Time-restricted eating is often framed as a way to “sync” metabolism with the body clock. That framing is partially right — meal timing does entrain peripheral organ clocks — but it collapses a hierarchy that matters: food moves the liver and adipose tissue, not the brain. The distinction has consequences for how you interpret the benefits, the risks of late eating, and what time-restricted eating can and cannot do.
This article covers the animal science establishing food as a peripheral zeitgeber, the first controlled human evidence of peripheral uncoupling from meal delays, and a 2025 randomized controlled trial testing whether clock-entrainment from time-restricted eating produces cardiometabolic benefits. It does not cover time-restricted eating as a caloric restriction strategy — that relationship is addressed in Does Your Circadian Clock Control When Insulin Works?. For the broader picture of how circadian disruption affects insulin, glucose, and metabolic health, see Circadian Rhythm Disruption.
Can Food Timing Uncouple Your Peripheral Clocks From the Master Clock?
Damiola et al. (2000) established the primary evidence for food as a peripheral zeitgeber. Mice kept under a standard light-dark regimen were fed exclusively during the light phase — their rest phase — for eight consecutive days. By the ninth day, the phase of circadian gene expression (including Dbp, Per1, Per2, Per3, Cry1, Rev-erbα, and Cyp2a5) in the liver had inverted by up to 12 hours relative to mice fed during the dark (active) phase. The same inversion occurred in the kidney, heart, and pancreas, though at different rates. The liver was the fastest-responding organ, achieving near-complete phase inversion within 4 to 7 days. The kidney, heart, and pancreas required longer exposure to the altered feeding schedule (Damiola et al., 2000).
The SCN was unaffected. In situ hybridization of coronal brain sections showed that Per1 and Per2 mRNA accumulation in the SCN maintained the same phase under both daytime and nighttime feeding conditions. The master clock stayed locked to the light-dark cycle regardless of when food arrived.
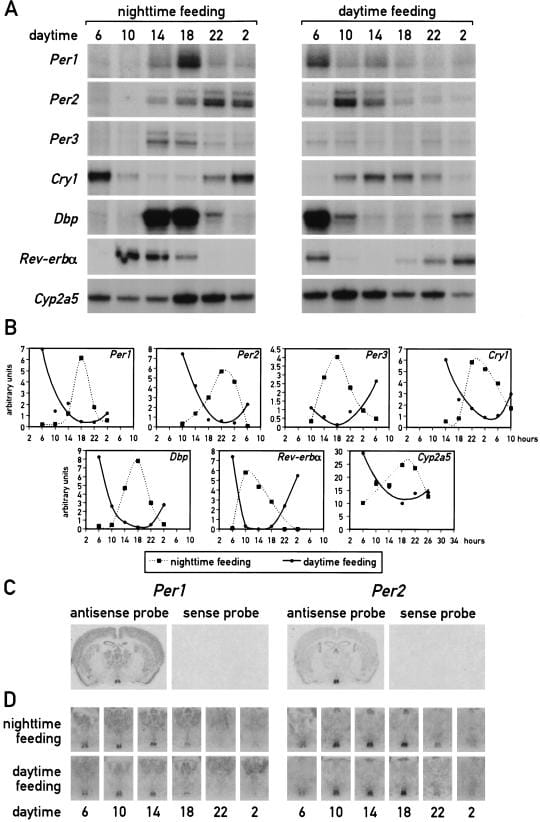
Identical results occurred under constant darkness — confirming that peripheral entrainment by food is independent of photic input and not mediated through any light-driven pathway. The implication: metabolic cues triggered by food availability — including insulin, glucocorticoids, and body temperature fluctuations — serve as timing cues capable of overriding SCN output in peripheral organs. This was the first demonstration that peripheral clocks can be decoupled from the master clock by a single behavioral input, and every subsequent time-restricted eating study builds on what Damiola et al. established.
How Fast Do Peripheral Clocks Respond to Meal Timing Changes?
Stokkan et al. (2001) used transgenic rats expressing a luciferase reporter driven by the Period1 promoter, allowing continuous visualization of circadian oscillations in living tissue. When food access was restricted to the light (rest) phase, the liver clock moved approximately 10 hours within 48 hours. SCN bioluminescence rhythms remained phase-locked to the light-dark cycle throughout — no centrally driven compensation occurred (Stokkan et al., 2001).
