Consumer vagus nerve stimulation devices — Pulsetto, Nurosym, Truvaga — are marketed for sleep, but the available information is dominated by brand sales pages and dense journal articles. People considering these devices need to know what the published evidence shows: effect sizes, limitations, and how the mechanism works.
This article covers how transcutaneous auricular vagus nerve stimulation (taVNS) affects sleep physiology, what the randomized controlled trials found (including the 2024 JAMA trial and 2025 meta-analysis), what brain imaging reveals about the mechanism, and what consumers should know before purchasing. It does not cover the full autonomic overview — for that, see Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It. Vagal tone is one of several autonomic factors affecting sleep; the pillar covers the broader picture.
How Does Vagus Nerve Stimulation Affect Sleep?
The auricular branch of the vagus nerve — the only peripheral branch that surfaces at the skin — runs through the outer ear (the cymba conchae and tragus). Stimulating this branch with a small electrical current sends afferent input along the vagus nerve to the nucleus tractus solitarius (NTS) in the brainstem. The NTS connects to the locus coeruleus (which regulates norepinephrine and arousal), the thalamus (which gates sensory input to the cortex), and cortical regions involved in attention and emotion regulation.
In insomnia, the thalamus is often over-connected to arousal-related cortical regions, keeping the brain alert at sleep onset. A 2023 fMRI study compared resting-state brain scans in 20 people with insomnia before and after taVNS, with 20 healthy controls as a reference (Zhao et al., 2023). At baseline, the insomnia group showed increased connectivity between the thalamus and the right insula and inferior frontal gyrus — regions associated with interoceptive awareness and arousal. After taVNS, connectivity between the thalamus and the anterior cingulate cortex and precuneus decreased. These are core nodes of the default mode network, the brain circuit active during ruminative thought at sleep onset.
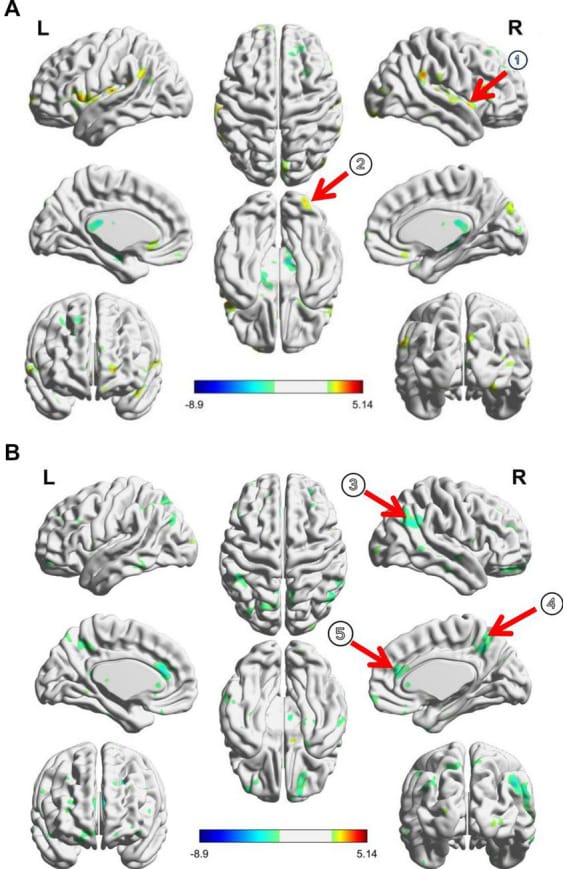
A 2025 randomized controlled trial with fMRI examined a different circuit: the basal forebrain-cortical pathway (Qi et al., 2025). The basal forebrain is a major source of acetylcholine, a neurotransmitter involved in cortical arousal. In 67 people with primary insomnia randomized to active or sham taVNS for four weeks, active taVNS decreased connectivity between the basal forebrain and visual and somatosensory cortical regions. The reduction in basal forebrain-occipital connectivity correlated with insomnia improvement (r = 0.490, p = 0.008), and a predictive model using baseline connectivity identified who would respond to taVNS (r = 0.520 in the primary cohort, r = 0.443 in validation).
The pattern across both imaging studies: taVNS appears to reduce cortical hyperarousal by normalizing connectivity in thalamocortical and basal forebrain-cortical circuits.
