The circadian timing network is not one clock — it is a collection of semi-autonomous clocks in every organ, coordinated by a master pacemaker in the brain called the SCN. Rotating night schedules disrupt this coordination from the top: the SCN stays locked to the light-dark cycle and refuses to adjust, while peripheral organs respond to behavioral cues like meal timing and temperature and begin moving off-phase. The result is internal misalignment, not schedule misalignment alone. This article covers the SCN-peripheral desynchronization mechanism, the organ-specific re-entrainment hierarchy, phototherapy as a rescue strategy, and the cardiometabolic disease burden accumulated from chronic rotating schedules. It does not address every health consequence of irregular work hours. For the broader picture of how circadian rhythm disruption drives 3 AM wakeups, see the Circadian Rhythm Disruption overview on circadian sleep disruption.
How Do Rotating Night Schedules Desynchronize Your Peripheral Clocks From Your Brain Clock?
The SCN is entrained primarily by the light-dark cycle, and controlled laboratory and field data show that SCN-driven markers — melatonin and cortisol — do not adjust across successive night rotations (Boivin et al., 2022). The central clock holds its day orientation even when the work schedule demands overnight wakefulness.
This creates a two-tier misalignment. The first tier is between the SCN and peripheral tissue clocks. The second tier emerges between peripheral clocks and the plasma metabolome. Boivin et al. (2022) documented that most rhythmically expressed transcripts remain entrained to a day-oriented schedule with dampened amplitudes, while plasma metabolites move by several hours in a different direction. The metabolome and the transcriptome — both downstream of the SCN — end up on different schedules.
The human quantification of this peripheral dampening comes from Kervezee et al. (2018), who studied 8 healthy subjects using genome-wide blood sampling at baseline and after 4 days of simulated night work (a 10-hour sleep delay). Of the transcripts rhythmically expressed at baseline, 73% remained rhythmic under the night-work condition — but their amplitude dropped from a mean of 0.185 to 0.115 (p < 0.0001). That is a 38% reduction in the strength of peripheral clock gene expression, not a loss of rhythmicity. The distinction matters: the clocks are not stopping. They are running with weaker temporal output.
