The experience of being physically exhausted yet unable to sleep reflects a measurable state where sympathetic activation persists into the hours that should be dominated by parasympathetic recovery. This has consequences beyond poor sleep quality — it affects cardiovascular function, brain health, and long-term longevity outcomes.
This article covers the autonomic nervous system mechanics behind the wired-but-tired state, what brain imaging and EEG data reveal during sleep, how hyperarousal differs from anxiety, and the cardiovascular risk evidence. It does not cover the self-reinforcing cortisol loop or broader vagal tone pathways — for the full autonomic overview, see Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It.
Autonomic hyperarousal is one of several causes of disrupted sleep. Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It covers the broader picture, including vagal tone, GABA pathways, and neuroinflammation.
What is happening in the autonomic nervous system when you feel wired but tired?
The autonomic nervous system has two branches: the sympathetic branch drives arousal — it raises heart rate, sharpens alertness, and mobilizes energy. The parasympathetic branch drives recovery — it lowers heart rate, promotes digestion, and supports the physiological conditions needed for restorative sleep. Sleep onset normally requires the parasympathetic branch to become dominant. In hyperarousal insomnia, that transition is incomplete.
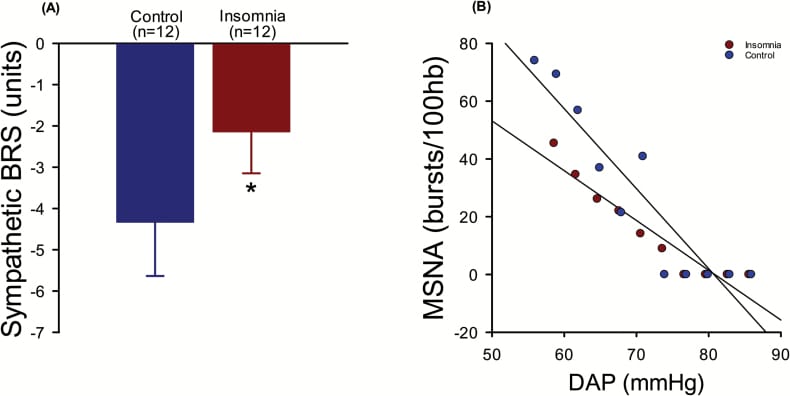
The strongest direct evidence for this comes from Carter et al. (2018), who used microneurography — a technique that records electrical activity from sympathetic nerve fibers during live recordings — to measure sympathetic output in 12 people with chronic insomnia and 12 matched controls. This is the gold-standard method for quantifying sympathetic nervous system activity, and it remains the only study to use it in an insomnia population.
The results identified a measurable impairment in autonomic regulation. Baroreflex sensitivity — the feedback mechanism that detects changes in blood pressure and adjusts sympathetic firing accordingly — was blunted in the insomnia group: -2.1 bursts per 100 heartbeats per mmHg, compared to -4.3 in controls (p < 0.001). In practical terms, the insomnia group's autonomic feedback loop was roughly half as effective at dampening sympathetic activity when it should have been winding down.
During a cold pressor stress test (hand submerged in ice water), sympathetic nerve activity increased by 127% in the insomnia group versus 52% in controls (p = 0.02). Systolic blood pressure reactivity was also elevated: a 21 mmHg increase in the insomnia group versus 14 mmHg in controls (p = 0.04). The insomnia group’s nervous system responded to the same stressor with more than double the sympathetic output.
An important nuance: resting baseline sympathetic nerve activity did not differ between the two groups. The impairment was in regulation — the ability to dampen sympathetic activation once triggered — rather than in tonic output at rest. This distinction matters because it means the wired-but-tired state is not about chronically elevated baseline arousal. It is about an impaired ability to return to baseline after activation.
Riemann et al. (2010) placed this autonomic profile within a broader framework, synthesizing evidence across six levels — autonomic, neuroendocrine, neuroimmunological, electrophysiological, neuroimaging, and behavioral — all converging on the same conclusion: insomnia is a disorder of 24-hour hyperarousal. The elevated cortisol, increased brain glucose metabolism during sleep, and heightened beta-wave EEG activity documented across studies all point to a nervous system that does not downregulate, whether the person is lying in bed or sitting at their desk during the day.
Does the brain stay in a wake-like state even during sleep?
Polysomnography (PSG) measures brain wave activity during sleep using electrodes placed on the scalp. Different sleep stages produce distinct electrical patterns: deep sleep generates slow delta waves (0.5-4 Hz), while wakefulness produces faster alpha (8-13 Hz) and beta (13-30 Hz) waves. In hyperarousal insomnia, these boundaries blur.
