By the time many men reach their 50s, consolidated 7-8 hour sleep has become rare. Orexin (also called hypocretin), the neuropeptide that stabilizes wakefulness, undergoes two opposing changes with aging that together destabilize sleep architecture.
The first change: orexin-producing neurons die. Postmortem human studies document a measurable decline in orexin neuron count with age. The second change: the surviving neurons become hyperexcitable — they fire more readily than they did in younger brains, making it easier for minor disturbances to trigger a full wake episode during sleep.
Below: orexin neuron loss, orexin neuron hyperexcitability, and how these interact with circadian amplitude decline to fragment sleep after midlife. For the broader picture of what orexin does, see What Is Orexin and Why Does It Keep You Awake?. For the full cause overview, see the circadian pillar.
How Many Orexin Neurons Do You Lose With Age?
The largest human postmortem study on age-related orexin neuron loss comes from Hunt et al. (2015), who examined hypothalamic tissue from individuals across a wide age range. They found a 23% decrease in orexin-immunoreactive neurons between younger and older adults. The loss was not evenly distributed — it concentrated in the dorsomedial and perifornical regions of the hypothalamus, the areas that receive direct circadian timing input from the suprachiasmatic nucleus (the brain’s master clock).
Earlier work by Kessler et al. (2011) established that orexin neuron attrition is a normal part of aging, not exclusively a disease process. Their rodent data showed more than 40% orexin neuron loss in aged rats, while human research has shown a similar trajectory at a lower magnitude. This was an important distinction: before this work, orexin neuron loss was primarily associated with narcolepsy, a condition involving 85-95% destruction of the orexin neuron population.
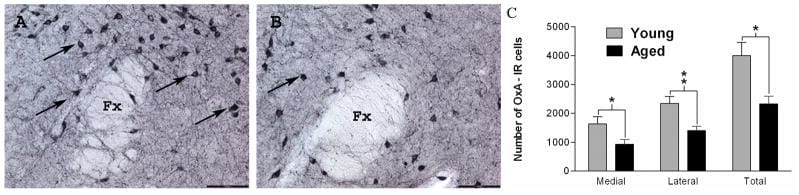
What does a 23% loss mean functionally? It is not narcolepsy — that requires near-total orexin neuron destruction. A 23% reduction measurably weakens the brain’s ability to hold a stable wake state. Orexin neurons do not just promote wakefulness — they stabilize it, preventing unwanted transitions between sleep and wake states. With fewer neurons maintaining that stability, the threshold for transitioning from sleep to wake drops, and each sleep cycle boundary becomes a point where arousal can intrude.
Neuron count, however, is not the whole story. The neurons that survive undergo their own changes — and those changes may matter more than the loss itself.
Why Do Remaining Orexin Neurons Become Hyperexcitable With Age?
A 2022 paper published in Science by Li, de Lecea, and colleagues provided the first mechanistic explanation for age-related sleep fragmentation tied to orexin neuron activity. Using fiber photometry (a technique that records calcium activity from specific neurons in live, behaving animals), they tracked orexin neuron firing across sleep-wake states in young and aged mice.
Aged mice showed higher spontaneous orexin neuron activity during sleep. When the researchers used optogenetic stimulation (activating orexin neurons with light pulses via a genetically encoded light-sensitive channel), aged mice produced longer wake bouts from the same stimulation intensity — their orexin neurons responded more aggressively to the same input.
The mechanism traced to KCNQ2/3 potassium channels. These channels produce the M-current, a stabilizing electrical current that keeps neurons from firing too easily. In aged orexin neurons, KCNQ2/3 channel expression was reduced, and the M-current was impaired. Without this restraint, orexin neurons sat closer to their firing threshold, ready to activate at smaller provocations.
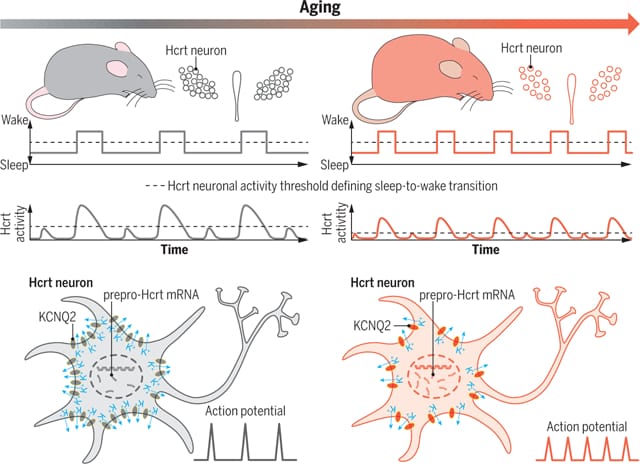
Two experiments made the causal link convincing. First, the researchers used CRISPR to knock out Kcnq2/3 genes specifically in orexin neurons of young mice. These young mice developed the same sleep fragmentation pattern seen in aged animals — frequent, brief wake episodes interrupting NREM sleep. The fragmentation was not a general aging effect; it was reproduced by disabling a single channel type in a single neuron population. Second, administering flupirtine (a KCNQ channel opener) to aged mice restored M-current function and reversed their sleep fragmentation, returning their sleep architecture closer to that of young animals.
