid-luteal sedation effect
During the mid-luteal phase, progesterone rises and is converted to allopregnanolone — a neurosteroid that enhances GABA-A receptor activity. GABA-A receptors are the primary inhibitory gatekeepers in the brain: when allopregnanolone binds them, neural excitability decreases and sleep is promoted. This is the same receptor site targeted by benzodiazepines and alcohol, though allopregnanolone modulates it through a different binding mechanism.
A 2023 review of reproductive hormones and sleep disturbances across the female lifespan documented that women report better sleep during the follicular and ovulatory phases — when progesterone is low and stable — and worse sleep during the premenstrual phase, when progesterone is declining. The review identified the late-luteal progesterone withdrawal as the primary cyclical insomnia trigger, characterizing it as an abrupt removal of GABAergic sedation (Haufe & Leeners, 2023).
The rate of change matters more than the peak
A study of 27 premenopausal women with regular cycles combined serial hormone measurements with home polysomnography during the luteal phase. The central finding: a steeper rate of progesterone rise from the follicular baseline to the mid-luteal peak predicted greater wake after sleep onset (WASO; p = 0.016). The velocity of the hormonal change — not the absolute peak concentration — was the driver of sleep fragmentation. Higher circulating estrone was independently associated with elevated WASO (p = 0.03), though this effect was attenuated when accounting for the rate of progesterone change (Sharkey et al., 2014).
This reframes premenstrual insomnia as a sensitivity to hormonal dynamics rather than a threshold effect of progesterone concentration. Women who mount rapid, large luteal progesterone rises may be disproportionately vulnerable to premenstrual sleep fragmentation.
The biphasic paradox
Progesterone’s relationship to sleep is not linear. During the mid-luteal phase, sustained progesterone and its metabolites carry sedative properties via GABA-A receptor modulation. But the late-luteal decline withdraws that sedation. A polysomnographic study of 20 midlife women found that when progesterone was elevated (3 ng/mL or above), both women with and without insomnia experienced more awakenings per hour (p = .003) and greater arousal frequency (p = .025) compared with the follicular phase (de Zambotti et al., 2015). The disruption is physiological — it occurs in healthy women, not only those with existing insomnia.
The 3am waking pattern that many women experience before their period may reflect this same progesterone withdrawal. For women in menopause, where progesterone decline is permanent rather than cyclical, the 3am waking pattern has its own set of drivers.
Does Body Temperature Rise After Ovulation Affect Sleep?
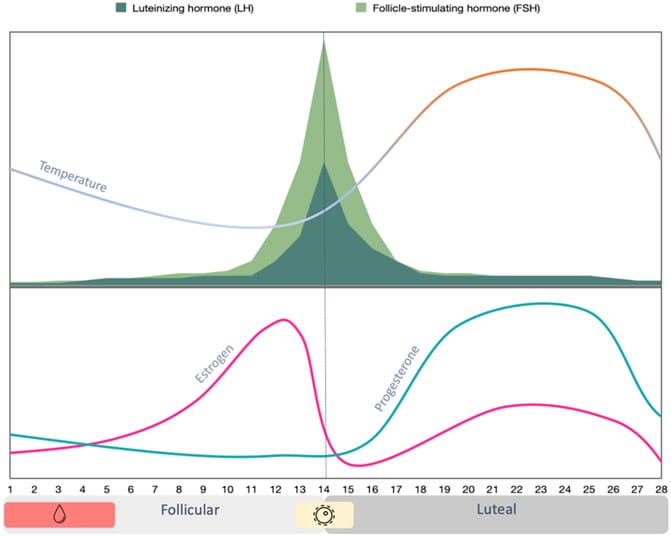
Core body temperature rises approximately 0.4 °C in the luteal phase, blunting the nocturnal temperature decline that normally facilitates sleep onset. Melatonin onset is delayed by up to 90 minutes, and the body resists melatonin’s temperature-lowering effect during this phase.
How temperature initiates sleep
Sleep onset depends on a drop in core body temperature. The body normally begins cooling in the evening — driven partly by melatonin, which promotes heat loss through peripheral vasodilation. This nocturnal temperature decline is one of the primary physiological gates for falling asleep. When the decline is blunted, sleep onset is delayed.
The luteal temperature elevation
After ovulation, progesterone raises the thermoregulatory set point. A 2023 review quantified this as an approximately 0.4 °C elevation in core body temperature during the luteal phase compared to the follicular phase, with a blunted nocturnal decline that reduces the thermal facilitation of sleep onset. REM sleep was reduced in the luteal phase and negatively correlated with both progesterone and estradiol levels — consistent with a thermogenic suppression of REM architecture (Alzueta & Baker, 2023).