What Did the Randomized Controlled Trials Find for Vagus Nerve Stimulation and Insomnia?
The Journal of the American Medical Association (JAMA) Network Open Trial (Zhang et al., 2024)
Zhang et al. enrolled 72 adults with chronic insomnia disorder (PSQI score of 8 or higher) and randomized them to active taVNS or sham stimulation (Zhang et al., 2024). Participants received 30-minute sessions twice daily, five days per week, for 8 weeks, with a 12-week observational follow-up.
The primary outcome: PSQI decreased by 8.2 points in the taVNS group versus 3.9 points in the sham group — a between-group difference of 4.2 points (p < 0.001, Cohen's d = 1.2). A d of 1.2 is considered a large effect by conventional benchmarks. Secondary outcomes including depression (Hamilton Depression Scale, HAMD), anxiety (Hamilton Anxiety Scale, HAMA), sleepiness (Epworth Sleepiness Scale, ESS), and fatigue (Flinders Fatigue Scale, FFS) all improved in the active group. The improvement was maintained through week 20, meaning the benefit persisted 12 weeks after stimulation ended.
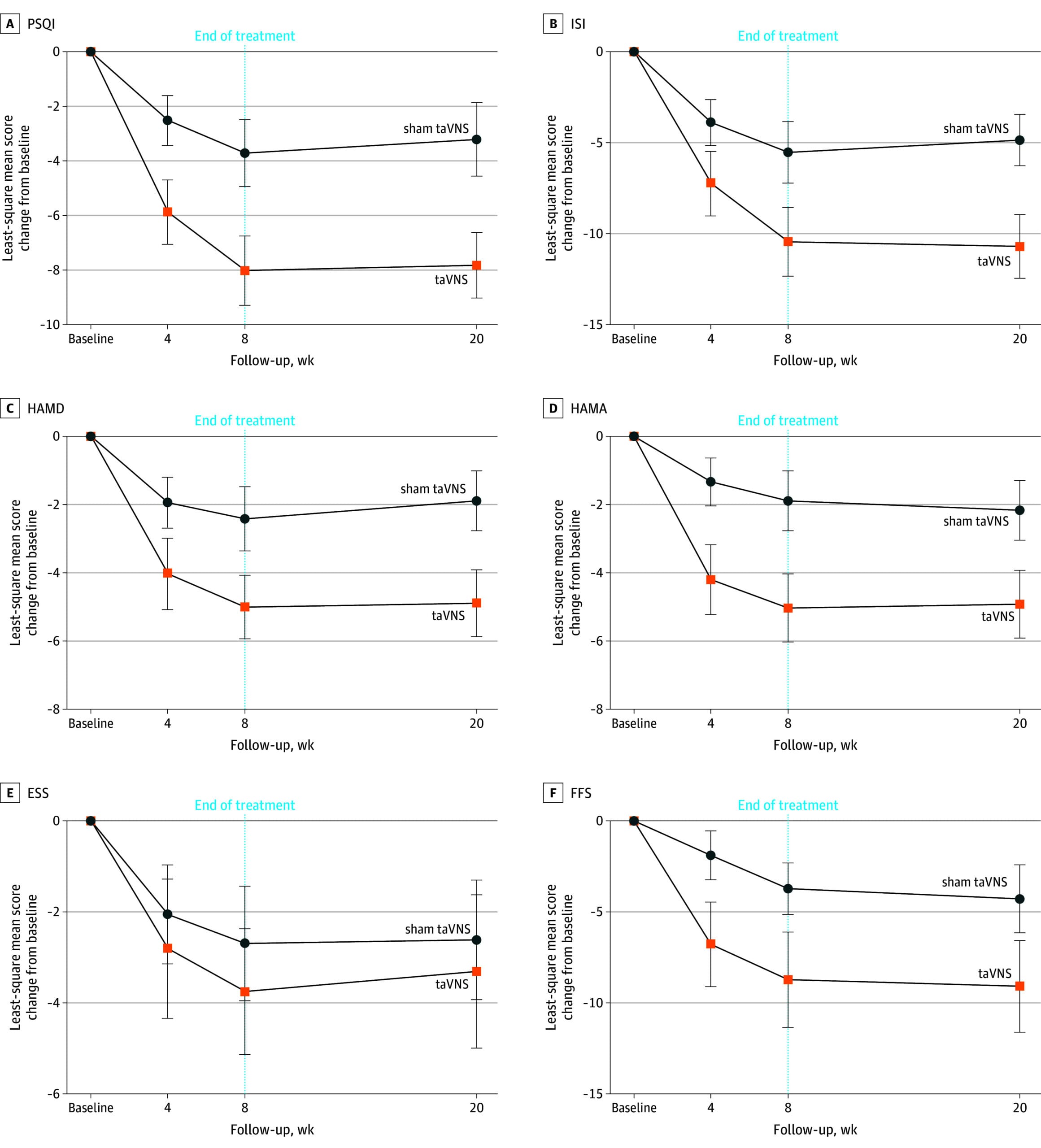
The Sleep Medicine Trial (Yeom et al., 2025)
A double-blind trial of 40 adults with chronic insomnia confirmed the JAMA direction (Yeom et al., 2025). Over six weeks of daily 30-minute sessions, PSQI decreased by 4.5 versus 1.9 points (d = -1.21, p = 0.009). The Insomnia Severity Index improved by 7.6 versus 4.25 points (d = -0.75, p = 0.023). Actigraphy showed total sleep time increased in the taVNS group while declining in the sham group (d = 0.69, p = 0.019). Quality of life also improved. No adverse events were reported.
The Meta-Analysis (de Oliveira et al., 2025)
A 2025 meta-analysis pooled 6 randomized controlled trials comprising 336 participants (de Oliveira et al., 2025). The pooled PSQI difference was -3.60 (95% CI: -4.98 to -2.22, p < 0.01). The pooled Insomnia Severity Index (ISI) difference was -5.24 (95% CI: -9.02 to -1.46, p < 0.01). Improvements appeared across PSQI subscales: sleep quality, latency, duration, and efficiency.
A PSQI improvement of 3 or more points exceeds the threshold for a measurable change in sleep quality. The pooled effect clears this threshold.
