Many men in their 40s and 50s notice their sleep deteriorating — more awakenings, less deep sleep, mornings that feel unrestored — and attribute it to stress or aging. The term “andropause” is less recognized than menopause, but the hormonal changes are measurable and well-documented: testosterone, DHEA, and growth hormone all decline on overlapping timelines.
This article covers what andropause is, which hormones decline together, how that compound decline affects sleep architecture, and what the evidence shows about DHEA and sleep. It does not cover testosterone-sleep interactions in isolation (covered separately) or the cortisol-testosterone ratio (covered separately). Andropause is one of several hormonal mechanisms that disrupt sleep in men — the full picture is in Hormonal Sleep Disruption in Men.
What Is Andropause and When Does It Start?
Testosterone decline begins around age 30 and proceeds at roughly 1-2% per year. In a prospective cohort of 1,312 men aged 65 and older from the MrOS study, lower testosterone was associated with lower sleep efficiency, more nocturnal awakenings, and less slow-wave sleep — with the relationship measurable on polysomnography (Barrett-Connor et al., 2008).
DHEA follows a steeper age curve. By age 50, DHEA production is roughly 50% of its peak; by ages 70-80, it has declined to 10-20% of peak levels. Growth hormone declines at approximately 14% per decade from young adulthood. These three declines — sometimes described as andropause, adrenopause, and somatopause — occur simultaneously, compounding each other.
Tanji et al. (2025) studied 1,489 Japanese men with a mean age of 49.8. Among this cohort, 46.5% met criteria for probable insomnia, and 8.4% had severe andropause as measured by the Aging Males’ scale. After multivariable adjustment, insomnia showed an odds ratio of 9.47 for severe andropause — higher than any other behavioral factor, including smoking and physical inactivity. The OR was even stronger for the somatic subdomain (12.86), indicating that insomnia is not a peripheral accompaniment to andropause but its strongest behavioral predictor.
The gradual onset is part of why men miss the connection. There is no equivalent of the menopause transition — no abrupt change that marks a before and after. Instead, sleep quality declines over years, and each year’s deterioration feels normal relative to the one before it.
What Are the Sleep Effects of Andropause?
Gonzalez et al. (2018) followed 78 men receiving androgen deprivation therapy for prostate cancer, 99 men who had undergone surgery only, and 108 men with no cancer history. Men on androgen deprivation therapy demonstrated worse subjective sleep disturbance throughout the 12-month study period (p ≤ .03) and greater objectively measured wake after sleep onset (p < .01). They also experienced more nocturia episodes (p < .001) and greater hot flash interference (p < .001). Mediation analyses identified nocturia as a partial mediator of objective sleep disruption, and hot flash interference as a partial mediator of subjective sleep disturbance — making these the pathways through which androgen removal disrupts sleep, paralleling the vasomotor disruption women experience during menopause.
Morssinkhof et al. (2023) provided causal evidence for testosterone’s effect on sleep architecture. In a controlled prospective study of 73 transgender individuals, transmasculine participants initiating testosterone therapy showed measurable changes within three months: slow-wave sleep decreased by 7 minutes (95% CI: −12 to −3), REM sleep latency decreased by 39% (95% CI: −52% to −22%), and REM sleep duration increased by 17 minutes (95% CI: 7 to 26). Transfeminine participants receiving estrogen plus antiandrogens showed no sleep architecture changes. The selectivity of the effect to testosterone — and not estrogen — indicates that androgens directly modulate sleep stage distribution.
