A two-hour weekend sleep delay desynchronizes your peripheral clocks — the oscillators in your liver, gut, pancreas, and muscle that run on their own circadian cycles. The central brain clock re-entrains to your Monday alarm in about one day, but peripheral clocks take 3 to 8 days depending on the organ. This means Monday through Wednesday, your organs are running on different time zones. Approximately 69 percent of working adults experience this desynchronization weekly.
Sleeping in on weekends feels like recovery. In practice, it creates a weekly cycle of internal desynchronization that goes unnoticed behind Monday exhaustion, metabolic changes, and mood patterns. The brain resets fast. The liver, gut, and pancreas do not.
This article covers how social jet lag desynchronizes central and peripheral clocks, why different organs re-entrain at different rates, and what the documented health consequences are. Social jet lag is one of several factors that disrupt circadian timing; the full cause overview covers the broader circadian disruption framework.
What Is Social Jet Lag and Why Does Approximately 69 Percent of the Population Have It?
Social jet lag is the difference between your biological sleep timing on free days and the schedule your work or social obligations impose on work days. A large-scale epidemiological study found that approximately 69 percent of the studying and working population have social jet lag exceeding one hour, and this mismatch is associated with higher odds of being overweight (Roenneberg et al., 2012). Evening chronotypes and younger adults carry the highest burden.
Wittmann and colleagues introduced the term in 2006, defining social jet lag as the discrepancy between sleep timing on work days and free days. The formula uses data from the Munich ChronoType Questionnaire (MCTQ), which collects separate sleep timing for work and free days (Wittmann et al., 2006). The corrected midpoint of sleep on free days (MSFsc) isolates circadian timing from weekend recovery behavior.
Roenneberg and colleagues applied this measurement to a large population-based sample of over 65,000 entries and found that 69 percent had social jet lag exceeding one hour. Social jet lag was associated with higher BMI and increased odds of being overweight, independent of total sleep duration (Roenneberg et al., 2012).
Evening chronotypes — people whose circadian clocks run later — experience the largest mismatch with standard work schedules. Younger adults also carry higher social jet lag, partly because adolescent circadian timing runs later while school and early-career job start times remain fixed.
Why Does the Brain Clock Re-Entrain in One Day While Your Liver Takes a Week?
The SCN (the brain’s master clock) receives direct light input and can adjust to a new schedule within approximately one day. Peripheral clocks in the liver, kidney, and muscle rely on indirect cues — glucocorticoids, body temperature, autonomic innervation, and feeding-fasting cycles — and need 5 to 8 additional days to fully realign. During this gap, your central clock says Monday but your liver clock says Sunday.
The SCN sits at the top of a hierarchy. It receives photic input directly from specialized retinal ganglion cells and uses this input to synchronize its own molecular oscillation within a single cycle. This is fast because the coupling between light exposure and SCN clock gene expression is direct and well-characterized (Albrecht, 2012).
Peripheral clocks do not receive light input. They depend on cues relayed from the SCN through at least four pathways: autonomic innervation, glucocorticoid secretion from the adrenal glands, body temperature rhythms, and feeding-fasting cycles (Buijs et al., 2016). Each pathway runs on its own timescale. Each organ responds to a different combination of these cues.
Cuesta and colleagues demonstrated that glucocorticoids — one of the primary SCN-to-peripheral relay cues — entrain molecular clock components in human peripheral blood cells. In their experiment, a 6-day glucocorticoid course displaced the phase of PER2-3 and BMAL1 expression in peripheral blood mononuclear cells by approximately 9.5 to 11.5 hours, while central clock markers remained unaffected. This confirmed that peripheral clocks can be re-timed independently of the SCN, and that re-entrainment of peripheral clock gene expression takes days rather than hours (Cuesta et al., 2015).
A 2025 review confirmed that re-entrainment is both tissue-specific and gene-specific — different clock genes within the same organ respond to different zeitgebers at different rates. The liver responds to feeding-fasting cycles as a primary entrainment cue, while skeletal muscle responds to glucocorticoid cues as well as feeding and exercise signals (Bautista et al., 2025).
