Diphenhydramine is the most widely used over-the-counter sleep aid in the United States. It produces sedation — that part is not in question. The problem is what it does to sleep architecture once you are asleep. Diphenhydramine blocks muscarinic acetylcholine receptors in the brainstem, and those receptors are required for the brain to enter and maintain REM sleep. The result is a night that feels like sleep but lacks the REM stages associated with emotional regulation, memory consolidation, and long-term cognitive health.
This article covers how anticholinergic drugs suppress REM through muscarinic receptor blockade, which drug classes carry this effect, how the trospium comparison proves the mechanism is central (meaning it requires brain penetration), and what the long-term dementia risk data shows. It does not cover the full autonomic model of sleep disruption — for that, see Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It. Medication-induced REM suppression is one of several autonomic causes of sleep disruption; the pillar covers the broader picture.
Why Does Diphenhydramine Suppress Rapid Eye Movement Sleep Instead of Improving Sleep?
The sedation from diphenhydramine comes from one mechanism — histamine H1 receptor blockade. Histamine is a wakefulness-promoting neurotransmitter, and blocking it at the H1 receptor reduces arousal, which is why the drug makes you drowsy. That effect is well-documented and begins within 30-60 minutes of oral dosing.
The REM suppression comes from a different mechanism. Diphenhydramine also binds to muscarinic acetylcholine receptors (M1 and M3 subtypes), and these receptors are part of the brainstem circuitry that initiates and sustains REM sleep. Cholinergic neurons in the pedunculopontine tegmental nucleus (PPT) and laterodorsal tegmental nucleus (LDT) activate cortical and thalamic circuits via muscarinic receptors to produce REM. When diphenhydramine blocks those receptors, REM onset is delayed and total REM time is reduced.
A 2025 expert consensus review (Ariza-Salamanca et al.) synthesized polysomnographic evidence showing that 50 mg of diphenhydramine prolongs REM latency (the time before the first REM episode begins) and reduces REM sleep as a percentage of total sleep time (Ariza-Salamanca et al., 2025). Sleep architecture redistributes toward lighter N1 and N2 stages — stages associated with less restorative function.
This means the drug achieves two things simultaneously: it shortens the time to fall asleep, and it degrades the composition of the sleep that follows. The sedation is real; the sleep architecture it produces is not equivalent to natural sleep.
A 1991 human study by Gillin et al. demonstrated this same mechanism using biperiden, a selective muscarinic antagonist. In 8 healthy volunteers, biperiden produced dose-dependent suppression of REM sleep time and prolongation of REM latency at all three tested doses (2, 4, and 8 mg), showing that muscarinic receptor blockade alone — without histamine involvement — is sufficient to suppress REM in humans (Gillin et al., 1991).
Are Muscarinic Receptors Required for Rapid Eye Movement Sleep?
The pharmacological evidence from drugs like biperiden and diphenhydramine shows that blocking muscarinic receptors suppresses REM. But a more definitive answer comes from genetics.
Niwa et al. (2018) used CRISPR-Cas9 gene editing to create mice lacking muscarinic acetylcholine receptor subtypes — Chrm1 (M1), Chrm3 (M3), or both. Mice missing M1 alone showed reduced REM sleep. Mice missing M3 alone also showed reduced REM sleep. Mice missing both M1 and M3 had virtually no REM sleep at all (Niwa et al., 2018).
M1 and M3 muscarinic receptors are required for REM generation — they are not modulatory. The two subtypes are partially redundant with each other (either one alone can sustain some REM), but together they are necessary for normal REM generation. Without both, the brainstem cannot produce REM sleep.
This has direct implications for any drug with muscarinic receptor binding affinity. Diphenhydramine blocks both M1 and M3 receptors. So do tricyclic antidepressants (amitriptyline, doxepin at higher doses) and bladder antimuscarinics (oxybutynin, tolterodine). Each of these drug classes suppresses REM through the same pathway the Niwa study mapped genetically.
Earlier pharmacological work by Nissen et al. (2006) examined the role of muscarinic receptor activation in REM regulation using RS-86, a selective M1 muscarinic agonist, and donepezil, an acetylcholinesterase inhibitor, in healthy volunteers. The study found that M1 receptor activation shortened REM latency and increased REM density, providing complementary evidence that the muscarinic cholinergic pathway is a primary regulator of REM sleep (Nissen et al., 2006).
Which Medications Suppress Rapid Eye Movement Sleep Through Anticholinergic Activity?
