Waking between 2am and 4am with a racing heart or a mind that will not settle is a frequent sleep complaint in men over 40. The pattern often involves cortisol, but the mechanism is more specific than “stress causes bad sleep.”
This article covers what triggers cortisol spikes during sleep, how cortisol directly suppresses testosterone production at the testicular level, what the combined hormonal picture looks like, and what 2025 evidence says about the cortisol awakening response. For the broader landscape of hormonal sleep disruption, see the full hormonal sleep disruption guide.
Cortisol dysregulation is one of several causes of 3am waking. GABA deficiency, blood sugar drops, circadian disruption, and testosterone decline each involve different mechanisms, and they can compound one another. Understanding cortisol’s role helps distinguish it from the other contributors.
What Causes Cortisol to Spike in the Middle of the Night?
Every arousal during sleep — even a brief one — activates the HPA axis and produces a measurable cortisol spike.
Spath-Schwalbe et al. (1991) studied 10 healthy men under three conditions — undisturbed baseline sleep, total sleep deprivation, and repeatedly induced arousals during sleep. Each time an arousal was induced, plasma cortisol increased immediately and measurably (Spath-Schwalbe et al., 1991). After each arousal-driven spike, a transient secondary inhibition of cortisol occurred — the HPA axis partially compensated. But the acute excursion had already occurred. Mean cortisol across the disrupted night was not markedly higher than baseline, because the feedback correction brought levels back down between events. The pattern of spikes, however, is what matters for testosterone production, which depends on sustained, uninterrupted low-cortisol windows during sleep.
The study also revealed that spontaneous cortisol pulses during undisturbed sleep did not produce the same secondary suppression pattern that arousal-driven spikes did. This indicates a qualitatively different regulatory state during sleep versus wakefulness — sleep attenuates HPA negative feedback, while waking (including brief arousals) restores it.
What happens the next night is where the damage compounds. Leproult et al. (1997) measured cortisol across the 18:00-23:00 window the evening after sleep loss in healthy young men. Partial sleep deprivation (losing approximately 4 hours) elevated next-evening cortisol by 37% (p=0.03). Total deprivation elevated it by 45% (p=0.003). Under both conditions, the onset of the nocturnal cortisol quiescent period — the low-cortisol trough when testosterone production peaks — was delayed by at least one hour (Leproult et al., 1997).
One bad night does not end when morning arrives. The cortisol elevation persists into the following evening, compressing the testosterone production window for the subsequent night. An arousal spikes cortisol, which narrows the low-cortisol window testosterone depends on, which degrades the next night’s sleep architecture, which increases the probability of another arousal. Each night feeds the next.
How Does a Cortisol Spike Suppress Testosterone During Sleep?
Cortisol acts on the Leydig cell — the testicular cell responsible for manufacturing testosterone — directly, without requiring the brain to send a suppressive message to the testes.
Cumming et al. (1983) elevated cortisol in healthy men through two independent methods: insulin-induced hypoglycemia (which triggers endogenous cortisol release) and exogenous hydrocortisone administration. Under both conditions, serum testosterone fell rapidly following the cortisol rise. Luteinizing hormone (LH) and prolactin remained unchanged, ruling out pituitary-mediated suppression (Cumming et al., 1983). Cortisol elevation produced a rapid suppression of circulating testosterone. This is the direct biochemical link between a 3am cortisol spike and the loss of the sleep-dependent testosterone pulse.