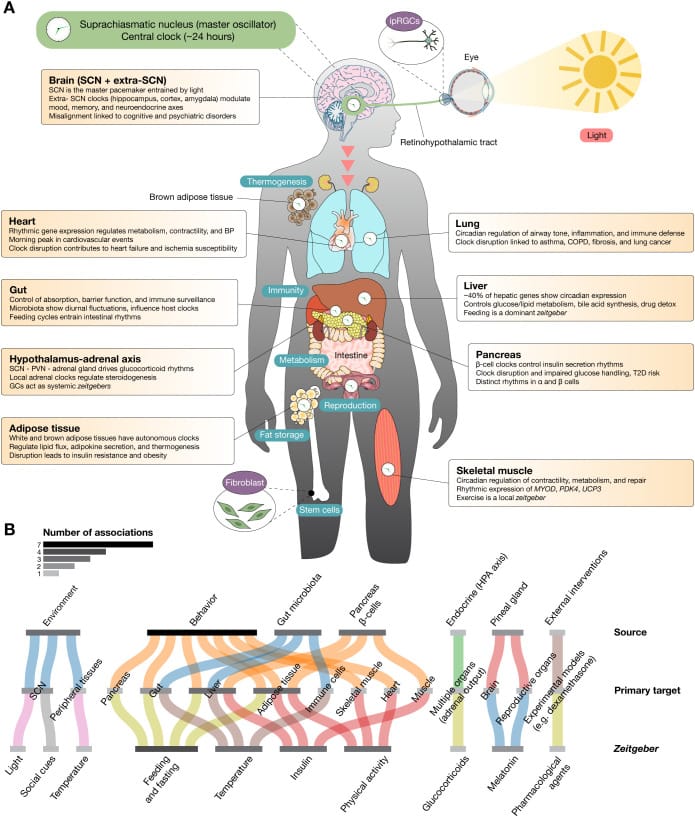
A two-hour weekend sleep delay does not produce a uniform internal delay. The SCN resets by Monday morning. The liver may not catch up until Wednesday or Thursday. The gut and pancreas follow their own timelines — several days each week where different organs are running on different internal schedules.
What Health Consequences Does Chronic Social Jet Lag Cause?
Chronic social jet lag independently predicts metabolic syndrome (odds ratio 1.92 for two or more hours of mismatch), lower HDL cholesterol, higher triglycerides, insulin resistance, and depressive mood. A meta-analysis found that social jet lag is independently associated with depressive symptoms (Gao et al., 2025). Wong et al. (2015) showed the metabolic effects hold after controlling for sleep quality, duration, and physical activity — isolating circadian misalignment as the independent variable.
Metabolic consequences
Wong and colleagues found that social jet lag was independently associated with lower HDL cholesterol, higher triglycerides, higher fasting insulin, and greater insulin resistance — after controlling for sleep quality, sleep duration, insomnia, physical activity, and caloric intake (Wong et al., 2015).
In a study of 1,164 Japanese working adults, Islam and colleagues found that social jet lag of two hours or more was associated with an odds ratio of 1.92 for metabolic syndrome (95% CI: 1.01-3.67) compared to less than one hour. The abdominal obesity component showed a stronger association, with an odds ratio of 2.26 for elevated waist circumference (95% CI: 1.33-3.84). These associations held after adjusting for age, sex, smoking, alcohol, physical activity, and sleep duration (Islam et al., 2018).
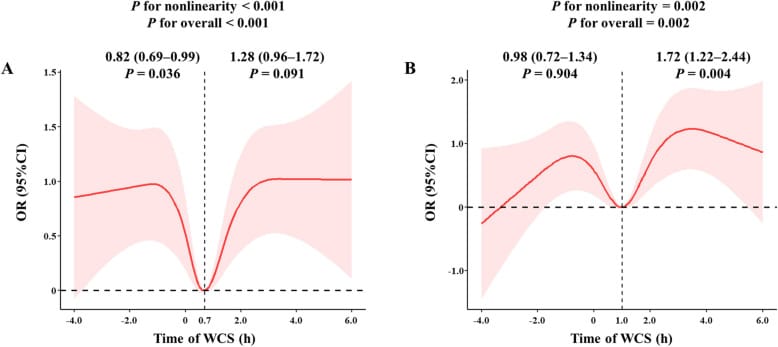
Weekend catch-up sleep itself follows a U-shaped relationship with insulin resistance. Liu and colleagues showed that approximately 0.7 to 1.0 hours of extra weekend sleep corresponds to the minimum insulin resistance risk. Beyond that range, both insufficient and excessive weekend extension predict worsening insulin resistance (Liu et al., 2025).
Mental health consequences
A 2025 meta-analysis of 25 studies found that social jet lag is independently associated with depressive symptoms. The pooled correlation was small (r = 0.049) but statistically consistent across study designs and geographic populations. No consistent association with anxiety was found (Gao et al., 2025).
Cardiovascular risk
A 2025 review in Circulation Research positioned sleep irregularity and circadian misalignment — including social jet lag — as independent risk factors for cardiometabolic disease and mortality, compounding with the metabolic and psychological consequences (Huang et al., 2025).