One limitation: the GRADE evidence rating was Very Low, meaning the evidence base is still small and variable across trials. The authors called for larger, longer-term randomized controlled trials to strengthen the evidence.
Does Vagus Nerve Stimulation Work for Insomnia Caused by Anxiety or Post-Traumatic Stress Disorder (PTSD)?
The randomized controlled trials above enrolled people with primary insomnia. But many people searching for vagus nerve stimulation devices have insomnia driven by anxiety or trauma.
Bottari et al. (2024) conducted a crossover polysomnography study in 13 veterans with PTSD (Bottari et al., 2024). Each veteran received one hour of active taVNS at bedtime on one night and sham on another, with full polysomnography (PSG) recording both nights. Active taVNS produced a modest increase in N3 (deep/slow-wave) sleep (Cohen’s d = 0.23) and a moderate decrease in the cyclic alternating pattern (CAP) rate — an electroencephalography (EEG) measure of sleep instability (d = -0.65). Respiratory sinus arrhythmia, a direct measure of parasympathetic cardiac activity, increased during the pre-bedtime period. This is the first study to combine polysomnography with a direct autonomic outcome measure in taVNS sleep research.
Rapid eye movement (REM) sleep showed a small-to-moderate reduction (d = -0.48), and total sleep time decreased modestly (d = -0.31). The sample was small and the design used a single session per condition, so these findings are preliminary.
Srinivasan et al. (2024) tested taVNS in 42 elderly healthcare workers with post-COVID anxiety and sleep disturbances (Srinivasan et al., 2024). Participants were randomized to taVNS or progressive muscle relaxation (an evidence-based active comparator), with 30-minute sessions three times weekly for four weeks. The taVNS group showed greater improvement in both sleep quality and anxiety (p = 0.001). Using progressive muscle relaxation as the comparator strengthens this finding by ruling out nonspecific relaxation effects.
taVNS effects on sleep appear consistent whether the insomnia is primary, anxiety-comorbid, or PTSD-related — suggesting it may target a shared autonomic mechanism (parasympathetic insufficiency and cortical hyperarousal) rather than being condition-dependent.
What Should Consumers Know Before Buying a Vagus Nerve Stimulation Device for Sleep?
