Millions of people now see HRV data from Oura, WHOOP, or Apple Watch every morning. A low overnight reading generates concern — but wearable dashboards don’t explain what the number represents in terms of the autonomic nervous system. Understanding vagal tone — the degree of parasympathetic activity mediated by the vagus nerve — reframes the question from “is my HRV bad?” to “what is my autonomic nervous system doing during sleep?”
This article covers what HRV measures during sleep, how vagal tone affects sleep onset and sleep architecture, what low overnight HRV indicates about autonomic balance, and what the evidence shows about improving vagal tone. Vagal tone is one of several autonomic factors that affect sleep; Autonomic Sleep Disruption covers the full picture of how autonomic regulation shapes sleep quality.
What Does Heart Rate Variability During Sleep Measure?
Your heart does not beat at a fixed interval. The time between each beat varies by milliseconds, and that variation is governed by the competing inputs of two branches of the autonomic nervous system: the sympathetic branch (which raises heart rate, increases alertness, and mobilizes energy) and the parasympathetic branch (which slows heart rate, supports digestion, and promotes recovery).
HRV captures this competition. When parasympathetic activity is high, the vagus nerve modulates the heart on a beat-to-beat basis, producing greater variation between heartbeats. When sympathetic activity dominates, the heart rate becomes more uniform and elevated — less variability, higher rate.
During sleep, HRV follows a pattern tied to sleep stages. A 2012 review by Stein and Pu mapped this relationship: during non-rapid eye movement (NREM) slow-wave sleep (the deepest sleep stage), parasympathetic tone predominates, producing lower heart rate and higher HRV in frequencies associated with respiratory sinus arrhythmia — the natural coupling between breathing rhythm and heart rate. During rapid eye movement (REM) sleep, sympathetic activity increases and vagal tone withdraws, reducing HRV in a pattern that resembles waking stress responses.
Consumer wearables estimate HRV using photoplethysmography (PPG) — optical sensors that detect blood volume changes at the wrist or finger. The metrics reported (RMSSD, HRV score, or “recovery”) are proxies for the high-frequency component of HRV, which is primarily vagally mediated. A 2026 analysis of approximately 2 million nocturnal heart rate readings from over 21,000 wearable users confirmed that overnight HRV varies with alcohol consumption, physical activity levels, sleep duration, and sleep timing consistency — and that normative values differ by age and sex (Grosicki et al., 2026).
The distinction matters: a single low HRV reading on one night may reflect alcohol, a late meal, or acute stress. A low overnight HRV trend that persists over weeks reflects a sustained autonomic state — one where parasympathetic recovery is not engaging during sleep.
How Does Vagal Tone Affect the Transition into Sleep?
Falling asleep is not a passive event. It requires an active autonomic transition: the parasympathetic branch takes over from the sympathetic branch. Heart rate declines. Core temperature drops as peripheral vasodilation moves heat away from the body’s core. Breathing rate and depth change. This transition is driven in large part by vagal activity — the parasympathetic output of the vagus nerve to the heart, lungs, and vasculature.
Ma et al. (2024) quantified what happens when this transition is insufficient. In a study of 328 participants — 123 normal sleepers, 69 with insomnia alone, 70 with insomnia plus mild obstructive sleep apnea, and 66 with insomnia plus moderate-to-severe sleep apnea — people with insomnia showed a measurable parasympathetic deficit at sleep onset. Heart rate was elevated (p = 0.004), SDNN (a time-domain measure of overall HRV) was lower (p = 0.003), and pNN20 (a measure of beat-to-beat parasympathetic variation) was reduced (p < 0.001). High-frequency HRV power — the primary spectral index of vagal modulation — was lower in people with insomnia (p < 0.001).
After adjusting for age, sex, BMI, and sleep apnea severity, insomnia independently remained associated with higher heart rate, lower pNN20, and lower high-frequency HRV power. This isolates a core finding: blunted vagal activity during sleep onset is a feature of insomnia itself, not a byproduct of concurrent sleep-disordered breathing.
Joubert et al. (2024) extended this finding beyond insomnia populations studied in sleep labs. In university students, lower overnight parasympathetic activity — measured by HRV during sleep — was associated with higher perceived stress, poorer wellbeing, and reduced daytime functioning. This suggests that the autonomic transition into sleep is not only disrupted in insomnia: it is sensitive to stress load in otherwise healthy adults.
The implication is direct: when the parasympathetic branch does not become dominant at sleep onset, the body enters sleep with sympathetic activity still elevated — meaning heart rate, blood pressure, and arousal-related neural firing remain higher than they should be during the transition into rest.