Women tracking with wearable devices (Oura, RingConn, Apple Watch) often see this temperature rise reflected in their data during the second half of the cycle. The elevation is not subtle — 0.4 °C is large enough to measurably delay the evening cooling that facilitates sleep.
Melatonin onset delay and resistance
A study measuring circadian core body temperature and melatonin rhythms across cycle phases found that in the luteal phase, melatonin onset was delayed by 90 minutes compared to the follicular phase (p < 0.01). Mean 24-hour core body temperature was elevated by 0.3 °C, and the circadian temperature amplitude was reduced by 40%. When 2.5 mg of exogenous melatonin was administered during daytime, it lowered core body temperature in the follicular phase but had no effect in the luteal phase — demonstrating a progesterone-associated resistance to melatonin's hypothermic action (Cagnacci et al., 1996).
This finding has direct relevance for women who take melatonin before their period and find it less effective. The resistance is not a dosing problem — it is a progesterone-driven change in how the body responds to melatonin’s temperature-lowering function. For women in menopause experiencing a related phenomenon where melatonin supplementation underperforms, that mechanism involves estrogen decline rather than progesterone elevation.
A 2024 review of 25 studies on biological rhythm disturbances in PMS and PMDD populations found that lower nighttime melatonin secretion and elevated nighttime body temperature co-occur in these groups, pointing toward a circadian rhythm disruption that goes beyond isolated progesterone sensitivity (Nexha et al., 2024).
Does the Menstrual Cycle Change Sleep Architecture?
Polysomnographic studies show that sleep architecture changes during the luteal phase. Sleep efficiency drops by approximately 5%, total sleep time decreases by about 25 minutes premenstrually, and awakenings increase. Sleep spindle density rises — a GABA-A receptor effect of progesterone metabolites on thalamocortical circuits.
What polysomnography shows
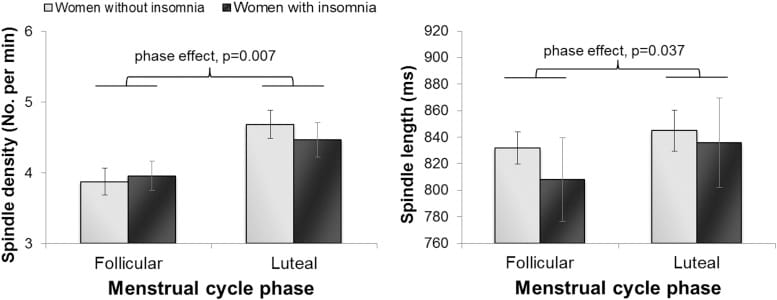
The sleep changes women feel before their period are measurable on polysomnography (PSG). In the de Zambotti et al. (2015) study of 20 midlife women, the luteal phase produced reduced slow-wave sleep (p = .024), increased spindle density (p = .007), longer spindles (p = .037), and elevated 14-17 Hz EEG power (p < .05). These changes occurred in both women with and without insomnia — confirming that luteal-phase sleep disruption is a physiological feature of the cycle, not an artifact of pre-existing sleep conditions (de Zambotti et al., 2015).
Women with insomnia showed additional deficits across all phases: shorter total sleep time (p = .012), elevated wake after sleep onset (p = .031), and lower sleep efficiency (p = .034). The luteal phase compounded these existing vulnerabilities.
The spindle increase
The rise in sleep spindle density during the luteal phase is attributed to progesterone metabolites — particularly allopregnanolone — acting at GABA-A receptors on thalamocortical circuits. Spindles are brief bursts of oscillatory brain activity (11-16 Hz) generated during NREM sleep. While spindles are associated with memory consolidation and sleep stability, the progesterone-driven increase in spindle density during the luteal phase co-occurs with reduced slow-wave sleep and more awakenings — indicating that the GABA-A receptor modulation does not uniformly improve sleep.
Sleep difficulty patterns vary between women
A large cycle-tracked study of 213 participants found that sleep difficulty patterns across the menstrual cycle are heterogeneous: 46% showed no menstrual phase relationship, 25% experienced mid-cycle increases in difficulty, and 29% reported perimenstrual increases. Late-reproductive women demonstrated a 5% decline in sleep efficiency and a 25-minute reduction in total sleep time during the premenstrual week compared with other phases (Alzueta & Baker, 2023).
A 2023 narrative review found that women with PMS show poorer sleep across studies, and that progesterone’s biphasic effect — sedative when sustained, disruptive when declining — is a consistent finding in the literature. The review emphasized that these are physiological changes occurring in healthy women, not only those with existing sleep conditions (Andersen et al., 2023).
Which Factors Might Be Involved in Your Premenstrual Insomnia?
Cyclical hormonal insomnia often compounds with other contributors — including cortisol dysregulation, inflammatory load, metabolic disruption, and circadian misalignment — each of which may worsen during the premenstrual window.
