Orexin is the brain’s primary wake-stabilizing neuropeptide. Newer sleep medications — suvorexant, lemborexant, daridorexant — work by blocking orexin receptors. But orexin neurons are not autonomous — they respond to metabolic and environmental inputs as they change, and several of those inputs are modifiable.
This article covers the specific inputs that raise and lower orexin neuron activity, with evidence from animal and human studies. Each input represents a timing decision: when to eat, when to exercise, when to expose yourself to light. The cumulative effect of these decisions determines how much orexin-driven arousal is present at bedtime.
Orexin modulation through lifestyle timing is part of broader circadian regulation. For the full picture of how circadian mechanisms fragment sleep, see the parent pillar on Circadian Rhythm Disruption and Sleep.
How Does Blood Glucose Affect Orexin?
Orexin neurons are glucose sensors. This has been established at the single-neuron level: when blood glucose rises, orexin neurons reduce their firing rate. When glucose falls, orexin neurons become more active. This makes orexin part of a metabolic feedback loop — the brain’s wake drive increases when energy availability drops.
Burdakov et al. (2006) identified the molecular mechanism. Using patch-clamp recordings from orexin neurons in mouse brain slices, the researchers showed that glucose inhibits orexin neurons through tandem-pore potassium (K2P) channels. When glucose concentration increases at the neuron’s surface, an intracellular messenger activates these K2P channels, hyperpolarizing the neuron and reducing its firing rate. The intermediate messenger is not ATP, calcium, or glucose itself — it works through a pathway distinct from the pancreatic beta-cell glucose-sensing mechanism. This was one of the first demonstrations that orexin neurons could track physiologically relevant glucose fluctuations, not just extreme concentrations.
Viskaitis et al. (2024) extended this to behaving animals. Using two-photon calcium imaging of 913 individual orexin neurons in freely moving mice alongside continuous blood glucose telemetry, the researchers found that 98% of orexin neurons responded to blood glucose changes. Many of these neurons did not track absolute glucose concentration — they tracked the rate of glucose change. The maximal orexin neuron response preceded the glucose peak by several minutes, meaning orexin neurons were suppressed when glucose was rising fastest, not when it was highest. Additionally, 65% of orexin neurons multiplexed glucose and locomotion information, integrating metabolic state with physical activity.
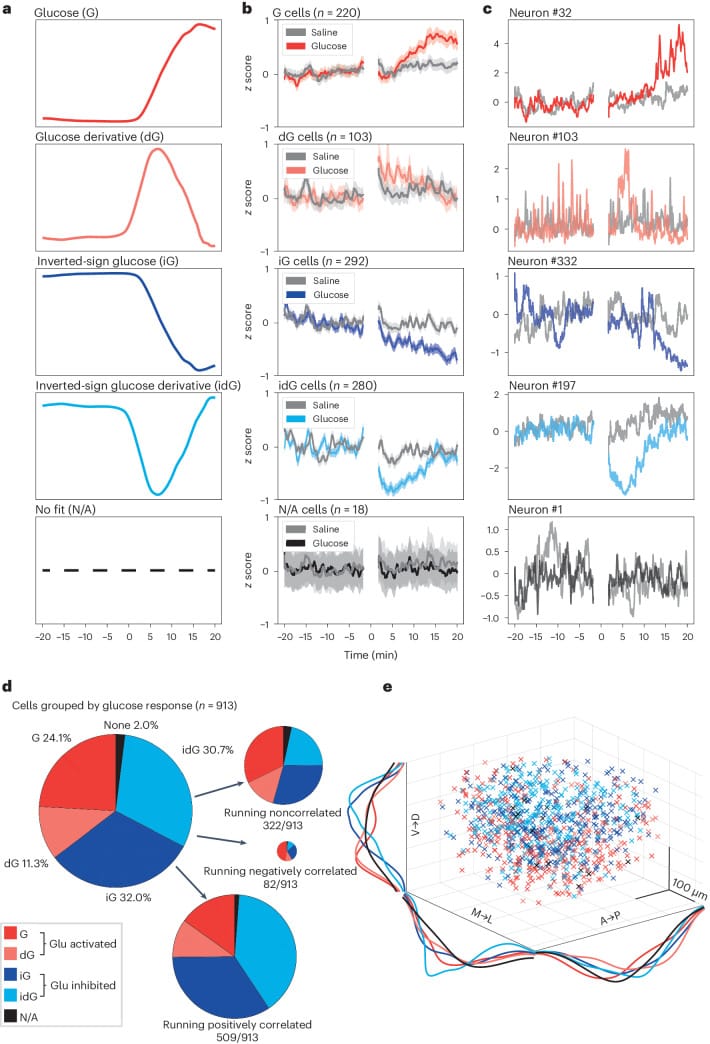
Leptin — the satiety hormone released by fat tissue — provides an additional glucose-aligned orexin suppression pathway. Goforth et al. (2014) showed that leptin does not inhibit orexin neurons directly. Instead, leptin activates neurotensin-containing neurons in the lateral hypothalamus that express the leptin receptor. These neurotensin neurons then suppress orexin firing through two GABA-independent mechanisms: reducing excitatory input to orexin neurons, and activating ATP-sensitive potassium channels that hyperpolarize orexin cells. The neuropeptide galanin, co-expressed with neurotensin, was required for this effect — blocking galanin receptors prevented leptin-driven orexin suppression.