How Does Meal Timing Compound the Weekend Clock Delay?
Social jet lag drives later timing of breakfast, afternoon snack, and dinner. This creates an additional source of desynchronization because the liver and gut clocks are independently entrained by feeding cycles, not just light. Individuals with social jet lag exceeding one hour ate later and showed worse metabolic control, suggesting the meal timing change adds to the damage from the sleep timing change.
Mota and colleagues documented that social jet lag pushes breakfast, afternoon snack, and dinner later on free days. When sleep timing changes, eating timing changes with it, and the two misalignments may be additive (Mota et al., 2019).
Peripheral clocks in the liver and gut respond directly to feeding-fasting cycles as a primary entrainment cue, independent of what the SCN is doing with light-based cues (Bautista et al., 2025). Light input resets the SCN on Monday morning. But if weekend meal timing also moved later, the liver and gut received a separate delayed timing cue through feeding. These organs are now re-entraining to two conflicting reset cues — the hormonal and autonomic inputs from the SCN pulling toward weekday timing, and the residual feeding-based entrainment pulling toward the weekend schedule.
For individuals with more than one hour of social jet lag, the eating pattern change is consistent enough to produce measurable metabolic differences. Maintaining weekday meal timing over the weekend — even if sleep timing varies — may reduce the peripheral clock disruption.
Many people have more than one cause contributing to their sleep disruption. Social jet lag and peripheral clock desynchronization may compound with metabolic, hormonal, inflammatory, or autonomic factors. Identifying which causes might be involved is a useful next step.
Find out which causes might be driving your 3 AM wakeups →
Frequently Asked Questions
How Do You Fix Social Jet Lag Without Quitting Your Job?
The effective approach is reducing the gap between weekday and weekend sleep midpoints to under one hour. This means limiting weekend sleep-in to no more than 30 minutes beyond your weekday wake time. Aligning meal timing and morning light exposure on weekends to match weekday patterns helps peripheral clocks maintain synchrony.
The MSFsc measurement from the Munich ChronoType Questionnaire provides the benchmark. Reducing the gap between weekday and free-day sleep midpoints below one hour corresponds to the lower-risk category in epidemiological studies (Roenneberg et al., 2012). The U-shaped insulin resistance data from Liu and colleagues narrows the target further: 0.7 to 1.0 hours of extra weekend sleep is the minimum risk point (Liu et al., 2025).
Three approaches reduce social jet lag without requiring identical schedules seven days a week. First, anchor your wake time within a 30-minute window across the week. Second, maintain consistent meal timing on weekends — eat breakfast within an hour of your weekday breakfast time, even if you woke up later. Third, get bright light exposure within 30 minutes of waking on weekend mornings to reinforce the brain clock’s weekday-aligned schedule.
Can Sleeping In on Weekends Hurt Your Health Even if You Feel Rested?
Yes. Weekend catch-up sleep follows a U-shaped relationship with insulin resistance — approximately 0.7 to 1.0 hours of extra weekend sleep is the minimum risk point. Beyond that, both insufficient and excessive weekend extension predict worsening insulin resistance.
The NHANES data from Liu and colleagues makes this point at the population level. Subjective restedness reflects sleep homeostatic pressure — the adenosine-driven need for sleep that accumulates during waking hours. It does not reflect circadian alignment between organs. A person who sleeps in three hours on Saturday and Sunday may feel recovered, but their liver, pancreas, and gut clocks have received a timing cue that pushes them days out of alignment with their central clock (Liu et al., 2025).
Does Social Jet Lag Cause Weight Gain?
Social jet lag is associated with higher odds of being overweight or obese, independent of total sleep duration. The mechanism involves circadian misalignment of metabolic hormone rhythms — insulin sensitivity, leptin, and ghrelin all follow circadian patterns that change timing when sleep timing changes.
Roenneberg and colleagues documented an association between social jet lag and BMI across a large population sample. The effect is not about sleeping less, but about sleeping at the wrong time relative to your circadian clock (Roenneberg et al., 2012).
The hormonal pathways linking circadian misalignment to weight gain involve insulin, leptin, and ghrelin, all of which follow circadian expression patterns. When peripheral clocks in the pancreas and adipose tissue are misaligned from the central clock, insulin peaks when the body is not expecting food, and leptin and ghrelin rhythms arrive out of phase with meals. Over weeks and months, this reduced efficiency in nutrient processing accumulates as weight gain.