First-generation antihistamines — diphenhydramine (Benadryl), doxylamine (Unisom SleepTabs), and hydroxyzine (Vistaril) — all have muscarinic receptor binding activity in addition to their H1 receptor effects. Muscarinic binding is an inherent property of these drugs’ receptor binding profiles, not a secondary side effect. All three suppress REM sleep when they cross the blood-brain barrier and reach central muscarinic receptors.
Bladder antimuscarinics — oxybutynin (Ditropan), tolterodine (Detrol), and solifenacin (Vesicare) — are prescribed for overactive bladder. Their primary mechanism is blocking muscarinic receptors in the bladder wall to reduce involuntary contractions. But oxybutynin and tolterodine also cross the blood-brain barrier, and once they reach the brainstem, they block the same M1/M3 receptors required for REM generation.
The trospium comparison provides the clearest evidence that central penetration is the determining factor. Diefenbach et al. (2005) compared oxybutynin (which crosses the blood-brain barrier) to trospium (which does not) in a randomized, placebo-controlled sleep study. Oxybutynin suppressed REM sleep. Trospium did not (Diefenbach et al., 2005). Both drugs block muscarinic receptors with comparable affinity — the difference is that trospium cannot reach the brain. This shows that the REM suppression requires brain penetration, not peripheral muscarinic blockade alone.
Tricyclic antidepressants — amitriptyline (Elavil), doxepin (at higher doses), nortriptyline, and imipramine — are among the most potent anticholinergic drugs in common use. Goerke et al. (2014) demonstrated that amitriptyline suppressed REM sleep and impaired sleep-dependent visual discrimination learning in healthy volunteers compared to placebo, providing direct evidence that anticholinergic-mediated REM suppression degrades the cognitive functions that depend on REM (Goerke et al., 2014).
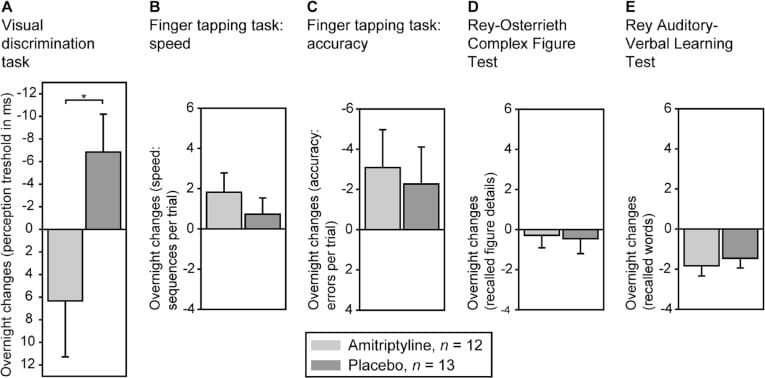
The common thread across all three drug classes is muscarinic receptor blockade in the brainstem. The Niwa 2018 genetic data and the Diefenbach 2005 trospium comparison converge on the same conclusion: any drug that blocks central muscarinic receptors will suppress REM sleep in proportion to its receptor affinity and ability to cross the blood-brain barrier.
Does Long-Term Anticholinergic Use Increase Dementia Risk?
Gray et al. (2015) followed 3,434 community-dwelling adults aged 65 and older in western Washington state for a mean of 7.3 years. Anticholinergic drug use was quantified using total standardized daily doses (TSDDs) calculated from 10 years of pharmacy dispensing records. Participants with cumulative exposure exceeding 1,095 TSDDs (equivalent to approximately three years of daily use) had a 54% higher hazard of all-cause dementia (HR 1.54; 95% CI 1.21-1.96) and a 63% higher hazard of Alzheimer’s disease compared to non-users. The relationship showed a statistically significant dose-response trend (P < 0.001 for trend) — higher cumulative exposure corresponded to higher dementia risk at each exposure level (Gray et al., 2015).
The most commonly implicated drug categories among high-exposure users were tricyclic antidepressants, first-generation antihistamines (including diphenhydramine), and bladder antimuscarinics — the same three drug classes that suppress REM through central muscarinic blockade.