This creates a counterintuitive picture. In narcolepsy, too few orexin neurons means the brain cannot sustain wakefulness — the result is excessive sleepiness and sudden sleep attacks. In aging, there are fewer orexin neurons, but the survivors fire too easily — the result is fragmented sleep because the brain cannot sustain sleep through normal cycle transitions. The same neuropeptide, two opposite outcomes depending on whether the problem is neuron loss alone or neuron loss combined with survivor hyperexcitability.
The connection to the 2-hour wake cycle that many middle-aged and older adults report is direct. Normal sleep cycles last roughly 90-120 minutes. At each cycle boundary, sleep becomes briefly lighter. If the threshold for a sleep-to-wake transition has dropped because of hyperexcitable orexin neurons, these natural cycle boundaries become points where full arousal occurs instead of smooth continuation into the next cycle.
One caveat: this mechanistic data comes from mice. No equivalent human tissue study has confirmed KCNQ2/3 impairment in aged human orexin neurons. The 23% neuron loss described in the previous section is from human postmortem data; the hyperexcitability mechanism is from animal research. The mouse findings are consistent with human sleep fragmentation patterns, but the direct translation has not been established.
How Does Orexin Decline Interact With Other Age-Related Circadian Changes?
Mander, Winer, and Walker (2017), in a comprehensive review published in Neuron, documented the scope of age-related sleep deterioration. Slow-wave sleep (the deepest stage of NREM sleep) drops from approximately 19% of total sleep time in young adults to as low as 3.4% by midlife in some studies. This decline begins in the late 20s and accelerates after 50.
Orexin neuron loss and hyperexcitability are two factors within a larger set of age-related circadian changes that compound each other:
- Weakened suprachiasmatic nucleus output. The suprachiasmatic nucleus loses neurons with age, and the amplitude of its circadian output declines. Because the suprachiasmatic nucleus normally provides precise timing input to the orexin neuron population — activating orexin during the day and withdrawing activation at night — a weaker suprachiasmatic nucleus means less precise gating of orexin neuron activity. The result is orexin activity that bleeds into the sleep period.
- Reduced melatonin production. Pineal calcification reduces melatonin synthesis with age. Melatonin suppresses orexin neuron firing, so lower melatonin at night means less restraint on orexin activity during the sleep window, increasing the probability of wake intrusions.
- Cortisol phase advance. The cortisol rhythm advances earlier with age, meaning cortisol levels begin rising in the early morning hours — often 1-2 hours earlier than in younger adults. Since cortisol promotes wakefulness and can activate orexin neurons, an earlier cortisol rise contributes to the early-morning waking pattern common after midlife.
Each of these changes is individually documented. Their compounding is what makes age-related sleep fragmentation so resistant to single approaches: addressing one factor (for example, supplementing melatonin) may not overcome the combined effect of the others.
Is Orexin Linked to Neurodegeneration?
Liguori et al. (2020) reported that cerebrospinal fluid orexin-A levels correlated with tau pathology and amyloid-beta42 levels in individuals with Alzheimer’s disease. Higher orexin-A in cerebrospinal fluid was associated with greater Alzheimer’s-related protein accumulation. This is an association finding, not a proven causal relationship — it is not established whether orexin changes contribute to neurodegeneration or are a consequence of it. For individuals focused on long-term cognitive health, the connection between sleep fragmentation, orexin changes, and Alzheimer’s biomarkers is an active area of research.
Do Dual Orexin Receptor Antagonists Help Older Adults Sleep?
Gotfried et al. (2024) published a pooled analysis of lemborexant placebo-controlled trials in adults aged 65 and older. The results showed improvements in both sleep onset and wake after sleep onset (the total time spent awake during the night after initially falling asleep). In this age group, lemborexant did not impair next-day memory, balance, or attention — risks that are well-documented with benzodiazepines and Z-drugs in older populations. Twelve-month safety data showed a consistent profile without tolerance development.
The trial data demonstrates that the orexin component of age-related fragmentation is pharmacologically modifiable — the fragmentation is not a fixed, irreversible consequence of aging. That said, orexin receptor antagonists are one tool among several, and individual responses vary.
Age-related orexin changes are one of several circadian mechanisms that can fragment sleep after midlife. Cortisol phase advance, melatonin decline, peripheral clock decoupling, and hormonal changes might also be contributing. Identifying which causes might be active gives a more complete picture than addressing any single factor.
What Else Do People Ask About Orexin and Aging?
Does Aging Mean Fragmented Sleep Is Normal?
The distinction matters. “Common” and “inevitable” are not the same thing. Many age-related changes in sleep architecture — lighter sleep, reduced slow-wave sleep, more frequent brief awakenings — do appear to be part of normal aging physiology. The severity of fragmentation that leads people to wake up at 2am or 3am and remain awake for an hour or more may involve addressable contributors on top of that baseline.
