NMN is a widely used NAD+ precursor supplement, and "NMN insomnia" is one of the higher-volume searches in the NAD+ sleep space. Supplement brands say take it in the morning. Longevity forums say afternoon. Human trials have tested both — and the answers are more specific than either side admits.
This article covers what the human trial evidence shows about NMN timing, dose thresholds, and sleep outcomes — including the studies where NMN did nothing to sleep and the populations where it made a measurable difference. NMN influences sleep through NAD+-dependent circadian mechanisms — the SIRT1-clock gene pathway covered in the parent Metabolic Sleep Disruption guide. This article narrows that broad mechanism to one variable: when and how much NMN you take.
Does NMN Cause Sleep Problems?
The safety data across published NMN trials point in the same direction. Yi et al. (2023) tested 300, 600, and 900 mg/day in 80 healthy middle-aged adults for 60 days — zero sleep-related adverse events at any dose. Morifuji et al. (2024) gave 250 mg/day for 12 weeks in older adults — no adverse effects related to the supplement. Yamaguchi et al. (2024) administered 250 mg/day to 11 healthy middle-aged men in an open-label trial for 8 weeks — well-tolerated.
The insomnia reports come from a different source: user communities. Reddit threads and supplement forums describe "enormous amounts of energy," "wide awake and struggling to sleep," and similar experiences. The pattern in these reports is the same — evening dosing, often above 400 mg. The molecule itself does not appear in any controlled study as a sleep disruptor. The variable is when NAD+ levels peak.
The mechanism is mapped out in the literature. NMN is converted to NAD+, which activates SIRT1 — a protein deacetylase that regulates the core clock genes BMAL1 and PER2 (Weiss, 2025). SIRT1 activation strengthens circadian oscillation. When that activation peaks during the biological evening, it reinforces alertness at the wrong time.
Yamaguchi et al. (2024) adds an important null result. In 11 healthy middle-aged men with already-good sleep quality, NMN produced no change in sleep — positive or negative. That is a floor effect: there was no room to improve, and the supplement did not worsen what was already good.
When Should You Take NMN for Better Sleep — Morning or Afternoon?
Kim et al. (2022) is the only published trial that randomized NMN timing as a study variable. The trial enrolled 108 older Japanese adults across four arms: NMN morning, NMN evening (6pm-bedtime), placebo morning, and placebo evening. All participants received 250 mg/day for 12 weeks.
The evening NMN group had the larger effect size for drowsiness reduction: Cohen’s d = 0.64. The morning NMN group did not show this drowsiness benefit. All four groups (including placebo) improved in the five-times sit-to-stand test. The drowsiness finding was larger in the evening groups, with the NMN evening group showing the largest effect (d = 0.64) compared to the placebo evening group (d = 0.50).

Why did evening dosing work better for older adults? NAD+ levels decline with age, and the age-related NAD+ nadir (lowest point in the daily cycle) is deeper in the later part of the day. Replenishing NAD+ during that trough aligns the supplement’s peak effect with the body’s circadian need for it — rather than adding NAD+ when levels are already adequate.
User community data shows the opposite pattern in younger populations. Adults under 50 report that NMN taken after midday produces restlessness at bedtime. The likely explanation is that younger adults have higher baseline NAD+ levels, so the additional late-day NAD+ elevation raises circadian alertness above the threshold needed for comfortable sleep onset.
A practical starting point: morning dosing for adults under 50, especially if evening alertness is a concern. For adults over 60 with daytime drowsiness, evening dosing is the option with RCT support (Kim et al., 2022). In NMN user communities, magnesium glycinate is the pairing people report using frequently to offset any evening alertness — though no controlled trial has tested that combination.
What Dose of NMN Starts Disrupting Sleep?