A larger study by Coupland et al. (2019) examined 58,769 dementia cases and 225,574 matched controls from English general practice records. In the highest anticholinergic exposure group (more than 1,095 standardized daily doses), the adjusted odds ratio for dementia was 1.49 (95% CI 1.44-1.54). Drug class-specific analysis showed elevated risk for bladder antimuscarinics (OR 1.65), anticholinergic antidepressants (OR 1.29), and antipsychotics (OR 1.70). The associations persisted even when exposure was lagged by 15-20 years before diagnosis, reducing the likelihood that early dementia drove the medication use rather than the reverse (Coupland et al., 2019).
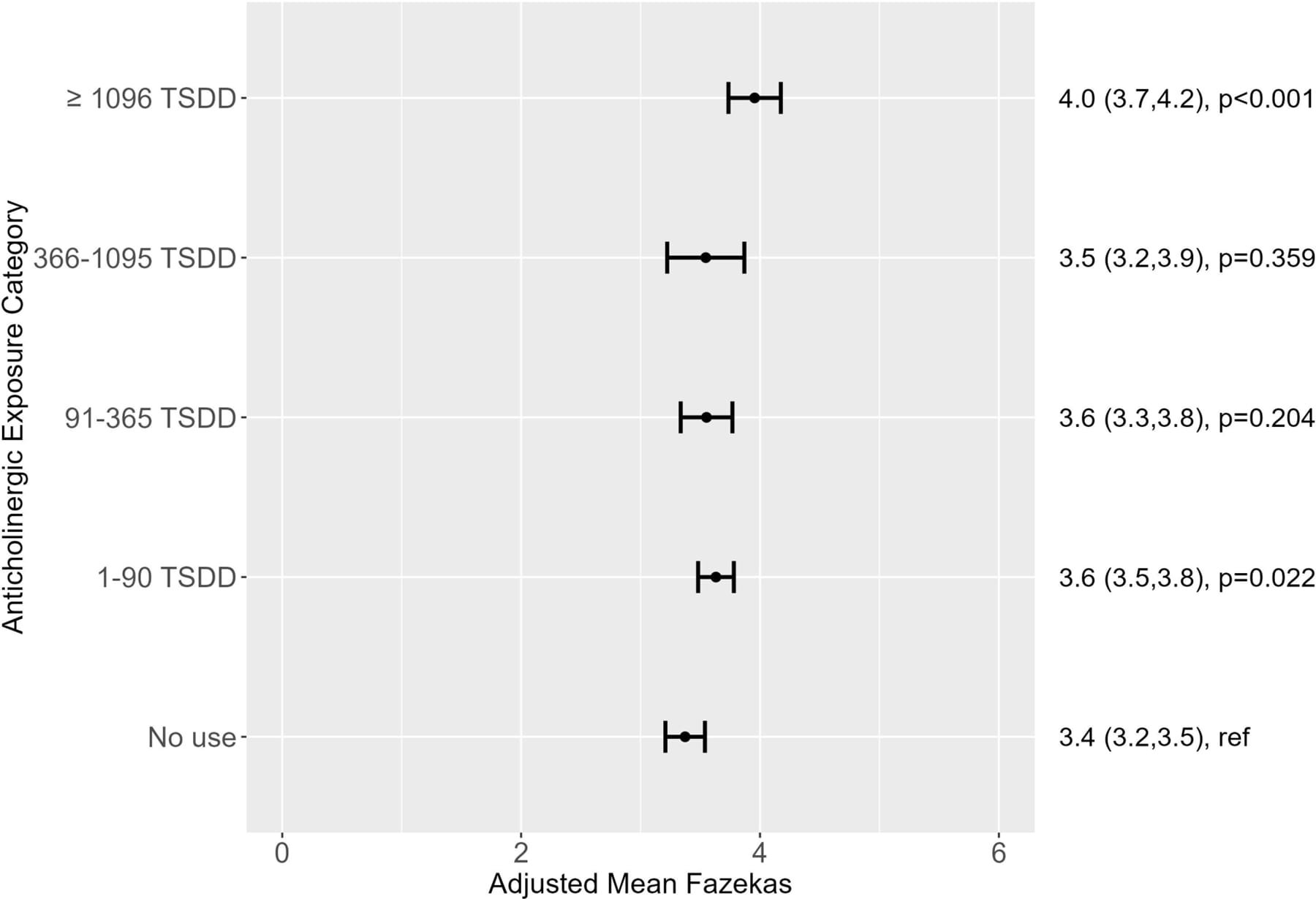
A 2025 brain MRI study by Li et al. added a structural dimension to this evidence. Among 1,043 community-dwelling older adults, those in the highest anticholinergic exposure group had greater white matter hyperintensity burden — a neuroimaging marker of cerebrovascular injury associated with cognitive decline — compared to non-users across all three validated imaging scales (Li et al., 2025).