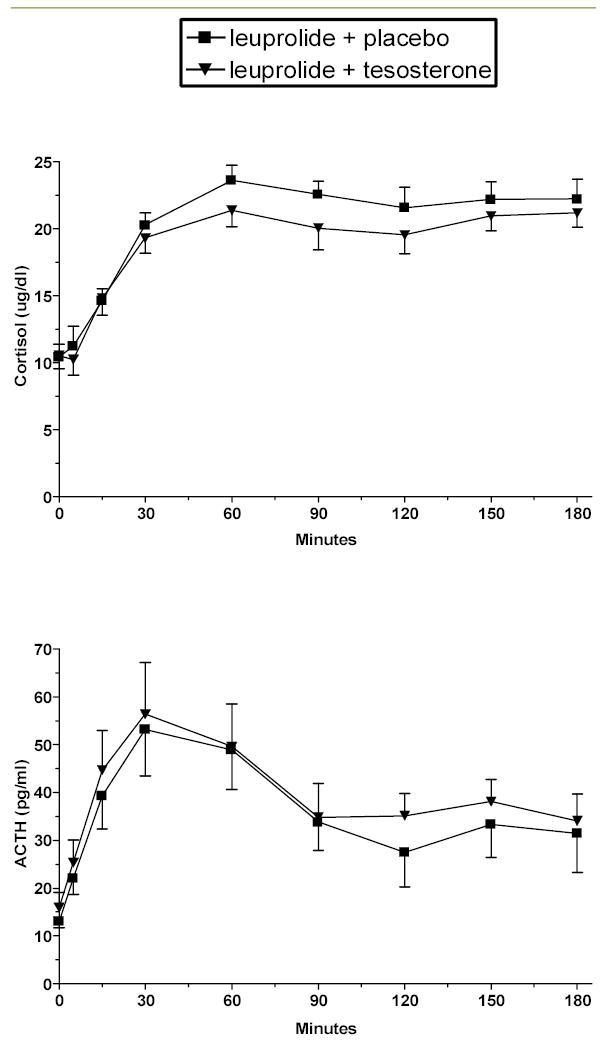
The relationship runs in both directions. Rubinow et al. (2005) studied 10 men under two pharmacological states: induced hypogonadism (using leuprolide to suppress gonadal function) and testosterone replacement added to leuprolide. When researchers stimulated the HPA axis with corticotropin-releasing hormone (CRH), cortisol output was reduced during testosterone replacement compared to the hypogonadal state. Peak cortisol was lower (p=0.005), and cortisol excursion — the total magnitude of the cortisol response — was reduced (p=0.01). ACTH levels increased with testosterone, meaning testosterone did not suppress the pituitary’s response to CRH. Instead, testosterone blunted the adrenal gland’s ability to convert ACTH into cortisol (Rubinow et al., 2005). Testosterone is a functional cortisol buffer at the adrenal level.
The bidirectional axis creates a self-reinforcing loop when it breaks down. Cortisol suppresses testosterone at the Leydig cell. Lower testosterone removes the cortisol buffer at the adrenal gland. The adrenal gland produces more cortisol per unit of ACTH. More cortisol suppresses more testosterone. This is why the cortisol-testosterone imbalance tends to persist and worsen once it begins, rather than correcting on its own.
Is the Cortisol Awakening Response an Arousal Spike or a Circadian Continuation?
The common assumption is direct: you wake up, cortisol spikes. A 2025 study challenges that framing.
Velazquez Sanchez and Dalley (2025) reviewed data from a study that used in-vivo microdialysis — a method that measures tissue-free interstitial cortisol, which is more precise than blood draws — in 201 healthy volunteers in their home environment. The central finding: the rate of cortisol increase did not change when participants woke up compared to the preceding hour when they were still asleep. The post-awakening cortisol rise showed no difference in rate from the pre-awakening cortisol rise (Velazquez Sanchez & Dalley, 2025). Between-subject variability in the post-awakening cortisol profile was partially explained by sleep duration and the timing of waking relative to the circadian cortisol curve.

Nocturnal cortisol spikes from arousals are a separate, documented phenomenon. Chapotot et al. (2001) measured cortisol alongside frontal gamma-band EEG activity (20-45 Hz) — a validated biomarker of central arousal — in 10 healthy young men. Under normal sleep conditions, cortisol secretory rate tracked changes in frontal gamma EEG with approximately a 10-minute lag (R=0.458, p<0.001). Sleep deprivation weakened this HPA-arousal coupling (R=0.276). Sleep deprivation also independently increased cortisol levels, secretory rate, and pulse amplitude, confirming that the HPA axis and the arousal state are co-regulated through a shared neural mechanism (Chapotot et al., 2001). Brief nighttime awakenings engage the cortisol axis within minutes.