Yi et al. (2023) established the dose-response curve. In 80 healthy middle-aged adults randomized to 300, 600, or 900 mg/day NMN or placebo for 60 days, blood NAD+ concentrations increased in all NMN groups (p <= 0.001 versus placebo at days 30 and 60). The 600 mg group reached the practical efficacy threshold — 900 mg did not produce meaningfully better NAD+ elevation or functional outcomes (six-minute walking distance, SF-36 quality of life).
Morifuji et al. (2024) demonstrated that 250 mg/day was sufficient to improve PSQI sleep scores in older adults — suggesting the effective dose for sleep is at the lower end of the range.
The sleep disruption reports from user communities cluster at 400 mg or higher when taken in the evening. Below 400 mg, or at any dose taken in the morning, the reports of sleep disruption are sparse.
Gao et al. (2023) chose 320 mg/day for the first dedicated insomnia RCT — a 400-patient multicenter trial testing NMN in people with chronic insomnia over 60 days. The dose falls within the lower range of NMN supplementation, consistent with the positive sleep findings at 250 mg in Morifuji (2024).
For sleep-focused supplementation, 250 mg is the dose with the better-supported positive sleep evidence. For general NAD+ goals (energy, metabolic function, biological aging markers), 600 mg appears to be the ceiling of useful dosing. Evening doses above 400 mg carry the higher likelihood of timing-related sleep disruption.
Can NMN Improve Sleep Quality Over Time?
Morifuji et al. (2024) is the direct evidence for sleep-quality scoring. Sixty older adults received 250 mg/day NMN or placebo for 12 weeks. The NMN group had lower (better) scores on both the PSQI Global score and the Daytime Impairment subscale at 12 weeks versus placebo. Blood NAD+ and its metabolites were elevated in the NMN group — verifying the supplement was absorbed and metabolically active.
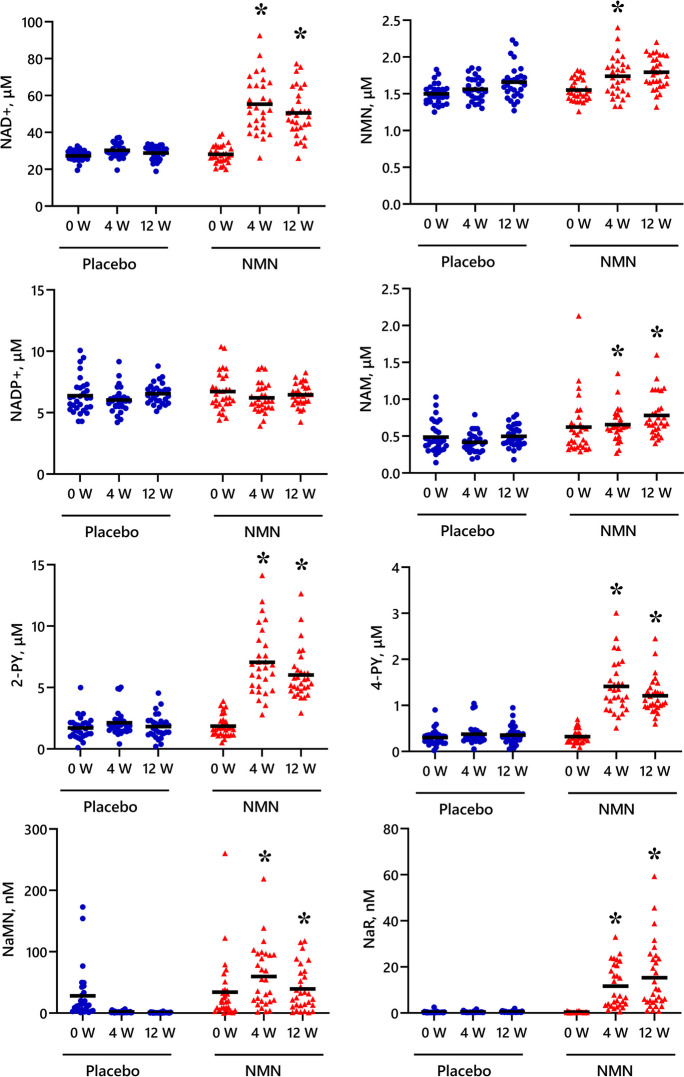
Kim et al. (2022) adds the timing dimension: the evening NMN group showed a drowsiness reduction with an effect size of d = 0.64 at 12 weeks. Both positive trials required a full 12-week supplementation period — there is no published evidence that NMN produces rapid sleep benefits within days or weeks.