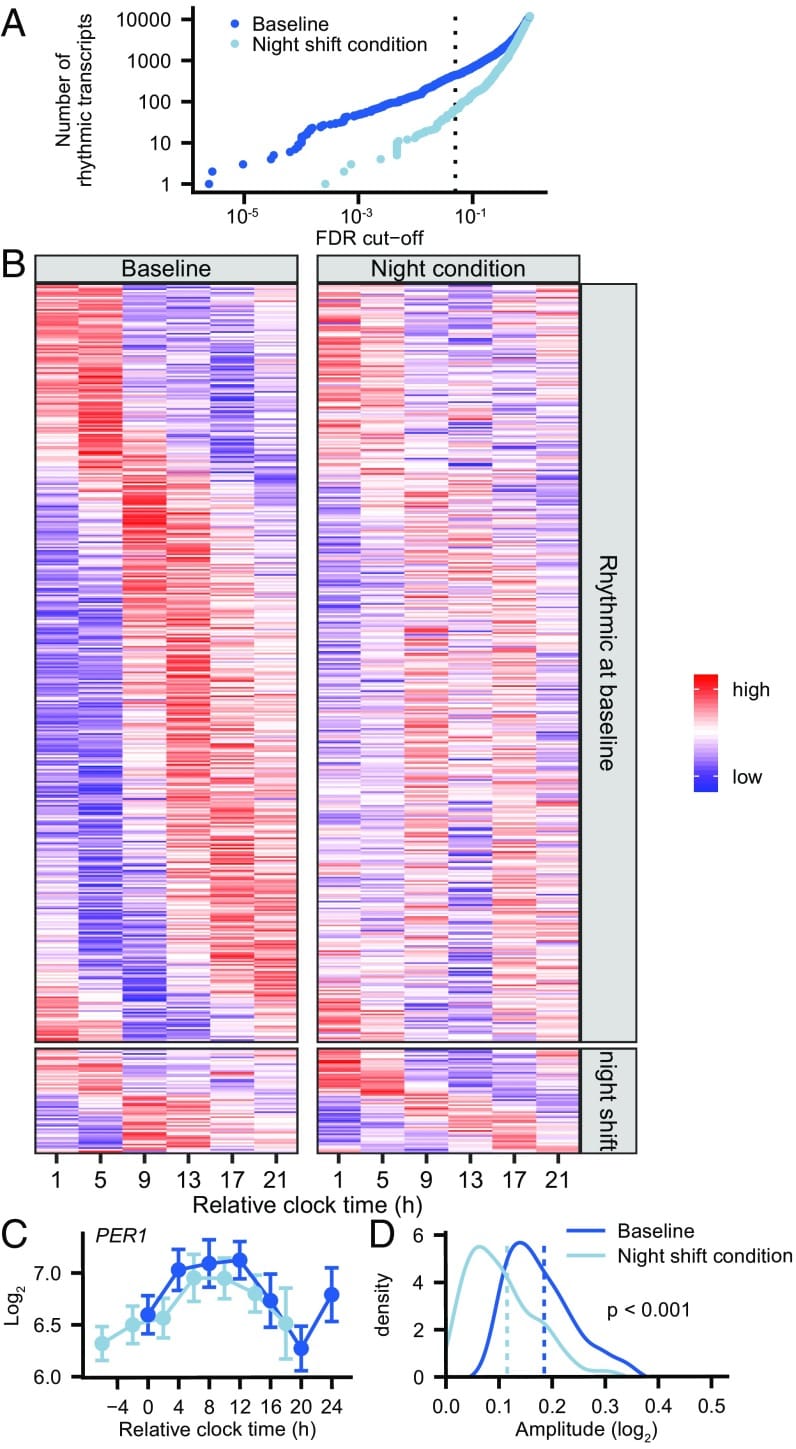
The pathways most affected included natural killer cell-mediated immune response — which accounted for 7 of the 23 phase-delayed transcripts — along with changes in Jun/AP1 signaling and STAT pathway expression (Kervezee et al., 2018) — connecting peripheral clock disruption directly to immune function. This provides a mechanistic basis for the elevated infection and cancer risks observed in long-term rotating-schedule workers.
The misalignment is therefore multilevel: the SCN maintains its day orientation, peripheral clock gene expression dampens while retaining phase, and the metabolome moves in yet another direction. No single “body clock” is off — the entire coordination architecture between central pacemaker, peripheral gene expression, and metabolic output is internally misaligned.
Do All Organs Re-Entrain at the Same Speed After a Schedule Change?
Koritala et al. (2023) tested this hierarchy directly in a mouse model by exposing 72 C57BL/6J mice to four conditions: standard light-dark control, short-term rotating schedules (15 days), long-term rotating schedules (45 days), and chronic jet lag (21 days of repeated 8-hour phase advances). The results revealed a tissue-specific vulnerability gradient.
Under short-term rotating conditions, liver clock gene expression showed no measurable disruption. Skin clock genes, however, were disrupted at the earliest time point measured. This means the skin peripheral clock is more vulnerable to early schedule instability than the liver clock, despite both receiving the same light-dark exposure. Whether this hierarchy translates directly to human tissue remains to be confirmed, but the organ-specific difference in clock vulnerability is consistent with human data showing tissue-dependent re-entrainment rates.
Under long-term rotating conditions (45 days), liver clock gene disruption caught up: 7 of 8 canonical clock genes were affected. In skin, 6 of 8 clock genes were disrupted. The genes showing the highest sensitivity across all conditions were Cry1, Cry2, and BMAL1 (Arntl), with Npas2, Per1, Per2, Per3, and Cry2 showing phase advances (p < 0.05 by CircaCompare analysis). The organ hierarchy matters for understanding which health risks accumulate first — skin-mediated processes may be affected before hepatic ones in workers with shorter rotating-schedule exposure.