Ten hours in two days was faster than light-driven resetting of the SCN produces in peripheral adaptation. The liver, receiving metabolic cues from food intake, responds to meal timing on its own timeline without waiting for the master clock.
The broader implication was that feeding cycles can entrain the liver independently of the SCN and the light cycle — a result the authors argued “suggest[s] the need to reexamine the mammalian circadian hierarchy.” The liver does not need the master pacemaker to respond to food timing, and feeding behavior is a sufficient, standalone entrainment cue for liver tissue.
This result explains why people engaged in night work or jet-lagged travel experience rapid hepatic metabolic desynchronization even when the SCN adapts slowly: the liver is already responding to food timing on an independent clock. A consistently late eating window does not wait weeks to move peripheral clocks. The entrainment begins within 48 hours.
What Happens When You Eat Late — Does It Move Your Glucose Clock?
Wehrens et al. (2017) conducted a 13-day inpatient within-subjects study with 10 healthy young men. All meals were delayed by 5 hours — from 30 minutes after waking to 5.5 hours after waking. The rest of the environment was held constant: light exposure, sleep timing, and activity levels were unchanged. Only the timing of food intake was manipulated (Wehrens et al., 2017).
The glucose clock moved with the meals. Plasma glucose rhythms delayed by 5.7 hours — a near-complete entrainment to the new meal schedule within the 13-day window. This was not a subtle change; the glucose rhythm tracked the new feeding time with high precision.
White adipose tissue PER2 mRNA — measured from surgical biopsies — moved approximately 1 hour later with the delayed meal schedule. This is direct molecular evidence of peripheral clock resetting in human tissue, not a surrogate marker. Meanwhile, salivary melatonin onset and serum cortisol morning peak — both robust proxies of SCN phase — were unaffected by meal timing in either condition. The SCN held its position while the peripheral metabolic clock in adipose tissue moved.
Average 24-hour glucose concentration decreased by 0.27 mmol/L under the delayed-meal condition despite equivalent food amounts and composition — a timing-driven metabolic change, independent of caloric intake.
The implications extend beyond habitual late eaters. People doing night work and those with chronically delayed eating schedules may accumulate peripheral misalignment without any subjective sense that their body clock is moving, because their SCN-driven melatonin and cortisol rhythms remain on schedule. The peripheral metabolic clocks move, but the conscious experience of circadian timing — driven by the SCN — stays put.
Does Clock-Entrainment From Time-Restricted Eating Improve Metabolic Health?
The claim that restricting your eating window aligns your peripheral clocks, and that alignment produces metabolic improvements, has two parts. Peters et al. (2025) tested both halves directly (Peters et al., 2025).
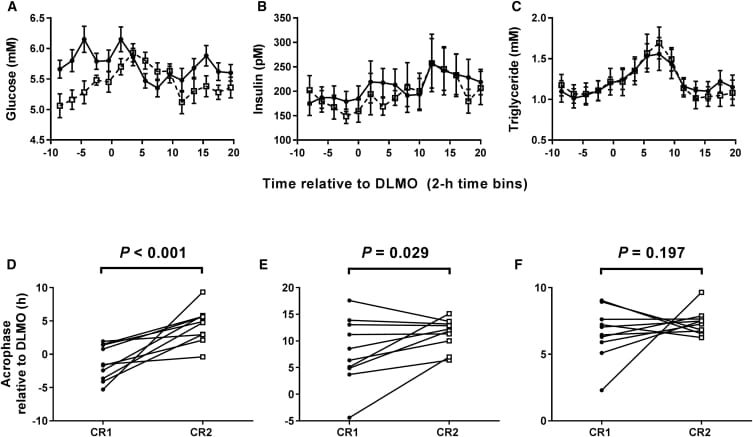
The trial enrolled 31 overweight or obese women in two consecutive 2-week conditions: early time-restricted eating (8:00-16:00) and late time-restricted eating (13:00-21:00), under intended isocaloric conditions. Peripheral circadian phase was measured directly via clock gene expression in blood monocytes — not estimated from behavioral proxies. Wrist actigraphy tracked sleep midpoint.
Clock-entrainment was demonstrated. Late time-restricted eating delayed monocyte circadian phase by approximately 24 minutes relative to early time-restricted eating (p=0.10) and moved sleep midpoint 15 minutes later (p<0.001). The eating window timing does entrain peripheral clocks in humans — that half of the hypothesis is validated.