The population matters. Yamaguchi et al. (2024) found no sleep improvement in 11 healthy middle-aged men — participants already in the "good sleep" range. The positive results from Morifuji (2024) and Kim (2022) came from older adults. NMN may improve sleep quality more readily in older adults, who have lower NAD+ levels, than in healthy middle-aged adults.
Gao et al. (2023) registered a 400-patient multicenter RCT testing 320 mg/day NMN in people with chronic insomnia, using PSQI as the primary outcome and the Insomnia Severity Index (ISI), Epworth Sleepiness Scale (ESS), sleep efficiency, and REM latency as secondary measures. Results are pending. This will be the first data on NMN in a population with chronic insomnia.
Supporting context from a related NAD+ precursor: Wu et al. (2025) tested 2000 mg/day nicotinamide riboside (NR) — a different NAD+ precursor that feeds the same downstream pathway — in 58 people with long-COVID. NAD+ levels increased 2.6- to 3.1-fold. Within-group PSQI scores improved by 0.74 points (p = 0.03) after 10 weeks. The primary between-group comparison was not statistically different (p = 0.69), but the within-group finding adds to the evidence that NAD+ precursors can move sleep quality scores in populations where sleep is impaired.
Does NMN Change Melatonin or Circadian Clock Activity?
The mechanistic link between NMN and circadian rhythm runs through SIRT1. Weiss (2025) reviewed the pathway: NAD+ serves as the essential cofactor for SIRT1, which is recruited to the CLOCK:BMAL1 chromatin complex. There, SIRT1 deacetylates BMAL1 and PER2, regulating the amplitude and duration of circadian gene expression. When NAD+ is abundant, SIRT1 activity is high, and circadian oscillation is stronger.
This explains why timing sensitivity exists. SIRT1 activation during the biological evening reinforces the wake-promoting arm of the circadian clock — increased alertness, elevated body temperature, sustained cortisol — at a point when the body should be transitioning toward sleep. During the biological morning, the same SIRT1 activation reinforces the daytime alertness phase, which is appropriate.
Animal data from the same review (Weiss, 2025) shows that nicotinamide riboside (NR) — an NAD+ precursor with the same downstream pathway — enhanced BMAL1 and PER2 expression in animal models, reduced NREM sleep by approximately 17%, and changed sleep architecture. Separately, earlier animal studies found that nicotinamide (a different vitamin B3 form) increased REM sleep. Together, the evidence suggests NAD+ precursors may change sleep architecture rather than increase total sleep time. Whether this translates to humans at supplemental NMN doses has not been tested.
NMN is not a melatonin replacement or equivalent. The tryptophan-kynurenine pathway — where tryptophan can be converted to either serotonin/melatonin or to NAD+ via the de novo pathway — creates a theoretical competition between melatonin production and NAD+ production for the same amino acid precursor. But NMN bypasses this competition because it enters the NAD+ salvage pathway, not the de novo pathway from tryptophan. Supplementing NMN does not divert tryptophan away from melatonin production.
If your sleep disruption goes beyond supplement timing — if you are waking at 3am, lying awake with a racing mind, or sleeping a full 7-8 hours but waking exhausted — the cause might not be your NMN dose or schedule. Metabolic imbalances, blood sugar regulation, cortisol timing, and circadian misalignment can all produce the same pattern. A supplement adjustment might help, but it might also be addressing only one contributor among several.