Di Marco et al. (2025) pooled PSG data from three independent datasets — 1,710 people with insomnia and 1,455 without — making this the largest analysis of sleep architecture in insomnia to date. The study found that people with insomnia had elevated relative alpha power during N1 (light sleep) and increased theta activity during wake periods, while delta power — the hallmark of deep, restorative sleep — was decreased. The brain was producing wake-like electrical patterns during stages that should be dominated by slower, deeper oscillations.
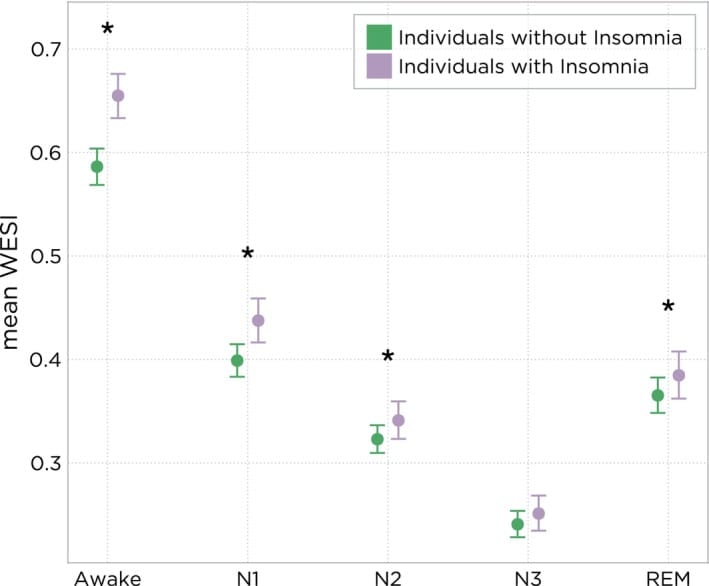
The researchers developed a machine-learning metric called the Wake Electroencephalographic Similarity Index (WESI), which scores how closely the brain’s electrical activity during each sleep stage resembles wakefulness. A score closer to 1.0 means the brain activity looks more like being awake; a score closer to 0 means it looks like sleep. In people with insomnia, WESI was elevated during wakefulness, N1, N2, and REM — four of five measured stages — with differences reaching statistical significance (p < 0.05). The brain was not fully transitioning into sleep-appropriate electrical states.
Sleep spindles — brief bursts of 11-15 Hz activity generated by the thalamus — normally protect sleep by blocking external sensory input from reaching the cortex. Di Marco et al. found that spindle density was reduced and spindle dispersion was increased in the insomnia group. This means fewer protective bursts were being generated, and the ones that did occur were more scattered and less coordinated. The thalamocortical circuits responsible for maintaining stable sleep were measurably impaired.
The study also quantified wake-transition probability — the likelihood of transitioning from any sleep stage into wakefulness. People with insomnia had elevated wake-transition probability across all sleep stages, with increases ranging from 0.6% to 14.9% depending on the stage (all p < 0.05). The highest increase was from N1 to wakefulness: a 14.9% greater probability of waking up from light sleep compared to controls.
Dressle and Riemann (2023) reviewed the broader evidence base for cortical hyperarousal and found that high-frequency beta and gamma EEG activity during NREM sleep is the finding replicated with the greatest consistency across insomnia studies. This elevated fast-wave activity reflects cortical neurons firing at rates characteristic of wakefulness during a period when they should be producing slower, synchronized oscillations. The subjective experience of “sleeping but not resting” has a measurable neural correlate.
Is hyperarousal the same as anxiety?
Anxiety involves conscious worry, fear, or apprehension — a cognitive and emotional experience the person is aware of. Hyperarousal, as measured in insomnia research, includes physiological markers that function below conscious awareness: elevated heart rate, reduced heart rate variability, blunted baroreflex sensitivity, increased sympathetic nerve firing. A person can have measurable autonomic hyperarousal without feeling anxious.
Dressle and Riemann (2023) organized the evidence into three domains. Cognitive-emotional hyperarousal — insomnia-related worry, rumination, and attentional bias toward sleep-related cues — has the broadest and best-replicated evidence base across studies. Cortical hyperarousal — elevated beta and gamma EEG activity during NREM sleep — is the physiological marker replicated with the greatest consistency. Physiological hyperarousal — autonomic markers like heart rate and HRV — shows more variable results across studies, though this variability may reflect methodological differences rather than a genuine absence of the effect.