Whether the dementia risk is driven primarily by chronic REM suppression, direct neurotoxic effects of muscarinic blockade, impaired glymphatic clearance during degraded sleep, or some combination remains an active area of research. A 2025 in-vitro study using human stem cell-derived neurons found that antihistamines including diphenhydramine did not show direct cytotoxicity at tested concentrations — suggesting their cognitive risk may be mediated through indirect mechanisms such as chronic REM suppression rather than direct neuronal damage (Schwarze-Taufiq et al., 2025). A 2023 population study by Gildengers et al. found that any anticholinergic drug use was associated with a 40% higher risk of developing mild cognitive impairment (HR 1.4, p=0.029) in 1,959 cognitively normal older adults (Gildengers et al., 2023).
REM suppression from anticholinergic medications might not be the only factor affecting your sleep. Autonomic hyperarousal, gamma-aminobutyric acid (GABA) receptor impairment, metabolic disruptions, or inflammatory processes might also be contributing. When multiple causes overlap, identifying which ones might be active is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Do You Build Tolerance to Antihistamines for Sleep?
The sedative effect of diphenhydramine depends on histamine H1 receptor blockade, and H1 receptors upregulate rapidly in response to chronic antagonism. The 2025 expert consensus review documented that tolerance to central nervous depression effects occurs within approximately three days, and by day four, performance metrics showed no difference from placebo (Ariza-Salamanca et al., 2025).
The anticholinergic effects — including REM suppression — do not develop tolerance on the same timeline. This creates a situation where continued nightly use provides diminishing sedative benefit while maintaining the same degree of REM disruption and muscarinic receptor blockade.
Are Second-Generation Antihistamines Safer for Sleep?
The structural difference between first- and second-generation antihistamines is central nervous system penetration. Second-generation compounds (cetirizine/Zyrtec, loratadine/Claritin, fexofenadine/Allegra) were designed to block peripheral H1 receptors without crossing the blood-brain barrier at standard doses. They also have lower muscarinic receptor binding affinity than diphenhydramine or doxylamine.
This parallels the trospium principle — drugs that cannot reach central muscarinic receptors do not suppress REM. Cetirizine can produce mild sedation in some individuals because it does cross the blood-brain barrier to a limited degree, but the effect is smaller than with diphenhydramine and the anticholinergic burden is lower.
Can Diphenhydramine Cause Rebound Insomnia?
During chronic diphenhydramine use, the brain compensates for persistent H1 receptor blockade by increasing histamine receptor sensitivity and histamine output. When the drug is removed, the heightened histaminergic activity produces a state of increased arousal that exceeds the original pre-drug baseline. The 2025 expert consensus review identified rebound insomnia as a recognized consequence of discontinuation after regular use (Ariza-Salamanca et al., 2025).
This can create a cycle where the drug appears to be necessary for sleep — not because the underlying sleep difficulty persists, but because the brain has adapted to the drug’s presence.
Does Benadryl Reduce Rapid Eye Movement Sleep?
Polysomnographic data cited in the 2025 expert consensus (Ariza-Salamanca et al.) showed that 50 mg of diphenhydramine reduced REM as a percentage of total sleep time and delayed the onset of the first REM period (Ariza-Salamanca et al., 2025). The mechanism is muscarinic receptor blockade in the brainstem — the same pathway identified by the Niwa 2018 genetic study and the Gillin 1991 pharmacological study using the selective muscarinic antagonist biperiden.
REM sleep is associated with emotional memory processing, procedural memory consolidation, and overnight cognitive maintenance. Reduced REM percentage may not produce noticeable effects on a single night, but chronic reduction over weeks, months, or years of nightly use represents a cumulative change in sleep composition.