Bautista et al. (2025) provide the broader framework for this hierarchy, mapping autonomous circadian oscillators across nine organs and tissues — hippocampus, heart, liver, gut, pancreas, white and brown adipose tissue, adrenal glands, lungs, and skeletal muscle. While the SCN coordinates approximately 20,000 neurons as the master pacemaker, peripheral clocks retain autonomy and can be independently entrained by local zeitgebers.
The mechanism by which behavioral schedule changes reach peripheral clocks — even when the light-dark cycle remains stable — involves zeitgebers spanning several categories described in Bautista et al. (2025): environmental (light, temperature), behavioral (feeding, exercise), hormonal (glucocorticoids, insulin, melatonin), and microbial (short-chain fatty acids, bile acids). Feeding, body temperature, and hormones are potentially conflicting re-entrainment cues that can pull peripheral clocks away from the SCN’s light-entrained phase. This is how internal desynchronization develops organ by organ, with each tissue responding to a different mix of zeitgeber inputs at a different rate.
Can Phototherapy Reset Both Central and Peripheral Clocks During Night Schedules?
Cuesta et al. (2017) assigned 18 healthy subjects to a 6-day simulated night-schedule protocol, with one group receiving bright-light phototherapy (three cycles of 8-hour exposure) and one control group receiving standard indoor lighting.
In the bright-light group, central clock markers responded first: plasma cortisol and melatonin delayed by approximately 7-9 hours (p < 0.001). Peripheral clock genes in peripheral blood mononuclear cells (PBMCs) followed in parallel. PER1 delayed by 7 hours 23 minutes (p < 0.001). PER2 delayed by 9 hours 24 minutes (p < 0.001). PER3 delayed by 8 hours 54 minutes (p < 0.001). REV-ERBa delayed by 6 hours 22 minutes (p < 0.001). BMAL1 advanced by 5 hours 29 minutes (p < 0.05).
In the control group — same schedule, no bright-light exposure — only PER1 and BMAL1 adjusted, and only by approximately 2.5-3 hours (p < 0.05). The remaining peripheral clock genes did not measurably adjust.
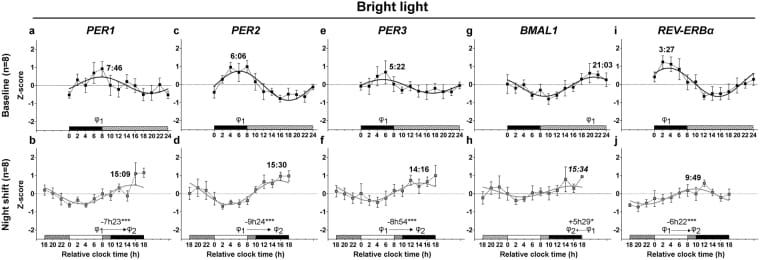
This study provided the first direct human evidence that phototherapy during night schedules can reset both SCN-driven markers and peripheral clock gene expression at the same time. The peripheral clocks followed the central zeitgeber once the central zeitgeber was properly moved — indicating that internal desynchronization is a consequence of conflicting zeitgebers, not an inherent feature of night work.
The practical constraint is timing. Phototherapy moves clocks — the direction of the move depends on when the bright light is applied relative to the individual’s circadian phase. Misapplied bright light at the wrong time can worsen desynchronization rather than correct it. The exposure must be timed to the target schedule, not applied broadly.
What Cardiometabolic Damage Accumulates From Chronic Rotating Schedules?
Ansu Baidoo and Knutson (2023) synthesized epidemiological evidence to quantify the cardiometabolic burden of circadian misalignment from rotating schedules. The findings:
- Obesity: 23% increased likelihood in rotating-schedule workers compared with day workers.
- Type 2 diabetes: 14% elevated risk of incident disease.
- Metabolic syndrome: 11-35% increased odds.
- Hypertension: 10% increased odds of prevalent hypertension and 30% increased odds of incident hypertension. Blood pressure was elevated across included studies.
These numbers come from large-scale epidemiological data, not small pilot studies.