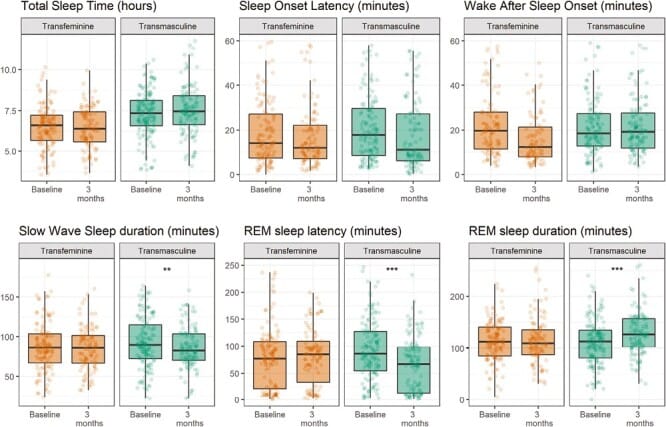
Barrett-Connor et al. (2008) documented the dose-dependent relationship in older men: among 1,312 men aged 65+, lower testosterone was associated with lower sleep efficiency, more awakenings, and less slow-wave sleep. DHEA supplementation has been shown to increase REM sleep duration in controlled studies (Friess et al., 1995), suggesting that as DHEA declines with age, REM architecture may weaken in parallel.
The accumulation is gradual. Unlike menopause, where sleep disruption can be acute and recognizable, andropause sleep disruption worsens over years. Many men adapt to the deterioration — adjusting their expectations of what sleep should feel like — making the connection to hormonal change harder to identify.
Why Do Men Sleep Worse After 50?
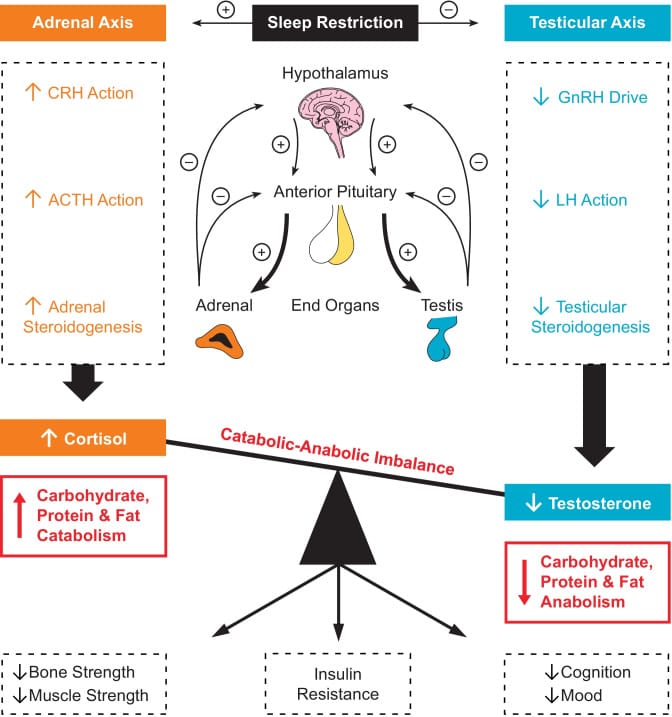
These declines reinforce each other. Low testosterone reduces the availability of neurosteroids like androstanediol that modulate GABA-A receptors — the same receptors involved in sleep maintenance. Less slow-wave sleep reduces both testosterone and growth hormone production, since both hormones are released predominantly during deep sleep. Cortisol elevation, which tends to accompany aging and stress, suppresses testosterone further.
Liu and Reddy (2022) synthesized the evidence on this bidirectional relationship: sleep loss and shorter sleep duration are associated with lower morning, afternoon, and 24-hour testosterone alongside higher afternoon cortisol. The testosterone increase that normally occurs during sleep is sleep-dependent — not circadian-driven — and requires at least 3 hours of sleep with normal architecture. When sleep is fragmented or shortened, the hormonal recovery window narrows.
Agrawal et al. (2024) quantified the relationship between sleep disorders and testosterone deficiency in a large U.S. claims database of men aged 40-70. After propensity score matching, circadian rhythm disorders carried an odds ratio of 2.63 for testosterone deficiency — higher than insomnia (OR = 1.74) or sleep apnea (OR = 1.66). The graded hierarchy suggests that disruption of circadian timing is more damaging to androgen production than fragmented sleep alone.
Hernández-Pérez et al. (2024) found an age-dependent pattern in NHANES data (n = 8,748): in men aged 41-64, longer sleep duration was associated with lower testosterone (OR = 2.03). The authors propose this may represent a compensatory response — men with poor sleep architecture sleep longer to compensate, but the extended time in bed does not restore the hormonal output that depends on normal sleep staging.