The distinction matters because cognitive-emotional hyperarousal responds well to cognitive behavioral approaches. Physiological hyperarousal may not. If the autonomic component is the primary driver, addressing only the cognitive component leaves the physiological state unchanged.
Ma et al. (2024) provided direct evidence for this distinction. In a study of 328 participants — 123 normal sleepers and 205 with insomnia, stratified by whether they also had sleep apnea — the insomnia group showed higher heart rate (p = 0.004), lower SDNN (a measure of overall heart rate variability; p = 0.003), and reduced pNN20 (a measure of beat-to-beat parasympathetic variation; p < 0.001) during the sleep-onset period. These differences persisted after adjusting for demographic and health-related confounders, including anxiety status. The autonomic profile was specific to insomnia, independent of anxiety diagnosis.
The authors described these altered heart rate dynamics as a potential “physiological biomarker for insomnia during bedtime wakefulness.” The parasympathetic withdrawal measured at sleep onset — the vagal tone dropping when it should be rising — reflects an autonomic state, not a psychological one.
The 2010 Riemann review framed this through the 3P model: predisposing factors (genetic or temperamental tendency toward heightened arousal), precipitating events (a period of stress, loss, or disruption that triggers insomnia), and perpetuating factors (conditioned arousal at bedtime, sleep-related anxiety, behavioral patterns that maintain the hyperaroused state). Under this model, anxiety can be a precipitating factor — it can trigger the onset of insomnia — but the hyperarousal that maintains it is physiological and can persist after the original anxiety resolves.
Does sleep fragmentation from hyperarousal carry cardiovascular risk?
The cardiovascular consequences of hyperarousal insomnia extend beyond nighttime discomfort. The autonomic profile documented in people with insomnia — impaired baroreflex, exaggerated sympathetic stress reactivity, parasympathetic withdrawal — is the same profile associated with elevated cardiovascular risk in other research contexts.
Chouchou et al. (2013) examined this in the PROOF-SYNAPSE study, a large population-based cohort of 780 healthy elderly participants (mean age 68.7 years, 57.4% female) with no pre-existing sleep disorders or cardiac disease. The researchers measured sleep fragmentation using an autonomic arousal index derived from EEG recordings, then tracked its relationship to blood pressure.
Participants with the highest sleep fragmentation had an odds ratio of 1.70 for hypertension (p = 0.036) — a 70% increase in odds — after controlling for BMI, sleep apnea, and other confounders. The mechanistic pathway was traceable through heart rate variability analysis: fragmented sleepers showed higher nocturnal and diurnal low-frequency HRV power and elevated LF/HF ratios, indicating sympathetic dominance that persisted from nighttime into daytime hours.
A finding with important implications: it was autonomic arousals (sympathetic activation events) rather than cortical arousals (EEG-level awakenings) that drove the blood pressure association. This distinction means the cardiovascular-relevant pathway is sympathetic nervous system activation — not the conscious experience of waking up. A person can experience repeated sympathetic arousals during sleep without being aware of them, and still accumulate cardiovascular risk.
Carter et al. (2018) connected this to the insomnia-specific autonomic profile. The combination of blunted baroreflex sensitivity and exaggerated sympathetic stress reactivity they documented provides a plausible mechanism for the estimated 45% elevation in cardiovascular disease risk observed in epidemiological studies of chronic insomnia. The baroreflex normally acts as a buffer — when blood pressure rises, it triggers the sympathetic nervous system to reduce firing, bringing pressure back down. When that buffer is impaired, blood pressure excursions go unchecked, and the cumulative effect over years of fragmented sleep is sustained hypertension.
The PROOF-SYNAPSE finding is notable because it was observed in healthy people with no identified sleep disorders — though the cohort was elderly (mean age 68.7), so the magnitude of these effects in younger populations remains to be established. This suggests that even subclinical sleep fragmentation can drive sustained autonomic imbalance with measurable cardiovascular consequences.
Hyperarousal insomnia might not be the only factor disrupting your sleep. Autonomic imbalance can overlap with cortisol imbalance, metabolic disruptions, hormonal changes, or inflammatory processes that compound one another. When multiple causes are active, the wired-but-tired experience might reflect several of these mechanisms at once.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Can cognitive behavioral therapy for insomnia address hyperarousal?