Find out which causes might be driving your 3am wakeups
Frequently Asked Questions
How Long Does It Take for NMN to Improve Sleep?
Morifuji et al. (2024) assessed participants at baseline, 4 weeks, and 12 weeks. The sleep improvements appeared at the 12-week mark. Kim et al. (2022) measured outcomes at 12 weeks only — no interim time points were tested for sleep endpoints. The Gao et al. (2023) insomnia trial uses a 60-day (approximately 8.5-week) design, which will provide data on a shorter timeline — but results are not yet available.
User community reports of early energy changes within the first week of NMN supplementation are common. These describe increased daytime alertness, not improved sleep quality. The two outcomes appear to work on different timescales — acute NAD+ elevation may increase daytime energy within days, while measurable sleep quality improvement requires weeks of sustained NAD+ restoration.
Should You Take NMN With Magnesium to Offset Sleep Disruption?
The community pattern is repeated across forums: across Reddit threads and supplement forums, magnesium glycinate is the frequently suggested addition when users report NMN-related sleep disruption. The logic — that magnesium’s role in GABAergic (inhibitory) neurotransmission could offset the alertness-promoting effects of elevated NAD+ — has biological plausibility but has not been tested in a controlled setting.
Adjusting NMN timing and dose has stronger evidence than adding a second supplement. Moving the dose from evening to morning, or reducing from 500+ mg to 250 mg, addresses the timing-dose interaction that produces sleep disruption in the first place. Magnesium glycinate has its own independent evidence base for sleep — but not as a specific counterbalance to NMN.
Does NMN Work Differently for Sleep in Older Versus Younger Adults?
The controlled evidence splits along age and baseline sleep quality. Morifuji et al. (2024) and Kim et al. (2022) — both positive for sleep outcomes — enrolled older adults. Yamaguchi et al. (2024) — null for sleep — enrolled 11 healthy middle-aged men.
The age-related NAD+ decline may explain the difference: older adults with lower NAD+ levels have more room for improvement from NAD+ restoration, while middle-aged adults with adequate NAD+ levels may not benefit measurably.
User community data reinforces the age split. Younger adults (under 40) report insomnia and evening restlessness from NMN more frequently than older adults. Older adults are more likely to report benefit — reduced daytime drowsiness, improved sleep quality. The likely explanation: younger adults have higher baseline NAD+ levels, so supplementation produces a surplus that drives circadian alertness. Older adults have depleted NAD+, so supplementation restores a deficit.
Can NMN Reduce Nighttime Awakenings?
The PSQI — used in Morifuji (2024), Kim (2022), and Yamaguchi (2024) — is a self-report questionnaire with seven components: subjective sleep quality, sleep latency, sleep duration, habitual sleep efficiency, sleep disturbances, use of sleeping medication, and daytime impairment. The global score captures an aggregate picture. It does not isolate wake-after-sleep-onset (WASO) or number of nighttime awakenings as standalone metrics.
Actigraphy and polysomnography data for NMN supplementation do not yet exist in the published literature. The Gao et al. (2023) insomnia RCT includes sleep efficiency as a secondary outcome — the ratio of time asleep to time in bed — which is the closest available measure to nighttime wakefulness. When those results are published, they will provide the first objective data on whether NMN reduces time spent awake during sleep.