The Li et al. (2022) KCNQ2/3 data showed that pharmacologically restoring channel function reversed fragmentation in aged mice. The Gotfried et al. (2024) lemborexant data showed that blocking orexin receptors improved sleep maintenance in adults over 65 without next-day cognitive costs. Both findings indicate that at least a portion of age-related sleep fragmentation involves mechanisms that can be influenced.
Can Orexin Impairment Contribute to Cognitive Decline?
Liguori et al. (2020) measured cerebrospinal fluid orexin-A alongside Alzheimer’s biomarkers and found that higher orexin-A levels correlated with greater tau and amyloid-beta42 accumulation. One hypothesis is that elevated orexin-A increases wakefulness and reduces the glymphatic clearance of metabolic waste that occurs during deep sleep — but this remains speculative.
What makes this relevant for individuals focused on longevity: sleep fragmentation and neurodegeneration may share upstream circadian drivers. If weakened suprachiasmatic nucleus output and orexin impairment both contribute to fragmented sleep and correlate with Alzheimer’s biomarkers, addressing the circadian contributors may have implications beyond sleep quality alone. This research is still in early stages, and causal claims are not supported by current data.
Are Dual Orexin Receptor Antagonists Safer Than Traditional Sleep Medications for Older Adults?
The Gotfried et al. (2024) pooled analysis of lemborexant trials included 12-month safety data in adults 65 and older. The medication improved both time to fall asleep and wake after sleep onset. It did not increase fall risk, impair next-day balance, or reduce next-day cognitive performance — the three primary safety concerns with sleep medications in older adults.
Kishi et al. (2025) conducted a network meta-analysis comparing dual orexin receptor antagonists to other sleep medication classes, finding favorable efficacy and tolerability profiles for orexin receptor antagonists in adults across age groups.
Side effects do occur with dual orexin receptor antagonists. Vivid or unusual dreams are the commonly reported side effect, along with next-morning drowsiness in some individuals. These side effects are generally milder than the cognitive impairment and fall risk associated with older medication classes, but they are not trivial.
This article does not recommend specific medications. For a detailed comparison of how orexin receptor antagonists work compared to other sleep medication classes, see How Do Orexin-Blocking Sleep Medications Compare to Ambien?.
Related Reading
- Circadian Rhythm Disruption and Sleep — The full cause reference page covering all five circadian mechanisms
- What Is Orexin and Why Does It Keep You Awake When Your Body Clock Says Sleep? — How the circadian clock controls orexin
- How Does Melatonin Suppress Orexin? — Melatonin-orexin opposition
- How Does Stress Hijack Your Orexin? — Stress-driven orexin overactivation
- Can You Lower Orexin Naturally? — Lifestyle approaches for orexin modulation
- How Do Orexin-Blocking Sleep Medications Compare to Ambien? — Mechanism comparison
References
1. Hunt, N. J., Rodriguez, M. L., Waters, K. A., & Machaalani, R. (2015). Changes in orexin (hypocretin) neuronal expression with normal aging in the human hypothalamus. Neurobiology of Aging, 36(1), 292-300. PMID: 25212464. https://pubmed.ncbi.nlm.nih.gov/25212464/
2. Kessler, B. A., Stanley, E. M., Frederick-Duus, D., & Fadel, J. (2011). Age-related loss of orexin/hypocretin neurons. Neuroscience, 178, 82-88. PMID: 21262323. https://pubmed.ncbi.nlm.nih.gov/21262323/
3. Li, S.-B., et al. (2022). Hyperexcitable arousal circuits drive sleep instability during aging. Science, 375(6583), eabh3021. PMID: 35201886. https://pubmed.ncbi.nlm.nih.gov/35201886/
4. Mander, B. A., Winer, J. R., & Walker, M. P. (2017). Sleep and human aging. Neuron, 94(1), 19-36. PMID: 28384471. https://pubmed.ncbi.nlm.nih.gov/28384471/
5. Liguori, C., et al. (2020). Sleep-wake cycle in Alzheimer’s disease is associated with tau pathology and orexin dysregulation. Journal of Alzheimer’s Disease, 74(2), 501-508. PMID: 32065791. https://pubmed.ncbi.nlm.nih.gov/32065791/
6. Gotfried, M. H., et al. (2024). Efficacy and safety of insomnia treatment with lemborexant in older adults: analyses from three clinical trials. Drugs & Aging, 41(9), 757-772. PMID: 39120786. https://pubmed.ncbi.nlm.nih.gov/39120786/
7. Kishi, T., et al. (2025). Comparative efficacy and safety of daridorexant, lemborexant, and suvorexant for insomnia: a systematic review and network meta-analysis. Translational Psychiatry, 15, 211. PMID: 40555730. https://pubmed.ncbi.nlm.nih.gov/40555730/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