The chronotype-misalignment data from the same review adds a second dimension. Evening chronotypes — people whose internal clock runs late — showed higher fasting glucose (mean difference 7.82 mg/dL; 95% CI: 3.18-12.45; 8 studies) and elevated HbA1c (mean difference 7.6; 95% CI: 3.1-12.2) compared with morning chronotypes. This occurred independent of formal work-schedule status. The internal clock-behavior mismatch alone, without any work-schedule component, produced measurable metabolic consequences.
Meal timing emerges as an independent driver in the same review. Late-night eating was associated with elevated metabolic syndrome prevalence (OR 1.25; 95% CI: 1.04-1.49), consistent with the peripheral clock sensitivity to food timing described in the organ re-entrainment hierarchy above. Sleep timing variability was associated with elevated BMI and HOMA-IR, and sleep irregularity was linked to higher diabetes prevalence and incident cardiovascular events over approximately 5 years in the MESA cohort.
For adults over 40 managing cardiometabolic health who work variable schedules, these risks represent measurably elevated baseline risk, quantified across tens of thousands of study participants.
Sleep disruption from rotating schedules is rarely a single-cause problem. Circadian misalignment from irregular work hours may compound with inflammatory, metabolic, hormonal, or autonomic factors — especially in adults managing health conditions alongside irregular schedules. Identifying which causes might be contributing is a useful next step.
Find out which causes might be driving your 3 AM wakeups →
Frequently Asked Questions
Does Internal Desynchronization Explain Why Some Rotating-Schedule Workers Tolerate Irregular Hours and Others Cannot?
Reinberg and Ashkenazi (2008) reanalyzed time-series data from 48 male French rotating-schedule workers divided into good tolerators (n = 14), poor tolerators (n = 19), and former rotating-schedule workers who returned to day work (n = 15).
Tolerant workers were older (mean age 44.9 +/- 2.1 vs. 40.1 +/- 2.6 years) and had longer shift-work experience. This suggests physiological adaptation or self-selection over time — workers whose internal clocks cannot maintain coherence under rotating conditions may leave those schedules earlier.
Non-tolerant workers presented with sleep disturbance, persistent fatigue, mood changes, and medication dependency. The internal desynchronization — rhythms whose free-running period deviated from 24 hours — was mechanistically linked to pathology, not a secondary consequence of the health effects.
Former rotating-schedule workers who returned to day schedules were assessed 1.5 to 20 years after the transition and showed resolution of circadian desynchronization and associated health effects. The desynchronization was reversible, but the recovery timeline varied widely between individuals.
Can Meal Timing Reduce Circadian Disruption From Rotating Schedules?
Bautista et al. (2025) identify feeding as a behavioral zeitgeber category, alongside light, temperature, exercise, and hormonal cues. Peripheral clocks in the gut, liver, and pancreas are particularly sensitive to food timing.
A practical approach is to align meal timing with the daylight schedule even during overnight work hours rather than eating primarily at night. This targets the peripheral clock network with a consistent zeitgeber — a strategy supported by the tissue-specific vulnerability described in the organ re-entrainment hierarchy above. Peripheral clocks respond to food timing independently of the light-dark cycle, meaning mealtime provides a separate entrainment channel that can either reinforce or conflict with SCN-driven coordination.
Meal timing does not substitute for phototherapy in resetting the central clock — the SCN responds primarily to light, not food. But restricting food intake to daytime hours can reduce the degree of peripheral desynchronization independently, limiting the extent to which gut, liver, and pancreatic clocks diverge from the SCN’s maintained day orientation.
How Long Does It Take Peripheral Clocks to Re-Synchronize After a Schedule Change?
Bautista et al. (2025) document that peripheral clocks retain autonomy and respond to local zeitgebers (feeding, temperature, hormones) that may work on different timescales than light. The SCN can adjust its phase within a few days when given a consistent light-dark cue — but downstream organs wait for their own relevant zeitgebers to arrive, and those zeitgebers may not all align at the same time.