CBT-I targets the perpetuating factors in the 3P model: conditioned arousal at bedtime, catastrophic thinking about sleep, and behavioral patterns like excessive time in bed. It is effective for the cognitive-emotional domain of hyperarousal, where the evidence base is broadest.
The physiological domain is less responsive. Dressle and Riemann (2023) noted that while cortical hyperarousal markers (elevated beta/gamma EEG) tend to improve with CBT-I, the autonomic markers — heart rate, HRV, baroreflex function — show more variable responses across studies. For people whose hyperarousal is primarily physiological rather than cognitive, CBT-I may be a necessary but not sufficient approach.
Can consumer wearables detect hyperarousal?
Ma et al. (2024) demonstrated that heart rate variability during the sleep-onset period can distinguish people with insomnia from normal sleepers. The metrics that showed the strongest separation — heart rate, SDNN, and pNN20 — are measured by many modern wrist-worn devices, though consumer-grade accuracy varies.
Wix-Ramos et al. (2024) went further by simultaneously recording polysomnography and an Empatica E4 wrist wearable in 32 people with insomnia and 19 controls. The wearable detected elevated heart rate throughout sleep, increased electrodermal activity (a marker of sympathetic arousal), and impaired thermoregulatory cooling (skin temperature did not show the normal nocturnal decline) in the insomnia group. These differences were detectable across all sleep stages.
Trends over multiple nights are more informative than single-night readings. A consistently elevated resting heart rate at sleep onset, persistently low HRV during the first hour of sleep, and absence of the normal nighttime temperature decline can collectively indicate an autonomic profile consistent with hyperarousal.
Does chronic fight-or-flight activation cause permanent changes?
Riemann et al. (2010) framed this through predisposing, precipitating, and perpetuating factors. A person with a genetic predisposition toward heightened arousal encounters a stressor that triggers insomnia. If the insomnia persists, conditioned arousal and behavioral changes perpetuate the hyperaroused state. Over time, what began as a temporary stress response can become a more stable trait — the nervous system recalibrates to a higher baseline of sympathetic activity.
Dressle and Riemann (2023) described hyperarousal as functioning “simultaneously as a state marker of active insomnia and as a vulnerability trait.” This means it can be both the current state (the person is hyperaroused right now) and a predisposition (the person is more likely to become hyperaroused again after recovery). The trait component does not mean irreversible damage — it means the threshold for re-triggering hyperarousal may be lower than in someone without that predisposition.
What does fight-or-flight feel like when falling asleep?
Normal sleep onset involves a coordinated set of autonomic changes: heart rate decreases, core body temperature drops (facilitated by peripheral vasodilation that radiates heat through the hands and feet), skin conductance decreases, and muscle tension releases. These changes are driven by increasing parasympathetic tone and decreasing sympathetic activity.
Wix-Ramos et al. (2024) documented what happens when this process is impaired. In their insomnia group, skin and body temperature did not show the normal nocturnal decline — thermoregulatory cooling, which is mediated by parasympathetic vasodilation, was blunted. Electrodermal activity (a measure of sweat gland activity driven by sympathetic nerves) remained elevated. Heart rate stayed higher than in controls across all sleep stages.
Ma et al. (2024) confirmed that the parasympathetic withdrawal is measurable during the specific window of bedtime wakefulness — the minutes when a person is lying in bed preparing to sleep. Heart rate was higher, overall HRV (SDNN) was lower, and beat-to-beat parasympathetic variation (pNN20) was reduced in the insomnia group. The body was in a physiological state of readiness rather than a state of recovery — exhausted, but autonomically unable to initiate the transition into sleep.