Does Low Heart Rate Variability During Sleep Indicate Autonomic Imbalance?
A low HRV reading from a wearable provides limited information on its own. The question is whether it represents a transient dip (a hard training day, poor timing of meals, alcohol) or a sustained pattern of reduced parasympathetic engagement.
Wix-Ramos et al. (2024) addressed this by simultaneously applying standard polysomnography (PSG) and the Empatica E4 wrist-worn wearable to 32 people with chronic insomnia and 19 healthy controls. People with insomnia showed elevated heart rate during both wakefulness and all sleep stages — not just at sleep onset — indicating sustained sympathetic activation that does not resolve when sleep begins. HRV was reduced during stage N3 slow-wave sleep in the insomnia group, indicating that the sleep stage where parasympathetic dominance is normally at its peak was autonomically compromised. Peripheral skin temperature did not show the normal nocturnal decrease in the insomnia group, and electrodermal activity (a marker of sympathetic arousal) remained elevated throughout the night.
Laharnar et al. (2020) quantified the heart rate difference over the full sleep period. In a pulse wave analysis study, people with insomnia had median overnight heart rates of 66-67 bpm compared to 58 bpm in healthy sleepers — a sustained elevation across the entire night, not limited to sleep onset or individual sleep stages.
The MESA cohort study (Castro-Diehl et al., 2016) — a large epidemiological analysis of 527 participants — found a dose-response relationship between sleep duration and vagal suppression. Participants sleeping fewer than 6 hours per night had elevated resting heart rate and heightened cardiovascular reactivity. Those sleeping 6-7 hours showed reduced high-frequency HRV, a direct index of cardiac parasympathetic modulation. Participants with insomnia combined with short sleep showed the lowest vagal HRV indices across all subgroups.
A 2025 large-scale analysis of 5,335 electrocardiograms from the Sleep Heart Health Study confirmed that insomnia elevates mean heart rate during sleep — a finding that distinguished the insomnia contribution from sleep apnea and provided a quantifiable nocturnal autonomic marker for insomnia (Martin-Montero et al., 2025).
Across these studies, insomnia is associated with reduced parasympathetic engagement that persists across the full night, not only at sleep onset. Low overnight HRV — especially when it persists over weeks — reflects an autonomic state where the parasympathetic branch is not adequately engaging during sleep.
Can Vagal Tone Be Improved for Better Sleep?
Vagal tone responds to sustained behavioral and physiological inputs. Two categories of evidence are relevant: respiratory-based approaches that modulate vagal activity through the cardiorespiratory coupling between breathing and heart rate, and direct vagal stimulation via transcutaneous devices.
Laborde et al. (2019) tested slow-paced breathing at 6 breaths per minute — a rate that maximizes respiratory sinus arrhythmia, the natural variation in heart rate that occurs with each breathing cycle. In a controlled study, 30 days of this practice improved cardiac vagal activity (measured by RMSSD, the root mean square of successive heartbeat interval differences) and improved subjective sleep quality compared to a control group breathing at their normal rate. The breathing rate matters: at approximately 6 breaths per minute, each inhalation and exhalation cycle aligns with the ~10-second baroreflex oscillation, producing the largest increase in vagal output to the heart.