The practical implication is consistent across these findings. After a meal, blood glucose rises and leptin increases — both suppress orexin neuron activity, reducing wake drive. Going to bed with low blood glucose (from skipped meals or prolonged fasting) keeps orexin neurons active, because the metabolic input that suppresses them is absent. A carbohydrate-containing snack before bed raises glucose, which — based on the Burdakov and Viskaitis findings — suppresses orexin firing through the K2P channel mechanism. The rate-of-change finding from Viskaitis et al. suggests the suppression is strongest during the rising phase of glucose, not at the peak.
How Does Light Exposure Control Orexin Timing?
Light does not affect orexin neurons directly. The suprachiasmatic nucleus (SCN) — the brain’s master circadian clock — receives light input from the retina and uses that input to gate when orexin neurons fire. During the circadian day, when light activates the SCN, the SCN suppresses orexin neuron activity. At dark onset, as SCN output decreases, orexin neurons are released from inhibition and become active.
This is counterintuitive. The SCN does not turn orexin on with light — it holds orexin in check during light, and releases it at dark onset.
Marston et al. (2008) demonstrated this relationship using c-Fos immunostaining (a marker of recent neuronal activation) in mice maintained under constant light conditions. Orexin neuron activation followed a circadian pattern, peaking during the subjective night — the animal’s endogenous dark period. When the researchers introduced a 6-hour dark pulse during the subjective day, this dark pulse suppressed SCN activity and activated orexin neurons in the tuberal hypothalamus. The effect was disinhibition: removing light input reduced SCN firing, which released orexin neurons from SCN-mediated suppression. Simultaneously, the dark pulse activated neurons in the median raphe nucleus, whose outputs further suppressed the SCN.
For humans, the SCN-orexin gating mechanism — established in animal models — has two practical implications. SCN entrainment by light is well-documented in humans, and the orexin gating pattern observed in mice is expected to apply given the conserved anatomy of the lateral hypothalamus. Morning bright light exposure entrains the SCN to the local light-dark cycle, which keeps orexin firing on its normal circadian schedule — peaking in the late afternoon and declining at night. Evening screen light or bright indoor light after sunset delays the SCN’s transition into its nighttime mode. If the SCN remains in its daytime pattern, it continues suppressing orexin through its normal daytime gating — but the downstream effect is that orexin’s scheduled release into its nighttime decline is delayed. The result is that the natural evening wind-down of orexin-driven arousal occurs later than it should relative to the person’s intended bedtime.
The timing of light exposure matters more than the intensity. Consistent morning light and dim evenings keep the SCN entrained so that the SCN-to-orexin gating occurs at the right time — orexin active during the day, declining in the evening, suppressed during sleep.
Does Exercise Timing Affect Orexin Activity?
Physical activity increases orexin neuron firing. This is consistent with orexin’s role as a wake-stabilizing neuropeptide — during movement, the brain benefits from sustained arousal and alertness.
Nigro et al. (2025) reviewed evidence that physical activity stimulates orexin release, enhancing neuroplasticity and cognitive performance. The orexin response to exercise is acute: it occurs during and shortly after activity. This orexin activation is part of why exercise tends to improve alertness and focus — the same wake-promoting neuropeptide that fragments sleep when active at night supports performance when active during the day.
Fasting amplifies orexin activity through a related mechanism. Almeneessier, Alzoghaibi, BaHammam et al. (2018) measured plasma orexin-A concentrations in eight healthy volunteers during Ramadan — a period of daytime fasting followed by evening eating. Orexin-A levels increased during fasting hours compared to non-fasting baseline measurements. At night, orexin-A concentrations decreased. This pattern aligns with the metabolic sensing function: low blood glucose during fasting keeps orexin neurons active (no glucose-mediated K2P channel suppression), while evening food intake raises glucose and suppresses orexin as the sleep period approaches.
The exercise-orexin relationship creates a timing consideration. Exercise earlier in the day — particularly in the morning — raises orexin during hours when orexin should be high, reinforcing the circadian wake drive. Exercise close to bedtime raises orexin during hours when orexin should be declining. A post-workout carbohydrate-containing meal provides the glucose input that suppresses orexin firing, helping transition from exercise-driven arousal back toward baseline.
The combination of morning exercise followed by breakfast produces two aligned effects: orexin activation from physical activity (appropriate for the morning wake phase) followed by orexin suppression from rising blood glucose (which resets the metabolic baseline). This combination uses both the exercise and glucose inputs in the direction that matches circadian timing.
Can Apigenin Act as a Natural Orexin Modulator?
Apigenin is a flavonoid found in chamomile, parsley, celery, and several other plants. It has received attention as a potential sleep-supporting compound through multiple mechanisms — not all of which are well-characterized in humans.