Related Reading:
- Autonomic Sleep Disruption: What It Is, How It Fragments Sleep, and How to Recognize It — full overview of how autonomic imbalance disrupts sleep architecture, including vagal tone, GABA pathways, and neuroinflammation
- Can Your Nervous System Get Stuck in Fight or Flight and Ruin Your Sleep? — the self-reinforcing cortisol loop and how chronic sympathetic activation perpetuates insomnia
- Why Won’t Your Brain Shut Off at Night? The Autonomic Nervous System Connection — neural mechanisms behind racing thoughts and inability to disengage at bedtime
- Does Benadryl Destroy Your Sleep? How Anticholinergic Drugs Suppress REM Sleep — how anticholinergic medications suppress REM sleep by blocking acetylcholine
- What Your Overnight HRV Is Telling You About Your Sleep: The Vagal Tone Connection — using heart rate variability as a window into parasympathetic recovery during sleep
- How Your Gut Talks to Your Brain Through the Vagus Nerve — and Why It Matters for Sleep — the gut-brain-vagus axis and its role in autonomic regulation during sleep
- Why Is Your REM Sleep Fragmented? The Brainstem Switch That Controls Dream Sleep — how brainstem circuits regulate transitions between REM and non-REM sleep
- Is It Just Stress? 4 Reasons You’re Waking at 3 A.M. — common causes of early-morning waking, including cortisol, blood sugar, and autonomic factors
- Alcohol Sedates You, Then Wakes You at 3AM — how alcohol disrupts sleep architecture and triggers sympathetic rebound in the second half of the night
- Why Won’t Your Brain Shut Off at Night? The Autonomic Connection — how GABA receptor impairment drives the sympathetic overactivation that prevents sleep
- Why Is Your Rapid Eye Movement Sleep Fragmented? The Brainstem Switch That Controls It — the brainstem circuit that controls dream sleep and which medications suppress it
- Does Benadryl Degrade Your Sleep? How Anticholinergic Drugs Suppress Rapid Eye Movement Sleep — how anticholinergic medications block the muscarinic receptors REM requires
- What Your Overnight Heart Rate Variability Is Telling You About Your Sleep: The Vagal Tone Connection — using HRV as a window into parasympathetic recovery during sleep
- How Your Gut Talks to Your Brain Through the Vagus Nerve — and Why It Matters for Sleep — gut bacteria, GABA production, and the vagal pathway to sleep regulation
- Do Antidepressants Suppress Rapid Eye Movement Sleep? — SSRIs, SNRIs, and tricyclics affect sleep architecture
- Can Vagus Nerve Stimulation Devices Improve Insomnia? — clinical trial evidence for taVNS devices
- Does Your Sleep Tracker Accuracy Matter? — what Oura, WHOOP, and Apple Watch get right and wrong
- Is Your Insomnia a Nervous System Problem? — how to tell if autonomic dysregulation is driving sleep disruption
References
Carter, J. R., Grimaldi, D., Engstrom, J., Infante, M. E., Tierney, B. T., & Van Cauter, E. (2018). Assessment of sympathetic neural activity in chronic insomnia: Evidence for elevated cardiovascular risk. Sleep, 41(6), zsy048. https://pubmed.ncbi.nlm.nih.gov/29522186/
Chouchou, F., Pichot, V., Pepin, J. L., Tamisier, R., Celle, S., Maudoux, D., Garcin, A., Levy, P., Barthelemy, J. C., & Roche, F. (2013). Sympathetic overactivity due to sleep fragmentation is associated with elevated diurnal systolic blood pressure in healthy elderly subjects: The PROOF-SYNAPSE study. European Heart Journal, 34(28), 2122-2131. https://pubmed.ncbi.nlm.nih.gov/23756334/
Di Marco, T., Kalkanis, A., Spaey, J., Testelmans, D., Buyse, B., & Van Huffel, S. (2025). Hyperarousal features in the sleep architecture of individuals with and without insomnia. Journal of Sleep Research, 34(1), e14256. https://pubmed.ncbi.nlm.nih.gov/38853521/
Dressle, R. J., & Riemann, D. (2023). Hyperarousal in insomnia disorder: Current evidence and potential mechanisms. Journal of Sleep Research, 32(6), e13928. https://pubmed.ncbi.nlm.nih.gov/37183177/
Ma, Y., Mullington, J. M., Wayne, P. M., & Yeh, G. Y. (2024). Heart rate variability during sleep onset in patients with insomnia with or without comorbid sleep apnea. Sleep Medicine, 122, 92-98. https://pubmed.ncbi.nlm.nih.gov/39137665/
Riemann, D., Spiegelhalder, K., Feige, B., Voderholzer, U., Berger, M., Perlis, M., & Nissen, C. (2010). The hyperarousal model of insomnia: A review of the concept and its evidence. Sleep Medicine Reviews, 14(1), 19-31. https://pubmed.ncbi.nlm.nih.gov/19481481/
Wix-Ramos, R., Gálvez-Goicuría, J., Verona-Almeida, M., Ayala, J. L., López-Viñas, L., Rocío-Martín, E., Luque-Cárdenas, C., Quintas, S., Gago-Veiga, A., & Pagán, J. (2024). Monitoring differences in the function of the autonomic nervous system in patients with chronic insomnia using a wearable device. Sleep Medicine, 115, 122-130. https://pubmed.ncbi.nlm.nih.gov/38359591/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