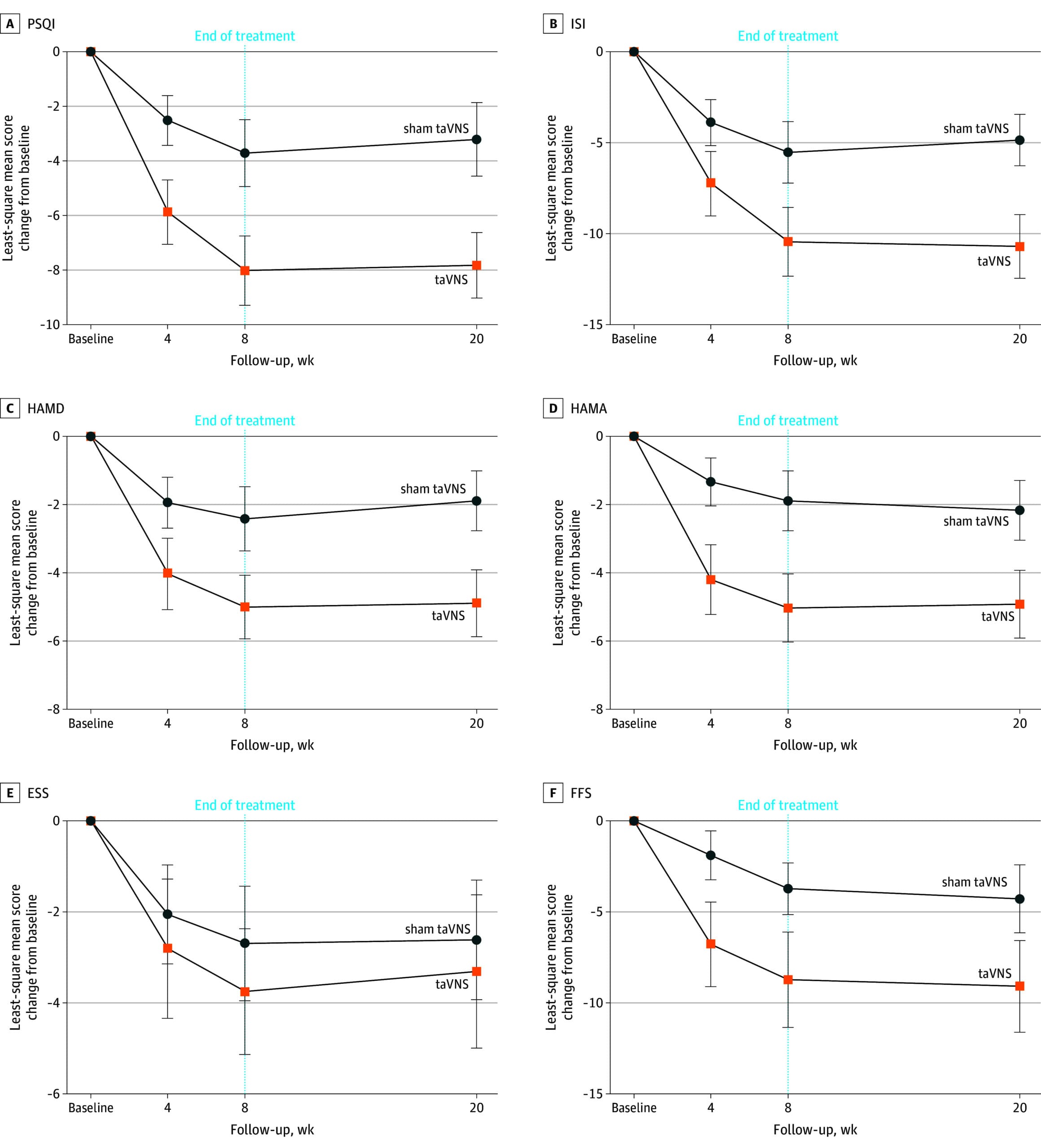
Zhao et al. (2024) conducted a randomized, sham-controlled trial of transcutaneous auricular vagus nerve stimulation (taVNS) for chronic insomnia, published in JAMA Network Open. Transcutaneous auricular vagus nerve stimulation delivers mild electrical pulses to the auricular branch of the vagus nerve — a branch accessible at the outer ear — without surgery or implantation. After 8 weeks, participants receiving active taVNS showed greater improvement in insomnia severity (measured by the Insomnia Severity Index) and sleep quality (measured by the Pittsburgh Sleep Quality Index) compared to sham stimulation. The improvements were sustained at 20-week follow-up, and the completion rate was 94.4%. The rationale: stimulating the auricular vagus branch increases afferent vagal input to the brainstem, which in turn increases parasympathetic output and may reduce the sympathetic hyperarousal associated with chronic insomnia.
Both approaches share a mechanism: they increase parasympathetic (vagal) activity. Slow-paced breathing does this through cardiorespiratory coupling — aligning breath rate with the heart’s baroreflex oscillation. Transcutaneous vagus nerve stimulation does this through direct electrical activation of vagal afferent fibers. Neither replaces addressing the upstream contributors to low vagal tone — chronic stress, inflammation, poor sleep duration — but both provide evidence that the parasympathetic branch responds to targeted input.
Low vagal tone might not be the only autonomic factor affecting your sleep. Parasympathetic insufficiency can overlap with cortisol-driven hyperarousal, GABA receptor impairment, or gut-brain pathway disruption. When multiple causes might be contributing, identifying which ones are active is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
How Do You Know if Your Vagus Nerve Is Underactive?
There is no single test that directly measures vagal tone. Instead, HRV — the high-frequency component in particular — serves as the primary proxy, because beat-to-beat heart rate variation in the respiratory frequency range is driven by vagal input to the sinoatrial node (the heart’s natural pacemaker).