What the trials used versus what consumers can buy
The published trials used medical-grade stimulators with controlled parameters: typically 25 Hz frequency, 0.2 ms pulse width, current below the pain threshold, and 30-minute sessions. Consumer devices target the same anatomical location and some publish similar specifications, but the devices themselves have not been independently tested in published randomized controlled trials.
The evidence supports taVNS as a mechanism and as an approach. It does not yet confirm that any one consumer device reproduces the effect observed in the trials. This does not mean the consumer devices are ineffective — it means independent validation has not been published.
What the Grading of Recommendations, Assessment, Development, and Evaluations (GRADE) rating means
The 2025 meta-analysis rated the overall evidence as Very Low on the GRADE scale (de Oliveira et al., 2025). In GRADE terminology, “Very Low” means the evidence base is small and the trials vary in design. It does not mean the approach is ineffective — it means larger, more standardized trials are needed to increase confidence in the effect size.
Practical considerations
Based on the published study methods: sessions lasted 30 minutes, frequency was daily or twice daily, and the minimum study duration showing benefit was 4 weeks (Srinivasan et al., 2024). The JAMA trial used twice-daily sessions for 8 weeks (Zhang et al., 2024), with benefits maintained through week 20. Consumers should expect a minimum commitment of 4-6 weeks of consistent daily use before assessing benefit.
Consumer taVNS devices range from approximately $200 to $900+. The published evidence does not support one brand over another, because no consumer brand has been tested in an independent randomized controlled trial.
Vagal tone may not be the only autonomic factor affecting your sleep. Parasympathetic insufficiency can overlap with cortisol-driven hyperarousal, GABA pathway impairment, circadian timing disruptions, or inflammatory mechanisms. When multiple causes might be contributing, identifying which ones are active is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Can Vagus Nerve Exercises Like Breathing and Cold Exposure Improve Sleep?
Slow-paced breathing at 6 breaths per minute activates the baroreflex arc, increasing vagal outflow and enhancing respiratory sinus arrhythmia — the same autonomic marker that improved in the Bottari et al. post-traumatic stress disorder study with taVNS. A 2019 study found that 30 days of twice-daily slow breathing improved cardiac vagal activity and sleep quality (Laborde et al., 2019). Cold water face immersion triggers the dive reflex through the trigeminal nerve, producing a rapid increase in parasympathetic tone through a different entry point.
The tradeoff: behavioral vagal activation requires sustained daily practice. taVNS is passive — the device delivers stimulation while the person sits or rests. For people who have difficulty maintaining a daily breathing practice, device-based stimulation offers more consistent delivery.
Does Vagus Nerve Stimulation Increase Deep Sleep?
The Bottari et al. (2024) study is the only published taVNS trial that measured sleep architecture with polysomnography. The N3 increase (d = 0.23) was modest and came from a single bedtime session in 13 participants. Whether repeated taVNS over weeks produces larger changes in deep sleep duration is an open question.
The longer trials — Zhang 2024 (8 weeks), Yeom 2025 (6 weeks), Srinivasan 2024 (4 weeks) — measured sleep quality using the PSQI and ISI, which are subjective questionnaires, and in one case actigraphy, which estimates sleep and wake periods but does not differentiate between sleep stages. The subjective improvements are well-documented. Whether those correspond to measurable changes in deep sleep duration or slow-wave activity remains to be tested in longer polysomnography studies.
How Long Does It Take for Transcutaneous Auricular Vagus Nerve Stimulation (taVNS) to Improve Sleep?
Srinivasan et al. (2024) showed improvement at 4 weeks with three sessions per week. Zhang et al. (2024) showed large improvements at 8 weeks with twice-daily sessions, and these improvements persisted through week 20 — suggesting taVNS-induced brain changes may be durable rather than dependent on continued stimulation.
The fMRI data from Zhao et al. (2023) and Qi et al. (2025) offer context: the brain connectivity changes reflect neural reorganization, not an acute pharmacological effect. A single session may produce measurable autonomic effects (as in the Bottari et al. study), but the larger improvements in sleep quality appear to require at least 4 weeks.
Can Vagus Nerve Stimulation Replace Sleeping Pills?
The meta-analysis authors (de Oliveira et al., 2025) identified this as a gap in the evidence. The effect sizes in the taVNS trials (d = 1.2 in the JAMA trial) are numerically comparable to those reported for some sleep medications — but different study populations and outcome measures make indirect comparison unreliable. For anyone currently taking sleep medication, changes should be discussed with their prescriber.