The reconciliation: nighttime arousals produce measurable cortisol pulses — the Spath-Schwalbe and Chapotot data are consistent on this. But the broader pre-dawn cortisol rise that begins around 2-4am appears to be circadian regardless of whether you wake. For the person waking at 3am, both processes are relevant. The arousal generates a measurable cortisol pulse on top of a circadian cortisol curve that was already rising. The practical implication is that cortisol is already elevated in the pre-dawn window, and an arousal amplifies it further — a combination that narrows the remaining testosterone production window for the rest of the night.
What Does the Combined Picture of High Cortisol and Low Testosterone Look Like?
The cortisol-testosterone interaction described above — cortisol suppressing testosterone at the Leydig cell, testosterone buffering cortisol at the adrenal — extends beyond the endocrine axis into daily function.
Park et al. (2024) collected salivary testosterone at waking, 30 minutes post-waking, and 60 minutes post-waking in 225 healthy men. Of those, 155 men (69%) showed a typical cortisol awakening response, and in this group, testosterone surged in parallel with cortisol during the post-awakening period. Post-awakening testosterone in these men correlated negatively with age and with 11 individual items on the Aging Males’ Symptoms (AMS) scale — spanning somatic complaints, psychological distress, and sexual function decline. The 70 men (31%) who did not exhibit a typical cortisol awakening response showed no meaningful post-awakening testosterone rise and no correlation with AMS scores (Park et al., 2024). The two axes appear yoked: when the cortisol awakening response is absent or blunted, the testosterone response is absent too, and the AMS correlations disappear.
The cascade extends beyond sleep. Jiao et al. (2025) synthesized evidence that sleep disorders elevate cortisol and dysregulate the HPG axis, reducing testosterone. The downstream consequences include impaired glucose tolerance, reduced insulin sensitivity, and increased inflammation — metabolic changes that are themselves associated with further testosterone decline and further sleep disruption (Jiao et al., 2025). Hernandez-Perez et al. (2024) analyzed NHANES data from 8,748 adults and found that the testosterone-sleep relationship is modulated by age: in middle-aged men (41-64 years), extended sleep (9 or more hours) was associated with low testosterone (OR=2.03; 95% CI: 1.10-3.73). Prolonged sleep in this population may reflect underlying hormonal or metabolic dysregulation rather than a protective effect (Hernandez-Perez et al., 2024).
The recognizable presentation in men with cortisol-testosterone imbalance includes elevated alertness at bedtime despite physical fatigue, waking at 3am with a racing mind, morning heaviness and difficulty initiating the day, declining energy through the afternoon, visceral fat accumulation around the midsection, and reduced libido. The cortisol-testosterone imbalance presents across multiple domains simultaneously.
Cortisol dysregulation is one contributor to 3am waking, but multiple causes often overlap. GABA deficiency, blood sugar instability, inflammation, and circadian disruption can each independently fragment sleep — and each may compound the cortisol-testosterone imbalance described above. Identifying which causes might be involved is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Why Do I Keep Waking Up at 3am?
The 2-4am window has a circadian explanation. Cortisol follows a circadian pattern that reaches its lowest point in the early hours of sleep and begins rising in the pre-dawn hours. As cortisol rises, the arousal threshold drops — meaning less of a stimulus is needed to produce a full awakening. Any additional trigger occurring during this rising cortisol baseline (a blood sugar dip, a bladder pressure cue, a brief episode of airway obstruction) can push past the threshold and produce a full awakening.
Multiple drivers produce awakenings in this same window: GABA deficiency reduces the brain’s ability to maintain sleep continuity, blood sugar drops trigger counter-regulatory cortisol release, and sleep disorders alter the timing and amplitude of the cortisol rhythm (Jiao et al., 2025). The consistency of the timing does not necessarily indicate a single cause — it may reflect multiple causes converging on the same vulnerable window.
Does Sleep Deprivation Raise Cortisol the Next Night?