From Koritala et al. (2023), skin clock genes are more vulnerable to short-term rotating-schedule disruption than liver clock genes in mice, which suggests that skin re-entrainment may also follow a different timeline during recovery. Liver clock recovery after 45-day rotating-schedule disruption — with 7 of 8 genes affected — implies that sustained exposure accumulates disruption that cannot be rapidly reversed.
Reinberg and Ashkenazi (2008) documented the longest-term recovery data: former rotating-schedule workers who returned to day schedules were assessed 1.5 to 20 years after the transition, showing resolution of circadian desynchronization. The wide range underscores that recovery is not a fixed timeline. The duration depends on the extent of accumulated desynchronization, individual physiology, and the consistency of zeitgeber inputs during recovery.
Related Reading
- Circadian Rhythm Disruption: How a Misaligned Internal Clock Fragments Sleep, Drives 3AM Wakeups, and Reduces Sleep Depth — the full overview of circadian sleep disruption, including melatonin, cortisol, body temperature, peripheral clocks, and early-morning waking
- How Does a Two-Hour Weekend Sleep Delay Desynchronize Your Peripheral Clocks for Days? — how repeated weekend schedule shifts create chronic circadian misalignment
- Why Does Eastbound Jet Lag Last Longer Than Westbound — and What Determines Recovery Rate? — why travel direction changes the speed of circadian recovery
- Night Owl or Delayed Sleep Phase Disorder? How to Know the Difference — how delayed circadian timing differs from preference alone
- Why Does Your Wake Time Advance by Decades — and Can You Reverse Circadian Phase Advance After 60? — why aging shifts wake time earlier and which parts remain reversible
- Which Circadian Mechanisms Change After 40 — and Which Ones Are Reversible? — the broader aging mechanisms that shift circadian timing and sleep depth
- Does Evening Exercise Delay Your Circadian Clock — or Is Disruption Smaller Than You Think? — how exercise timing affects the circadian clock and sleep onset
- Does Time-Restricted Eating Entrain Your Peripheral Clocks? — how meal timing signals peripheral clocks outside the brain
References
1. Ansu Baidoo, V., & Knutson, K. (2023). Associations between circadian disruption and cardiometabolic disease risk: A review. Obesity, 31(3), 615-624. https://pubmed.ncbi.nlm.nih.gov/36750239/
2. Bautista, J., Ojeda-Mosquera, S., Ordoñez-Lozada, D., & López-Cortés, A. (2025). Peripheral clocks and systemic zeitgeber interactions: From molecular mechanisms to circadian precision medicine. Frontiers in Endocrinology, 16, 1606242. https://pubmed.ncbi.nlm.nih.gov/40510487/
3. Boivin, D. B., Boudreau, P., & Kosmadopoulos, A. (2022). Disturbance of the circadian system in shift work and its health impact. Journal of Biological Rhythms, 37(1), 3-28. https://pubmed.ncbi.nlm.nih.gov/34969316/
4. Cuesta, M., Boudreau, P., Cermakian, N., & Boivin, D. B. (2017). Rapid resetting of human peripheral clocks by phototherapy during simulated night shift work. Scientific Reports, 7, 16310. https://pubmed.ncbi.nlm.nih.gov/29176713/
5. Kervezee, L., Cuesta, M., Cermakian, N., & Boivin, D. B. (2018). Simulated night shift work induces circadian misalignment of the human peripheral blood mononuclear cell transcriptome. Proceedings of the National Academy of Sciences, 115(21), 5540-5545. https://pubmed.ncbi.nlm.nih.gov/29735673/
6. Koritala, B. S. C., Dakup, P., Porter, K. I., & Gaddameedhi, S. (2023). The impact of shift-work light conditions on tissue-specific circadian rhythms of canonical clock genes: Insights from a mouse model study. F1000Research, 12, 762. https://pubmed.ncbi.nlm.nih.gov/37576540/
7. Reinberg, A., & Ashkenazi, I. (2008). Internal desynchronization of circadian rhythms and tolerance to shift work. Chronobiology International, 25(4), 625-643. https://pubmed.ncbi.nlm.nih.gov/18622820/
Written by Kat Fu, M.S., M.S. · Last reviewed: May 2026 · 7 references cited