Much of what men accept as “I don’t sleep like I used to” is measurable hormonal decline, not an inevitable consequence of aging. The earlier the contributing factors are identified, the less compounded the decline becomes.
Does Dehydroepiandrosterone Help With Sleep in Men?
Friess et al. (1995) administered DHEA to healthy participants and found that it increased REM sleep duration and elevated EEG sigma power — an indicator of sleep spindle activity associated with sleep stability. The mechanism appears to involve DHEA’s conversion to neuroactive steroids, including metabolites that act on GABA-A receptors (promoting inhibitory tone) and NMDA receptors.
DHEA’s decline is steep. By age 50, production is approximately 50% of peak. By 70-80, it may be only 10-20% of peak. This decline runs parallel to testosterone decline but follows a different trajectory — DHEA originates from the adrenal glands rather than the testes, so it represents an independent hormonal input into sleep regulation.
This distinction matters for men on testosterone replacement therapy who still experience poor sleep. Testosterone replacement addresses testicular decline but does not restore DHEA. If DHEA-mediated neurosteroid support is part of the sleep disruption picture, testosterone alone may be insufficient.
The evidence base for DHEA supplementation and sleep is smaller than for testosterone. Timing may matter — some evidence suggests evening dosing may be more effective than morning dosing — but the data are limited. What is established is the mechanism: DHEA functions as a neurosteroid precursor with direct effects on the receptor families that govern sleep maintenance and architecture.
Andropause rarely presents as a single-hormone problem. Testosterone, DHEA, growth hormone, and cortisol all change together, and weakened GABA receptor function might be compounding the picture. Many men experiencing deteriorating sleep after 50 have multiple contributing mechanisms — autonomic, metabolic, inflammatory, or circadian factors might also be involved. Identifying which causes might be active is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Is Waking Up at 3am a Sign of Andropause?
The 2-4am waking window coincides with the nadir of the circadian temperature rhythm and the transition from slow-wave-dominant to REM-dominant sleep. Any disruption to either — whether from cortisol elevation, reduced GABAergic tone, or hormonal changes — can surface as waking during this period. The distinguishing feature of andropause-related waking is the cluster of co-occurring changes: if 3am waking is accompanied by night sweats, reduced muscle recovery, increased abdominal adiposity, and lower daytime energy, hormonal decline becomes a more likely contributor. A single wake event in isolation does not indicate andropause.
How Long Does Andropause Insomnia Last?
Menopause involves a transition — estrogen declines, stabilizes, and the acute vasomotor disruption often resolves within 5-10 years. Andropause has no equivalent stabilization point. Testosterone, DHEA, and growth hormone continue to decline decade over decade. The rate of worsening depends on how many hormonal axes are declining and whether compounding factors — adiposity, inflammation, chronic stress — are present. Addressing the hormonal contributors can slow or partially reverse the sleep deterioration, but the underlying age-related hormonal trajectory continues.
How Do You Address Insomnia Caused by Andropause?
Identifying which hormones are involved determines the approach. Testosterone, DHEA, growth hormone, and cortisol each affect sleep through different pathways — testosterone and DHEA through neurosteroid-mediated GABA-A receptor modulation, growth hormone through its coupling with slow-wave sleep, and cortisol through its antagonism of both sleep continuity and testosterone production. Resistance training can support testosterone and growth hormone. Managing body composition addresses the adiposity pathway that Barrett-Connor et al. (2008) identified as a mediator. DHEA may support REM architecture when it is part of the decline. The approach with the strongest evidence base is identifying which mechanisms are contributing and addressing those directly rather than supplementing broadly.
What Is the Best Sleep Supplement for Men Over 50?