The Leproult et al. (1997) data on this point is specific. After partial sleep loss (approximately 4 hours lost), plasma cortisol measured between 18:00 and 23:00 the following evening was 37% higher than after a normal night of sleep. After total deprivation, the elevation was 45%. In both conditions, the onset of the nocturnal cortisol quiescent period was delayed by at least one hour (Leproult et al., 1997).
The quiescent period is when cortisol drops low enough for testosterone production to reach its peak output. Delaying this window by an hour means testosterone production starts later and has less time to accumulate before the pre-dawn cortisol rise begins. One disrupted night does not resolve in the morning — the hormonal consequences extend into the following evening and compress the testosterone production window for the next sleep cycle.
How Do I Stop Waking Up at 3am?
The approach depends on which arousal trigger is dominant. Cortisol spikes during sleep are driven by arousals, and arousals have multiple causes (Spath-Schwalbe et al., 1991). Addressing the arousal source reduces cortisol spike frequency more effectively than addressing cortisol in isolation.
Sleep-disordered breathing produces repeated micro-arousals that spike cortisol dozens of times per night — screening for this is a high-priority step for men with consistent 3am waking. Blood sugar instability triggers counter-regulatory cortisol release, and stabilizing overnight blood sugar can reduce one source of arousal. GABA deficiency impairs the brain’s ability to maintain sleep continuity, and supporting GABA tone through lifestyle and dietary approaches can improve sleep maintenance. Pre-sleep cortisol elevation from psychological stress narrows the buffer between cortisol levels and the arousal threshold before sleep even begins.
Each of these targets a different mechanism. For many men, more than one contributor is involved, and addressing only cortisol without identifying the other arousal sources produces incomplete results.
Does Cortisol Rise While You Sleep?
The circadian cortisol rhythm reaches its lowest point (the nadir) in the first half of sleep and begins rising in the pre-dawn hours — typically between 2am and 4am — regardless of whether you are asleep or awake. The 2025 microdialysis data from 201 volunteers showed no change in the rate of cortisol increase at the moment of awakening compared to the preceding hour of sleep, indicating that the pre-dawn rise is programmed by the circadian clock rather than triggered by waking (Velazquez Sanchez & Dalley, 2025).
Nocturnal arousals produce a different phenomenon: acute cortisol pulses that are event-driven, not circadian. These are the spikes measured by Spath-Schwalbe et al. (1991) — immediate, arousal-linked, and followed by a transient feedback correction. Both the circadian rise and the arousal-driven spikes are measurable. They involve different mechanisms and produce different cortisol patterns, but both contribute to the total cortisol exposure during the second half of the night.
Can Stress Hormones Disrupt Testosterone Production at Night?
The Cumming et al. (1983) data is direct. When cortisol was elevated through two independent methods — insulin-induced hypoglycemia and exogenous hydrocortisone — testosterone fell rapidly in both conditions. LH and prolactin did not change, confirming that the suppression occurs at the testis, not through central gonadotropin suppression (Cumming et al., 1983). Cortisol elevation produced a rapid suppression of circulating testosterone.
A cortisol spike at 3am does not merely disrupt sleep — it actively suppresses the testosterone production that was underway during that sleep. The nocturnal testosterone pulse depends on a sustained low-cortisol environment, and a cortisol spike interrupts that environment at the site of production. For a more detailed discussion of the testosterone-sleep relationship, see how declining testosterone fragments sleep architecture.