Kramer & Johnson (2024) reviewed apigenin’s molecular interactions relevant to sleep and aging. Their primary finding was that apigenin inhibits CD38, a glycoprotein expressed on immune cells that consumes NAD+ by converting it to ADP-ribose and cyclic ADP-ribose. By inhibiting CD38, apigenin increases the available pool of NAD+ — a coenzyme involved in circadian clock function and cellular repair processes that occur during sleep. Separately, they reported that apigenin demonstrated GABAergic activity in rats that was independent of GABA-benzodiazepine receptors and may be mediated through the GABA-A receptor directly. The review also identified an inverse correlation between dietary apigenin intake and poor sleep quality in a large cohort of adults — though this is observational data, not causal evidence.
A computational gene expression analysis has suggested that apigenin’s molecular profile resembles that of an orexin receptor (OX2R) antagonist. This comes from database-level comparison of gene expression signatures, not from direct binding assays or receptor pharmacology studies. No published research has validated that apigenin binds to or blocks OX2R in human tissue. The observation is an emerging area of investigation that remains unvalidated by direct experimental evidence.
Chamomile tea is the traditional application of apigenin for sleep. The apigenin content in a cup of chamomile tea is relatively low — concentrated extracts deliver more, but dosing has not been standardized in controlled trials for sleep outcomes. The CD38 inhibition and GABAergic effects observed in animal studies provide mechanistic plausibility, but translating these to specific human dosing recommendations is premature.
Orexin is one of several circadian mechanisms that can fragment sleep. Melatonin timing, cortisol phase advance, peripheral clock decoupling, metabolic or hormonal factors might also be contributing. Identifying which causes might be active is a useful next step.
Find out which causes might be driving your 3am wakeups →
Frequently Asked Questions
Does Fasting Raise or Lower Orexin?
Can Eating Before Bed Help With Sleep?
Does Temperature Affect Orexin?
Related Reading:
- Circadian Rhythm Disruption and Sleep — The full cause reference page covering all five circadian mechanisms
- What Is Orexin and Why Does It Keep You Awake When Your Body Clock Says Sleep?
- How Does Orexin Change With Age? Why Sleep Fragments More After 50
- How Melatonin Suppresses Orexin: The Push-Pull That Decides Whether You Sleep or Stay Awake
- How Stress Hijacks Your Orexin: Why “Wired but Tired” Is a Real Brain State
- DORAs vs. Ambien: How Orexin-Blocking Sleep Medications Work Differently in Your Brain
References
Almeneessier, A. S., Alzoghaibi, M., BaHammam, A. A., Ibrahim, M. G., Olaish, A. H., Nashwan, S. Z., & BaHammam, A. S. (2018). The effects of diurnal intermittent fasting on the wake-promoting neurotransmitter orexin-A. Annals of Thoracic Medicine, 13(1), 48–54. https://pubmed.ncbi.nlm.nih.gov/29387256/
Burdakov, D., Jensen, L. T., Alexopoulos, H., Williams, R. H., Fearon, I. M., O’Kelly, I., Gerasimenko, O., Fugger, L., & Verkhratsky, A. (2006). Tandem-pore K+ channels mediate inhibition of orexin neurons by glucose. Neuron, 50(5), 711–722. https://pubmed.ncbi.nlm.nih.gov/16731510/
Goforth, P. B., Leinninger, G. M., Patterson, C. M., Satin, L. S., & Myers, M. G. Jr. (2014). Leptin acts via lateral hypothalamic area neurotensin neurons to inhibit orexin neurons by multiple GABA-independent mechanisms. Journal of Neuroscience, 34(34), 11405–11415. https://pubmed.ncbi.nlm.nih.gov/25143620/
Kramer, D. J., & Johnson, A. A. (2024). Apigenin: a natural molecule at the intersection of sleep and aging. Frontiers in Nutrition, 11, 1359176. https://pubmed.ncbi.nlm.nih.gov/38476603/
Marston, O. J., Williams, R. H., Canal, M. M., Samuels, R. E., Upton, N., & Piggins, H. D. (2008). Circadian and dark-pulse activation of orexin/hypocretin neurons. Molecular Brain, 1, 19. https://pubmed.ncbi.nlm.nih.gov/19055781/
Nigro, E., Argentino, F., Musumeci, G., & Daniele, A. (2025). Orexin and lifestyle habits: a meaningful connection among nutrition, physical activity, and sleep pattern in health and diseases. International Journal of Molecular Sciences, 26(18), 8980. https://pubmed.ncbi.nlm.nih.gov/41009546/
Viskaitis, P., Tesmer, A. L., Liu, Z., Karnani, M. M., Arnold, M., Donegan, D., Bracey, E. F., Grujic, N., Patriarchi, T., Peleg-Raibstein, D., & Burdakov, D. (2024). Orexin neurons track temporal features of blood glucose in behaving mice. Nature Neuroscience, 27(7), 1299–1308. https://pubmed.ncbi.nlm.nih.gov/38773350/
Written by Kat Fu, M.S., M.S. · Last reviewed: April 2026 · 7 references cited