The metabolic half did not hold up. Insulin sensitivity showed no meaningful change between arms (difference: -0.07; 95% CI: -0.77 to 0.62; p=0.60). Twenty-four-hour glucose profiles, lipid panels, inflammatory markers, and oxidative stress indices were unchanged within or between conditions. Adherence was high — 96.5% in the early arm and 97.7% in the late arm — ruling out compliance as an explanation.
Minor weight loss did occur: 1.08 kg in the early time-restricted eating arm and 0.44 kg in the late arm. But dietary analysis revealed an inadvertent caloric deficit of approximately 167 kcal/day in the early arm. When calories are controlled, the clock-entrainment produced by time-restricted eating does not produce cardiometabolic improvements in this population over a 2-week period.
The implication: metabolic improvements in prior time-restricted eating trials may have been driven by caloric restriction rather than circadian alignment. The dissociation between clock-entrainment and metabolic outcomes complicates the assumption that misalignment is the primary driver of metabolic impairment. Time-restricted eating does move peripheral clocks — but moving peripheral clocks, by itself, may not be sufficient to improve metabolic markers when energy intake is held constant.
Meal timing is one of several inputs that can move your peripheral clocks. If your eating schedule conflicts with your light exposure, exercise timing, or work schedule, the result might be overlapping circadian misalignment. Identifying which causes might be contributing is a useful next step.
Find out which causes might be driving your 3 AM wakeups →
Does Intermittent Fasting Reset Circadian Rhythm?
Isherwood et al. (2023) addressed this in an 8-day controlled laboratory study with 24 healthy adult men. One group consumed two large meals at 7.5 and 14.5 hours after waking for 6 days, then entered a 37-hour constant routine with no food and continuous interstitial glucose monitoring every 15 minutes (Isherwood et al., 2023).
During the constant routine — when no food was provided — glucose concentrations in the two-meal group showed an anticipatory nadir at habitual meal times. Hunger ratings rose before the times that had been habitual meal windows (p<0.05). The body had encoded the meal timing pattern and was reproducing it even without food.
Melatonin-derived circadian phase did not differ between meal groups, confirming that these anticipatory metabolic and behavioral rhythms were encoded without moving the SCN. This is among the first controlled human demonstrations that the food-entrainable oscillator — studied extensively in rodents but rarely characterized in humans — encodes habitual timing and reproduces it in the absence of meals. The rhythm is not a response to food arriving; it is a learned anticipatory pattern that the body maintains independently.
What Is the Best Eating Window for Circadian Alignment?
Rovira-Llopis et al. (2024) synthesized 18 controlled studies involving 1,169 participants. Across the pooled data, HbA1c was reduced (effect size -0.27; p=0.01) and fasting insulin decreased (effect size -0.40; p=0.01). HOMA-IR showed a declining trend (effect size -0.32; p=0.06) (Rovira-Llopis et al., 2024).
Sub-group analysis revealed that early time-restricted eating — eating windows aligned with the morning — produced the largest effects, including fasting glucose reductions in overweight and obese populations compared to control or late-window conditions. The earliest eating windows produced the greatest glycemic improvements. Late eating windows showed smaller or absent metabolic benefits across the pooled studies.
Heterogeneity across trials in window duration (6-10 hours), population characteristics, and adherence monitoring limits effect size precision. But the pattern was consistent: the circadian timing of the eating window — not the restriction alone — modulates glycemic outcomes.
Can Time-Restricted Eating Help People Who Do Night Work?
McHill and Butler (2024) synthesized the evidence across species in the Annual Review of Nutrition. In rodent studies, restricting high-fat diet access to the biologically active (dark) phase prevented obesity and metabolic impairment compared to identical caloric consumption during the inactive phase — demonstrating food timing as a causal metabolic variable independent of calories (McHill & Butler, 2024).
Feeding schedules can re-entrain peripheral clocks independently of SCN phase — Stokkan et al. (2001) demonstrated a 10-hour liver phase shift within 48 hours in rats — with insulin and glucose fluctuations acting as food-related entraining signals. For people doing night work, this means that aligning food intake to the biological day — even when the work schedule requires nighttime wakefulness — may reduce peripheral clock desynchronization by removing one of the conflicting zeitgeber inputs.