Related Reading
- Hormonal Sleep Disruption in Men: The Complete Guide — How testosterone, cortisol, growth hormone, and other hormones disrupt sleep in men
- Does Low Testosterone Cause Sleep Problems in Men? — How declining testosterone fragments sleep architecture and the bidirectional feedback loop
- Does Growth Hormone Decline Affect Your Sleep After 40? — How age-related growth hormone loss disrupts slow-wave sleep
- Does Andropause Cause Insomnia? What Men Over 50 Need to Know — Compound hormonal aging and sleep disruption in men over 50
- Can Belly Fat Lower Your Testosterone and Disrupt Your Sleep? — How visceral fat suppresses testosterone through aromatase and disrupts sleep
- Can Ultra-Processed Food Lower Testosterone and Disrupt Sleep? — How endocrine-disrupting chemicals in processed food suppress testosterone and degrade sleep
- Can Inflammation Suppress Testosterone and Disrupt Your Sleep? — How inflammatory cytokines suppress testosterone production at the brain and testicular level
- Does Testosterone Replacement Therapy Affect Sleep? — What the evidence says about TRT and sleep quality
- Can Low GABA Cause Waking Up at 3am? — How insufficient GABA fails to maintain sleep through the night
- What Are the Signs of GABA Deficiency? — Recognizing GABA deficiency indicators that disrupt sleep
- Do GABA Supplements Help You Stay Asleep? — Evaluating GABA supplementation evidence for sleep maintenance
- Does GABA Affect Testosterone and Sleep in Men? — The GABA-testosterone connection in male sleep disruption
- How Do You Increase GABA Levels Naturally for Better Sleep? — Lifestyle and dietary approaches to support GABA function
References
1. Spath-Schwalbe, E., Gofferje, M., Kern, W., Born, J., & Fehm, H. L. (1991). Sleep disruption alters nocturnal ACTH and cortisol secretory patterns. Biological Psychiatry, 29(6), 575-584. https://pubmed.ncbi.nlm.nih.gov/1647222/
2. Leproult, R., Copinschi, G., Buxton, O., & Van Cauter, E. (1997). Sleep loss results in an elevation of cortisol levels the next evening. Sleep, 20(10), 865-870. https://pubmed.ncbi.nlm.nih.gov/9415946/
3. Cumming, D. C., Quigley, M. E., & Yen, S. S. (1983). Acute suppression of circulating testosterone levels by cortisol in men. The Journal of Clinical Endocrinology and Metabolism, 57(3), 671-673. https://pubmed.ncbi.nlm.nih.gov/6348068/
4. Chapotot, F., Buguet, A., Gronfier, C., & Brandenberger, G. (2001). Hypothalamo-pituitary-adrenal axis activity is related to the level of central arousal: effect of sleep deprivation on the association of high-frequency waking electroencephalogram with cortisol release. Neuroendocrinology, 73(5), 312-321. https://pubmed.ncbi.nlm.nih.gov/11399904/
5. Rubinow, D. R., Roca, C. A., Schmidt, P. J., Danaceau, M. A., Putnam, K., Cizza, G., Chrousos, G., & Nieman, L. (2005). Testosterone suppression of CRH-stimulated cortisol in men. Neuropsychopharmacology, 30(10), 1906-1912. https://pubmed.ncbi.nlm.nih.gov/15841103/
6. Park, J. Y., Seok, J. H., Cho, K. S., Kang, D. H., Kim, J. S., Do, S. H., Na, H. S., Jang, S. A., & Ahn, R. S. (2024). Association of salivary testosterone levels during the post-awakening period with age and symptoms suggestive of late-onset hypogonadism in men. Annals of Medicine, 56(1), 2356667. https://pubmed.ncbi.nlm.nih.gov/38776237/
7. Velazquez Sanchez, C., & Dalley, J. W. (2025). The cortisol awakening response: Fact or fiction? Brain and Neuroscience Advances, 9, 23982128251327712. https://pubmed.ncbi.nlm.nih.gov/40297522/
8. Hernandez-Perez, J. G., Taha, S., Torres-Sanchez, L. E., Villasante-Tezanos, A., Milani, S. A., Baillargeon, J., Canfield, S., & Lopez, D. S. (2024). Association of sleep duration and quality with serum testosterone concentrations among men and women: NHANES 2011-2016. Andrology, 12(3), 518-526. https://pubmed.ncbi.nlm.nih.gov/37452666/
9. Jiao, Y., Butoyi, C., Zhang, Q., Intchasso Adotey, S. A. A., Chen, M., Shen, W., Wang, D., Yuan, G., & Jia, J. (2025). Sleep disorders impact hormonal regulation: unravelling the relationship among sleep disorders, hormones and metabolic diseases. Diabetology & Metabolic Syndrome, 17(1), 305. https://pubmed.ncbi.nlm.nih.gov/40750881/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 9 references cited