Related Reading:
- Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It — how autonomic imbalance disrupts sleep architecture
- Can Your Nervous System Get Stuck in Fight or Flight and Ruin Your Sleep? — the self-reinforcing cortisol-sleep loop
- What Is Hyperarousal Insomnia? Why You’re Wired but Tired Every Night — autonomic mechanics behind the wired-but-tired state
- Why Won’t Your Brain Shut Off at Night? The Autonomic Connection — GABA receptor impairment and sympathetic overactivation
- Why Is Your Rapid Eye Movement Sleep Fragmented? The Brainstem Switch That Controls It — the brainstem circuit and cholinergic pathway
- What Your Overnight HRV Is Telling You About Your Sleep: The Vagal Tone Connection — HRV as a window into parasympathetic recovery
- How Your Gut Talks to Your Brain Through the Vagus Nerve — and Why It Matters for Sleep — gut-vagus-brain pathway
- I’ve Been Taking Benadryl Every Night for Years. Is It Bad? — anticholinergic burden and long-term cognitive risk
- Alcohol Sedates You, Then Wakes You at 3AM — GABA-A rebound and REM disruption
- What Your Overnight Heart Rate Variability Is Telling You About Your Sleep: The Vagal Tone Connection — using HRV as a window into parasympathetic recovery during sleep
- How Your Gut Talks to Your Brain Through the Vagus Nerve — and Why It Matters for Sleep — gut bacteria, GABA production, and the vagal pathway to sleep regulation
- Do Antidepressants Suppress Rapid Eye Movement Sleep? — SSRIs, SNRIs, and tricyclics affect sleep architecture
- Can Vagus Nerve Stimulation Devices Improve Insomnia? — clinical trial evidence for taVNS devices
- Does Your Sleep Tracker Accuracy Matter? — what Oura, WHOOP, and Apple Watch get right and wrong
- Is Your Insomnia a Nervous System Problem? — how to tell if autonomic dysregulation is driving sleep disruption
References
Ariza-Salamanca, D. F., et al. (2025). Expert consensus on the use of diphenhydramine for short-term insomnia: Efficacy, safety, and clinical applications. Journal of Clinical Medicine, 14(10), 3297. https://pmc.ncbi.nlm.nih.gov/articles/PMC12112657/
Coupland, C. A. C., et al. (2019). Anticholinergic drug exposure and the risk of dementia: A nested case-control study. JAMA Internal Medicine, 179(8), 1084-1093. https://pubmed.ncbi.nlm.nih.gov/31233095/
Diefenbach, K., et al. (2005). Effects on sleep of anticholinergics used for overactive bladder treatment in healthy volunteers aged >= 50 years. BJU International, 95(3), 346-349. https://pubmed.ncbi.nlm.nih.gov/15679791/
Gildengers, A., et al. (2023). Anticholinergic drug burden and risk of incident MCI and dementia: A population-based study. Alzheimer Disease and Associated Disorders, 37(1), 44-50. https://pubmed.ncbi.nlm.nih.gov/36706325/
Gillin, J. C., et al. (1991). Dose dependent inhibition of REM sleep in normal volunteers by biperiden, a muscarinic antagonist. Biological Psychiatry, 30(2), 151-156. https://pubmed.ncbi.nlm.nih.gov/1912107/
Goerke, M., et al. (2014). The influence of sleep on the consolidation of visual discrimination learning: Comparing amitriptyline treatment and normal conditions. Journal of Sleep Research, 23(Suppl 1), 41. https://pmc.ncbi.nlm.nih.gov/articles/PMC3985094/
Gray, S. L., et al. (2015). Cumulative use of strong anticholinergics and incident dementia: A prospective cohort study. JAMA Internal Medicine, 175(3), 401-407. https://pubmed.ncbi.nlm.nih.gov/25621434/
Li, K. H., et al. (2025). Cumulative anticholinergic exposure and white matter hyperintensity burden in community-dwelling older adults. Journal of the American Geriatrics Society, 73(4), 1100-1108. https://pubmed.ncbi.nlm.nih.gov/39697086/
Niwa, Y., et al. (2018). Muscarinic acetylcholine receptors Chrm1 and Chrm3 are essential for REM sleep. Cell Reports, 24(9), 2231-2247.e7. https://pubmed.ncbi.nlm.nih.gov/30157420/
Nissen, C., et al. (2006). Differential effects of the muscarinic M1 receptor agonist RS-86 and the acetylcholine-esterase inhibitor donepezil on REM sleep regulation in healthy volunteers. Neuropsychopharmacology, 31(6), 1294-1300. https://pubmed.ncbi.nlm.nih.gov/16192980/
Schwarze-Taufiq, T. A., et al. (2025). Anticholinergic drugs and dementia risk: Using stem cell-based studies to complement pharmacoepidemiology. Alzheimer’s & Dementia: Translational Research & Clinical Interventions, 11(1), e70057. https://pubmed.ncbi.nlm.nih.gov/39911736/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 11 references cited