Related Reading:
- Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It — how autonomic imbalance disrupts sleep architecture
- Can Your Nervous System Get Stuck in Fight or Flight and Ruin Your Sleep? — the self-reinforcing cortisol-sleep loop
- What Is Hyperarousal Insomnia? Why You’re Wired but Tired Every Night — autonomic mechanics behind the wired-but-tired state
- Why Won’t Your Brain Shut Off at Night? The Autonomic Connection — GABA receptor impairment and sympathetic overactivation
- Why Is Your Rapid Eye Movement Sleep Fragmented? The Brainstem Switch That Controls It — the brainstem circuit and cholinergic pathway
- Does Benadryl Destroy Your Sleep? How Anticholinergic Drugs Suppress REM — anticholinergic medications and REM suppression
- What Your Overnight HRV Is Telling You About Your Sleep: The Vagal Tone Connection — HRV as a window into parasympathetic recovery
- How Your Gut Talks to Your Brain Through the Vagus Nerve — and Why It Matters for Sleep — gut-vagus-brain pathway
- Do Antidepressants Suppress Rapid Eye Movement Sleep? What SSRIs, SNRIs, and Tricyclics Do to Sleep Architecture — serotonergic REM suppression from medications
- Does Your Sleep Tracker Accuracy Matter? What Oura, WHOOP, and Apple Watch Get Right and Wrong About Sleep Stages — wearable validation data
- Is Your Insomnia a Nervous System Problem? How to Tell the Difference — identifying whether autonomic imbalance is driving your insomnia
- Do Antidepressants Suppress Rapid Eye Movement Sleep? — SSRIs, SNRIs, and tricyclics affect sleep architecture
- Does Your Sleep Tracker Accuracy Matter? — what Oura, WHOOP, and Apple Watch get right and wrong
- Is Your Insomnia a Nervous System Problem? — how to tell if autonomic dysregulation is driving sleep disruption
References
Bottari, S. A., et al. (2024). Preliminary evidence of transcutaneous vagus nerve stimulation effects on sleep in veterans with post-traumatic stress disorder. Journal of Sleep Research, 33(1), e13891. https://pubmed.ncbi.nlm.nih.gov/37039398/
de Oliveira, H. M., et al. (2025). Transcutaneous auricular vagus nerve stimulation in insomnia: A systematic review and meta-analysis. Neuromodulation. https://pubmed.ncbi.nlm.nih.gov/40323248/
Laborde, S., et al. (2019). Influence of a 30-day slow-paced breathing intervention compared to social media use on subjective sleep quality and cardiac vagal activity. Journal of Clinical Medicine, 8(2), 193. https://pubmed.ncbi.nlm.nih.gov/30736268/
Qi, M., et al. (2025). Baseline functional connectivity of the basal forebrain-cortical circuit predict taVNS treatment response in primary insomnia: A randomized controlled trial and fMRI study. BMC Medicine, 23, 340. https://pubmed.ncbi.nlm.nih.gov/40629377/
Srinivasan, V., et al. (2024). Effect of vagus nerve stimulation (taVNS) on anxiety and sleep disturbances among elderly health care workers in the post COVID-19 pandemic. Work, 77(1), 351-358. https://pubmed.ncbi.nlm.nih.gov/38143418/
Yeom, J. W., et al. (2025). Transcutaneous auricular vagus nerve stimulation (taVNS) improves sleep quality in chronic insomnia disorder: A double-blind, randomized, sham-controlled trial. Sleep Medicine, 129, 36-43. https://pubmed.ncbi.nlm.nih.gov/40398066/
Zhang, S., et al. (2024). Transcutaneous auricular vagus nerve stimulation for chronic insomnia disorder: A randomized clinical trial. JAMA Network Open, 7(12), e2451217. https://pubmed.ncbi.nlm.nih.gov/39680406/
Zhao, B., et al. (2023). Altered functional connectivity of the thalamus in patients with insomnia disorder after transcutaneous auricular vagus nerve stimulation therapy. Frontiers in Neurology, 14, 1164869. https://pubmed.ncbi.nlm.nih.gov/37483453/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 8 references cited