Related Reading
- Circadian Rhythm Disruption: How a Misaligned Internal Clock Fragments Sleep, Drives 3AM Wakeups, and Reduces Sleep Depth — the full overview of circadian sleep disruption, including melatonin, cortisol, body temperature, peripheral clocks, and early-morning waking
- Can Rotating Night Schedules Permanently Desynchronize Your Peripheral Clocks? — how rotating night work affects peripheral clocks and metabolic timing
- Why Does Eastbound Jet Lag Last Longer Than Westbound — and What Determines Recovery Rate? — why travel direction changes the speed of circadian recovery
- Night Owl or Delayed Sleep Phase Disorder? How to Know the Difference — how delayed circadian timing differs from preference alone
- Why Does Your Wake Time Advance by Decades — and Can You Reverse Circadian Phase Advance After 60? — why aging shifts wake time earlier and which parts remain reversible
- Which Circadian Mechanisms Change After 40 — and Which Ones Are Reversible? — the broader aging mechanisms that shift circadian timing and sleep depth
- Does Evening Exercise Delay Your Circadian Clock — or Is Disruption Smaller Than You Think? — how exercise timing affects the circadian clock and sleep onset
- Does Time-Restricted Eating Entrain Your Peripheral Clocks? — how meal timing signals peripheral clocks outside the brain
References
Albrecht, U. (2012). Timing to perfection: The biology of central and peripheral circadian clocks. Neuron, 74(2), 246-260. https://pubmed.ncbi.nlm.nih.gov/22542179/
Bautista, J., Ojeda-Mosquera, S., Ordóñez-Lozada, D., & López-Cortés, A. (2025). Peripheral clocks and systemic zeitgeber interactions: From molecular mechanisms to circadian precision medicine. Frontiers in Endocrinology, 16, 1606242. https://pubmed.ncbi.nlm.nih.gov/40510487/
Buijs, F. N., León-Mercado, L., Guzmán-Ruiz, M., Guerrero-Vargas, N. N., Romo-Nava, F., & Buijs, R. M. (2016). The circadian system: A regulatory feedback network of periphery and brain. Physiology, 31(3), 170-181. https://pubmed.ncbi.nlm.nih.gov/27053731/
Cuesta, M., Cermakian, N., & Boivin, D. B. (2015). Glucocorticoids entrain molecular clock components in human peripheral cells. FASEB Journal, 29(4), 1360-1370. https://pubmed.ncbi.nlm.nih.gov/25500935/
Gao, L., Gao, J., & Zeng, Z. (2025). Social jet lag and mental health outcomes: A systematic review and meta-analysis. Acta Psychologica, 259, 105419. https://pubmed.ncbi.nlm.nih.gov/40818420/
Huang, T. (2025). Sleep irregularity, circadian disruption, and cardiometabolic disease risk. Circulation Research, 137(5), 709-726. https://pubmed.ncbi.nlm.nih.gov/40811504/
Islam, Z., Akter, S., Kochi, T., Hu, H., Eguchi, M., Yamaguchi, M., Kuwahara, K., Kabe, I., & Mizoue, T. (2018). Association of social jetlag with metabolic syndrome among Japanese working population: The Furukawa Nutrition and Health Study. Sleep Medicine, 51, 53-58. https://pubmed.ncbi.nlm.nih.gov/30099352/
Liu, X., Chu, A., & Ding, X. (2025). Investigating the associations between weekend catch-up sleep and insulin resistance: NHANES cross-sectional study. BMC Medicine, 23(1), Article 311. https://pubmed.ncbi.nlm.nih.gov/40437485/
Mota, M. C., Silva, C. M., Balieiro, L. C. T., Gonçalves, B. F., Fahmy, W. M., & Crispim, C. A. (2019). Association between social jetlag food consumption and meal times in patients with obesity-related chronic diseases. PLoS ONE, 14(2), e0212126. https://pubmed.ncbi.nlm.nih.gov/30753224/
Roenneberg, T., Allebrandt, K. V., Merrow, M., & Vetter, C. (2012). Social jetlag and obesity. Current Biology, 22(10), 939-943. https://pubmed.ncbi.nlm.nih.gov/22578422/
Wittmann, M., Dinich, J., Merrow, M., & Roenneberg, T. (2006). Social jetlag: Misalignment of biological and social time. Chronobiology International, 23(1-2), 497-509. https://pubmed.ncbi.nlm.nih.gov/16687322/
Wong, P. M., Hasler, B. P., Kamarck, T. W., Muldoon, M. F., & Manuck, S. B. (2015). Social jetlag, chronotype, and cardiometabolic risk. Journal of Clinical Endocrinology and Metabolism, 100(12), 4612-4620. https://pubmed.ncbi.nlm.nih.gov/26580236/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 12 references cited