Menuet et al. (2025) recommended that vagal tone assessment should account for breathing rate, because respiratory sinus arrhythmia — the primary contributor to high-frequency HRV — is modulated by respiratory depth and frequency. A high breathing rate can reduce high-frequency HRV even when vagal tone is normal. Some consumer wearables now track respiratory rate alongside HRV, which adds context to overnight readings.
Patterns that suggest reduced vagal tone over time include: low HRV across multiple nights, an elevated average sleeping heart rate (relative to your own baseline), and the absence of a heart rate dip during the first half of the night, when parasympathetic dominance is normally at its peak.
Does Deep Breathing Activate the Vagus Nerve?
The mechanism is respiratory sinus arrhythmia: during inhalation, heart rate accelerates; during exhalation, heart rate decelerates. This deceleration is mediated by vagal output to the heart. Slower breathing extends the exhalation phase, increasing vagal input per breath cycle.
At approximately 6 breaths per minute, the breathing rhythm aligns with the ~10-second baroreflex cycle — a feedback loop between blood pressure sensors in the carotid arteries and vagal output to the heart. This alignment amplifies vagal modulation of heart rate, producing the largest measurable increase in high-frequency HRV. Laborde et al. (2019) demonstrated that sustained practice (30 days) at this rate improved both cardiac vagal activity and self-reported sleep quality.
The rate matters more than the depth. Breathing at 10-12 breaths per minute (a normal resting rate) does not produce the same baroreflex resonance effect. The 6 breaths per minute rate — approximately 5 seconds inhale, 5 seconds exhale — is the range where vagal effects are maximized in the available research.
Can You Measure Vagal Tone at Home?
Wearable devices estimate HRV by measuring pulse wave timing through optical sensors. The resulting metric — typically RMSSD or a proprietary “HRV score” — captures the high-frequency, vagally mediated component of heart rate variability. These estimates correlate with ECG-derived HRV, though absolute accuracy varies between devices and sensor placements (wrist vs. finger vs. chest).
Grosicki et al. (2026) established that a minimum of five consecutive nights of data is needed to produce reliable 7-day HRV estimates from wearable devices. Single-night readings are influenced by too many transient factors — meal timing, alcohol, acute stress, exercise intensity — to reflect underlying vagal tone. The trend over 2-4 weeks is the informative measure, not the nightly fluctuation.
Interpreting wearable HRV data in context means accounting for age (vagal tone declines with age), sex (normative trajectories differ), physical fitness (trained individuals tend to have higher resting vagal tone), and breathing rate during sleep (which modulates the high-frequency HRV component directly).
Does Insomnia Lower Heart Rate Variability?
A 2017 review of 22 studies examining HRV in insomnia populations found that the theoretical model — insomnia involves autonomic hyperarousal with sympathetic excess and reduced vagal tone — is broadly supported but not demonstrated with uniform results across all studies (Dodds et al., 2017). Methodological differences in participant selection, HRV measurement protocols, and data-processing techniques accounted for the inconsistency. The review recommended standardized 24-hour HRV recordings and uniform participant selection criteria as necessary steps to resolve conflicting findings.
Since that review, larger studies with standardized methods have provided stronger evidence. Ma et al. (2024), in the largest controlled comparison of sleep-onset HRV in insomnia to date (n = 328), confirmed that insomnia independently predicts reduced high-frequency HRV and elevated heart rate at sleep onset after controlling for sleep apnea, age, sex, and BMI. Martin-Montero et al. (2025) confirmed elevated nocturnal heart rate in insomnia across 5,335 electrocardiograms.
The bidirectional nature means that addressing the upstream contributors to low vagal tone — chronic stress, inflammation, reduced physical activity, and sleep debt — may improve both HRV and sleep quality, while improving sleep may in turn support parasympathetic recovery.