Cyclical hormonal insomnia often compounds with other contributors. Cortisol dysregulation can elevate arousal at night. Inflammatory load can fragment sleep continuity. Metabolic disruption can alter blood glucose regulation during sleep. Circadian misalignment can delay or advance sleep timing. Each of these may worsen during the premenstrual window — and when multiple contributors overlap, the insomnia is more than the sum of its parts.
Identifying which causes might be involved is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
How Many Women Experience Sleep Changes Before Their Period?
Among cycle-tracked populations, 29% show perimenstrual sleep difficulty increases, while 25% experience mid-cycle increases. The pattern is not universal — 46% show no menstrual phase relationship (Alzueta & Baker, 2023).
Does the Progesterone Drop Before a Period Affect Serotonin and Melatonin?
Progesterone influences serotonin metabolism, and the late-luteal drop can reduce serotonin availability. Serotonin is the precursor to melatonin. Combined with the direct melatonin onset delay and blunted temperature response documented in the luteal phase, the premenstrual window faces a convergence of three disrupted sleep-promoting pathways: reduced GABAergic sedation, delayed melatonin onset, and lowered serotonin-to-melatonin conversion (Nexha et al., 2024).
When Does Progesterone Start to Drop Before a Period?
Progesterone begins declining approximately 5-7 days before menstruation in a typical 28-day cycle. The rate of this decline — not the absolute level — predicts the degree of sleep fragmentation, meaning women with steeper drops may experience more insomnia (Sharkey et al., 2014).
Does Oral Micronized Progesterone Help Cycle-Related Insomnia?
Oral micronized progesterone at bedtime produces sedative effects through allopregnanolone conversion and GABA-A receptor modulation. Whether supplemental progesterone during the late luteal phase can prevent cycle-related insomnia is an active area of investigation, with some evidence supporting improved sleep quality in perimenopausal women using this approach (Haufe & Leeners, 2023).
Related Reading
- Hormonal Sleep Disruption in Women — How estrogen, progesterone, and other hormones disrupt sleep across a woman’s lifespan
- Why Do You Wake Up at 3am During Menopause? — How cortisol and progesterone loss drive nocturnal waking in menopause
- Why Doesn’t Melatonin Work for Menopause Insomnia? — Why melatonin supplementation underperforms when estrogen decline is the upstream cause
- What Causes the “Wired but Tired” Feeling in Menopause? — How HPA axis changes and autonomic imbalance produce exhaustion with hyperarousal
- Does Hormone Replacement Therapy Help with Sleep in Menopause? — What the clinical evidence shows about HRT’s effect on sleep architecture and hot-flash-driven waking
- Does Progesterone Help You Sleep? — The GABA-A receptor mechanism behind progesterone’s sleep-promoting effects and why dose timing matters
References
1. Alzueta, E., & Baker, F. C. (2023). The menstrual cycle and sleep. Sleep Medicine Clinics, 18(4), 399-413. https://pubmed.ncbi.nlm.nih.gov/38501513/
2. Andersen, M. L., Hachul, H., Ishikura, I. A., & Tufik, S. (2023). Sleep in women: A narrative review of hormonal influences, sex differences and health implications. Frontiers in Sleep, 2, 1271827. https://pubmed.ncbi.nlm.nih.gov/41426438/
3. Cagnacci, A., Soldani, R., Laughlin, G. A., & Yen, S. S. (1996). Modification of circadian body temperature rhythm during the luteal menstrual phase: Role of melatonin. Journal of Applied Physiology, 80(1), 25-29. https://pubmed.ncbi.nlm.nih.gov/8847311/
4. de Zambotti, M., Willoughby, A. R., Sassoon, S. A., Colrain, I. M., & Baker, F. C. (2015). Menstrual cycle-related variation in physiological sleep in women in the early menopausal transition. Journal of Clinical Endocrinology & Metabolism, 100(8), 2918-2926. https://pubmed.ncbi.nlm.nih.gov/26079775/
5. Haufe, A., & Leeners, B. (2023). Sleep disturbances across a woman’s lifespan: What is the role of reproductive hormones? Journal of the Endocrine Society, 7(5), bvad036. https://pubmed.ncbi.nlm.nih.gov/37091307/
6. Nexha, A., Caropreso, L., Cardoso, T. A., Suh, J. S., Tonon, A. C., & Frey, B. N. (2024). Biological rhythms in premenstrual syndrome and premenstrual dysphoric disorder: A systematic review. BMC Women’s Health, 24(1), 551. https://pubmed.ncbi.nlm.nih.gov/39375682/
7. Sharkey, K. M., Crawford, S. L., Kim, S., & Joffe, H. (2014). Objective sleep interruption and reproductive hormone dynamics in the menstrual cycle. Sleep Medicine, 15(6), 688-693. https://pubmed.ncbi.nlm.nih.gov/24841109/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