A species difference limits direct translation: mice experience severe glucose decline during a 12-hour fast while humans maintain stable glucose. Rodent time-restricted eating schedules impose metabolic stress that has no equivalent in human time-restricted eating schedules. Human time-restricted eating trials therefore cannot be interpreted as direct replications of the rodent studies — the metabolic context differs between species, and the magnitude of peripheral clock re-entrainment from food restriction may not be equivalent.
Related Reading
- Circadian Rhythm Disruption: How a Misaligned Internal Clock Fragments Sleep, Drives 3AM Wakeups, and Reduces Sleep Depth — the full overview of circadian sleep disruption, including melatonin, cortisol, body temperature, peripheral clocks, and early-morning waking
- How Does a Two-Hour Weekend Sleep Delay Desynchronize Your Peripheral Clocks for Days? — how repeated weekend schedule shifts create chronic circadian misalignment
- Can Rotating Night Schedules Permanently Desynchronize Your Peripheral Clocks? — how rotating night work affects peripheral clocks and metabolic timing
- Why Does Eastbound Jet Lag Last Longer Than Westbound — and What Determines Recovery Rate? — why travel direction changes the speed of circadian recovery
- Night Owl or Delayed Sleep Phase Disorder? How to Know the Difference — how delayed circadian timing differs from preference alone
- Why Does Your Wake Time Advance by Decades — and Can You Reverse Circadian Phase Advance After 60? — why aging shifts wake time earlier and which parts remain reversible
- Which Circadian Mechanisms Change After 40 — and Which Ones Are Reversible? — the broader aging mechanisms that shift circadian timing and sleep depth
- Does Evening Exercise Delay Your Circadian Clock — or Is Disruption Smaller Than You Think? — how exercise timing affects the circadian clock and sleep onset
References
1. Damiola, F., Le Minh, N., Preitner, N., Kornmann, B., Fleury-Olela, F., & Schibler, U. (2000). Restricted feeding uncouples circadian oscillators in peripheral tissues from the central pacemaker in the suprachiasmatic nucleus. Genes & Development, 14(23), 2950-2961. https://pubmed.ncbi.nlm.nih.gov/11114885/
2. Stokkan, K. A., Yamazaki, S., Tei, H., Sakaki, Y., & Menaker, M. (2001). Entrainment of the circadian clock in the liver by feeding. Science, 291(5503), 490-493. https://pubmed.ncbi.nlm.nih.gov/11161204/
3. Wehrens, S. M. T., Christou, S., Isherwood, C., Middleton, B., Gibbs, M. A., Archer, S. N., Skene, D. J., & Johnston, J. D. (2017). Meal timing regulates the human circadian system. Current Biology, 27(12), 1768-1775.e3. https://pubmed.ncbi.nlm.nih.gov/28578930/
4. Isherwood, C. M., van der Veen, D. R., Hassanin, H., Skene, D. J., & Johnston, J. D. (2023). Human glucose rhythms and subjective hunger anticipate meal timing. Current Biology, 33(7), 1321-1326.e3. https://pubmed.ncbi.nlm.nih.gov/36822203/
5. Rovira-Llopis, S., Luna-Marco, C., Perea-Galera, L., Bañuls, C., Morillas, C., & Victor, V. M. (2024). Circadian alignment of food intake and glycaemic control by time-restricted eating: A systematic review and meta-analysis. Reviews in Endocrine and Metabolic Disorders, 25(2), 325-337. https://pubmed.ncbi.nlm.nih.gov/37993559/
6. McHill, A. W., & Butler, M. P. (2024). Eating around the clock: Circadian rhythms of eating and metabolism. Annual Review of Nutrition, 44(1), 25-50. https://pubmed.ncbi.nlm.nih.gov/38848598/
7. Peters, B., Schwarz, J., Schuppelius, B., Ottawa, A., Koppold, D. A., Weber, D., Steckhan, N., Mai, K., Grune, T., Pfeiffer, A. F. H., Michalsen, A., Kramer, A., & Pivovarova-Ramich, O. (2025). Intended isocaloric time-restricted eating shifts circadian clocks but does not improve cardiometabolic health in women with overweight. Science Translational Medicine, 17(822), eadv6787. https://pubmed.ncbi.nlm.nih.gov/41160666/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 7 references cited