Related Reading
- Metabolic Sleep Disruption: How Metabolic Impairment Fragments Sleep — the pillar article for metabolic sleep disruption, covering glucose regulation, cortisol rhythm, mitochondrial stress, NAD+ loss, and fat metabolism
- Which Nicotinamide Adenine Dinucleotide Precursor Improves Sleep — Nicotinamide Mononucleotide or Nicotinamide Riboside? — NMN and NR sleep evidence, timing, safety, and human trial differences
- Does Apigenin Protect Your NAD+ and Improve Sleep Through CD38 Inhibition? — CD38 inhibition, NAD+ depletion with age, and apigenin sleep evidence
- Does Your Body Divert Tryptophan From Melatonin to Make NAD+? — tryptophan allocation between melatonin, kynurenine, inflammation, and NAD+ synthesis
- Why Does Sleep Repair Your DNA — And Burn Through Your NAD+ to Do It? — PARP1, sleep pressure, DNA repair, and NAD+ demand during sleep
- Does Your Gut Decide Whether Your NAD+ Supplement Works? — gut microbiome conversion of NAD+ precursors and why supplement response varies
- Does NAD+ IV Therapy Improve Sleep? What the Evidence Shows — IV NAD+ evidence, safety, infusion effects, and comparison with oral precursors
- Why Does NAD+ Drop Faster in Women After 40 — And What Does That Mean for Sleep? — menopause, ovarian aging, NAD+ decline, and sleep disruption after 40
References
Gao, X., Li, J., Xu, S., Li, X., Wang, X., Li, Y., Huang, Y., Liu, S., & Zeng, Q. (2023). Oral nicotinamide mononucleotide (NMN) to treat chronic insomnia: protocol for the multicenter, randomized, double-blinded, placebo-controlled trial. Trials, 24(1), 340. https://pubmed.ncbi.nlm.nih.gov/37202819/
Kim, M., Seol, J., Sato, T., Fukamizu, Y., Sakurai, T., & Okura, T. (2022). Effect of 12-Week Intake of Nicotinamide Mononucleotide on Sleep Quality, Fatigue, and Physical Performance in Older Japanese Adults: A Randomized, Double-Blind Placebo-Controlled Study. Nutrients, 14(4), 755. https://pubmed.ncbi.nlm.nih.gov/35215405/
Morifuji, M., Higashi, S., Ebihara, S., & Nagata, M. (2024). Ingestion of beta-nicotinamide mononucleotide increased blood NAD levels, maintained walking speed, and improved sleep quality in older adults in a double-blind randomized, placebo-controlled study. GeroScience, 46(5), 4671-4688. https://pubmed.ncbi.nlm.nih.gov/38789831/
Weiss, C. (2025). Vitamin B3 Ameliorates Sleep Duration and Quality in Clinical and Pre-Clinical Studies. Nutrients, 17(12), 1982. https://pubmed.ncbi.nlm.nih.gov/40573093/
Wu, C. Y., Reynolds, W. C., Abril, I., McManus, A. J., Brenner, C., Gonzalez-Irizarry, G., Gutierrez-Martinez, L., Sun, O., Rosand, J., Tanzi, R. E., Arnold, S. E., & Guzman-Velez, E. (2025). Effects of nicotinamide riboside on NAD+ levels, cognition, and symptom recovery in long-COVID: a randomized controlled trial. EClinicalMedicine, 89, 103633. https://pubmed.ncbi.nlm.nih.gov/41357333/
Yamaguchi, S., Irie, J., Mitsuishi, M., Uchino, Y., Nakaya, H., Takemura, R., Inagaki, E., Kosugi, S., Okano, H., Yasui, M., Tsubota, K., Hayashi, K., Yoshino, J., & Itoh, H. (2024). Safety and efficacy of long-term nicotinamide mononucleotide supplementation on metabolism, sleep, and nicotinamide adenine dinucleotide biosynthesis in healthy, middle-aged Japanese men. Endocrine Journal, 71(2), 153-169. https://pubmed.ncbi.nlm.nih.gov/38191197/
Yi, L., Maier, A. B., Tao, R., Lin, Z., Vaidya, A., Pendse, S., Thasma, S., Andhalkar, N., Avhad, G., & Kumbhar, V. (2023). The efficacy and safety of beta-nicotinamide mononucleotide (NMN) supplementation in healthy middle-aged adults: a randomized, multicenter, double-blind, placebo-controlled, parallel-group, dose-dependent clinical trial. GeroScience, 45(1), 29-43. https://pubmed.ncbi.nlm.nih.gov/36482258/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 7 references cited