Related Reading:
- Autonomic Sleep Disruption — the full overview of how autonomic regulation shapes sleep quality
- Can Your Nervous System Get Stuck in Fight or Flight and Ruin Your Sleep? — the cortisol-sleep loop and sympathetic dominance
- What Is Hyperarousal Insomnia? Why You’re Wired but Tired Every Night — the wired-but-tired state and what drives it
- Why Won’t Your Brain Shut Off at Night? The Autonomic Connection — GABA and autonomic regulation at sleep onset
- Why Is Your Rapid Eye Movement Sleep Fragmented? The Brainstem Switch That Controls It — REM architecture and cholinergic pathway
- Does Benadryl Destroy Your Sleep? How Anticholinergic Drugs Suppress REM — medications and REM disruption
- How Your Gut Talks to Your Brain Through the Vagus Nerve — and Why It Matters for Sleep — gut-vagus pathway
- Does Benadryl Degrade Your Sleep? How Anticholinergic Drugs Suppress Rapid Eye Movement Sleep — how anticholinergic medications block the muscarinic receptors REM requires
- How Your Gut Talks to Your Brain Through the Vagus Nerve — and Why It Matters for Sleep — gut bacteria, GABA production, and the vagal pathway to sleep regulation
- Do Antidepressants Suppress Rapid Eye Movement Sleep? — SSRIs, SNRIs, and tricyclics affect sleep architecture
- Can Vagus Nerve Stimulation Devices Improve Insomnia? — clinical trial evidence for taVNS devices
- Does Your Sleep Tracker Accuracy Matter? — what Oura, WHOOP, and Apple Watch get right and wrong
- Is Your Insomnia a Nervous System Problem? — how to tell if autonomic dysregulation is driving sleep disruption
References
Castro-Diehl, C., Diez Roux, A. V., Redline, S., Seeman, T., McKinley, P., Sloan, R., & Shea, S. (2016). Sleep duration and quality in relation to autonomic nervous system measures: The Multi-Ethnic Study of Atherosclerosis (MESA). Sleep, 39(11), 1927-1940. https://pubmed.ncbi.nlm.nih.gov/27568797/
Dodds, K. L., Miller, C. B., Kyle, S. D., Marshall, N. S., & Gordon, C. J. (2017). Heart rate variability in insomnia patients: A critical review of the literature. Sleep Medicine Reviews, 33, 88-100. https://pubmed.ncbi.nlm.nih.gov/28187954/
Grosicki, G. J., et al. (2026). Heart rate variability coefficient of variation during sleep as a digital biomarker that reflects behavior and varies by age and sex. American Journal of Physiology – Heart and Circulatory Physiology. https://pubmed.ncbi.nlm.nih.gov/41309064/
Joubert, M., Beilharz, J. E., Fatt, S., Chung, Y. M., Cvejic, E., Vollmer-Conna, U., & Burton, A. R. (2024). Stress reactivity, wellbeing and functioning in university students: A role for autonomic activity during sleep. Sleep Medicine. https://pubmed.ncbi.nlm.nih.gov/39616430/
Laborde, S., Hosang, T., Mosley, E., & Dosseville, F. (2019). Influence of a 30-day slow-paced breathing intervention compared to social media use on subjective sleep quality and cardiac vagal activity. Journal of Clinical Medicine, 8(2), 193. https://pubmed.ncbi.nlm.nih.gov/30736268/
Laharnar, N., Grote, L., Engleman, H., Grolimund, E., Glos, M., Penzel, T., McNicholas, W. T., & Hedner, J. (2020). Overnight pulse wave analysis to assess autonomic changes in patients with sleep disorders. PLoS ONE, 15(5), e0232589. https://pubmed.ncbi.nlm.nih.gov/32379833/
Ma, Y., et al. (2024). Heart rate variability during sleep onset in patients with insomnia with or without comorbid sleep apnea. Sleep Medicine, 123, 51-57. https://pubmed.ncbi.nlm.nih.gov/39137665/
Martin-Montero, A., et al. (2025). Heart rate variability analysis in comorbid insomnia and sleep apnea (COMISA). Scientific Reports, 15, 18189. https://pubmed.ncbi.nlm.nih.gov/40399494/
Menuet, C., et al. (2025). Expert recommendation on respiratory-related heart rate variability. Physiological Measurement. https://pubmed.ncbi.nlm.nih.gov/40328963/
Stein, P. K., & Pu, Y. (2012). Heart rate variability, sleep and sleep disorders. Sleep Medicine Reviews, 16(1), 47-66. https://pubmed.ncbi.nlm.nih.gov/21658979/
Wix-Ramos, R., et al. (2024). Monitoring differences in the function of the autonomic nervous system in patients with chronic insomnia using a wearable device. Sleep Medicine, 115, 122-130. https://pubmed.ncbi.nlm.nih.gov/38359591/
Zhao, B., et al. (2024). Transcutaneous auricular vagus nerve stimulation for chronic insomnia: A randomized clinical trial. JAMA Network Open, 7(12), e2451217. https://pubmed.ncbi.nlm.nih.gov/39680406/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 12 references cited