Each supplement addresses a narrow slice of the overall picture. Melatonin supports circadian timing but does not address the slow-wave sleep and REM architecture changes driven by testosterone and DHEA decline. Magnesium supports GABA-A receptor function but does not restore hormonal levels. DHEA may support REM sleep through neurosteroid conversion (Friess et al., 1995), but the evidence base is limited. When multiple hormonal declines are compounding — as they typically do in andropause — addressing the upstream contributors (sleep architecture, body composition, stress load, hormonal status) is more effective than adding individual supplements without understanding which mechanisms are involved.
Related Reading
- Hormonal Sleep Disruption in Men — How testosterone, cortisol, growth hormone, and GABA interact to fragment sleep in men
- Can Low GABA Cause Waking Up at 3am? — The GABAergic mechanism behind 3am waking
- What Are the Signs of Low GABA at Night? — How to recognize impaired GABAergic function
- Do GABA Supplements Help You Stay Asleep Through the Night? — Trial evidence for oral GABA and the blood-brain barrier question
- Does GABA Affect Testosterone and Sleep in Men? — How testosterone-derived neurosteroids modulate GABA-A receptors
- How Do You Increase GABA Levels Naturally for Better Sleep? — Exercise, yoga, fermented foods, and gut health for GABA support
- Does Low Testosterone Cause Sleep Problems in Men? — The bidirectional testosterone-sleep feedback loop
- Can a Cortisol Spike Wake You Up at 3am? — Cortisol-testosterone axis and nocturnal waking
- Does Growth Hormone Decline Affect Your Sleep After 40? — GH-SWS coupling and age-related decline
- Can Belly Fat Lower Your Testosterone and Disrupt Your Sleep? — How visceral fat suppresses testosterone through aromatase and disrupts sleep
- Can Ultra-Processed Food Lower Testosterone and Disrupt Sleep? — How endocrine-disrupting chemicals in processed food suppress testosterone and degrade sleep
- Can Inflammation Suppress Testosterone and Disrupt Your Sleep? — How inflammatory cytokines suppress testosterone production at the brain and testicular level
- Does Testosterone Replacement Therapy Affect Sleep? — How TRT affects sleep quality, its interaction with sleep apnea, and the role of body weight
References
Agrawal, P., et al. (2024). Sleep disorders are associated with testosterone deficiency and erectile dysfunction — a U.S. claims database analysis. International Journal of Impotence Research, 36, 315-321. https://pubmed.ncbi.nlm.nih.gov/36473958/
Barrett-Connor, E., et al. (2008). The association of testosterone levels with overall sleep quality, sleep architecture, and sleep-disordered breathing. Journal of Clinical Endocrinology & Metabolism, 93(7), 2602-2609. https://pubmed.ncbi.nlm.nih.gov/18413429/
Friess, E., et al. (1995). DHEA administration increases rapid eye movement sleep and EEG power in the sigma frequency range. American Journal of Physiology, 268(1), E107-E113. https://pubmed.ncbi.nlm.nih.gov/7840167/
Gonzalez, B. D., et al. (2018). Sleep disturbance in men receiving androgen deprivation therapy for prostate cancer: The role of hot flashes and nocturia. Cancer, 124(3), 499-506. https://pubmed.ncbi.nlm.nih.gov/29072790/
Hernández-Pérez, J. G., et al. (2024). Association of sleep duration and quality with serum testosterone concentrations among men and women: NHANES 2011-2016. Andrology, 12(3), 487-497. https://pubmed.ncbi.nlm.nih.gov/37452666/
Liu, P. Y., & Reddy, R. T. (2022). Sleep, testosterone and cortisol balance, and ageing men. Reviews in Endocrine and Metabolic Disorders, 23, 1323-1339. https://pubmed.ncbi.nlm.nih.gov/36152143/
Morssinkhof, M. W. L., et al. (2023). Influence of sex hormone use on sleep architecture in a transgender cohort. Sleep, 46(12), zsad271. https://pubmed.ncbi.nlm.nih.gov/37715990/
Tanji, F., et al. (2025). Psychosocial factors and andropause symptoms among Japanese men: An internet-based cross-sectional study. American Journal of Men’s Health, 19(1). https://pubmed.ncbi.nlm.nih.gov/39945224/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 8 references cited
